
Elliott Management Takes Significant Stake in Dexcom, Pushes for Board Refresh
Dexcom gains Elliott stake; will add board members and restructure, betting on CGM growth; shares rose 6%.

Dexcom gains Elliott stake; will add board members and restructure, betting on CGM growth; shares rose 6%.
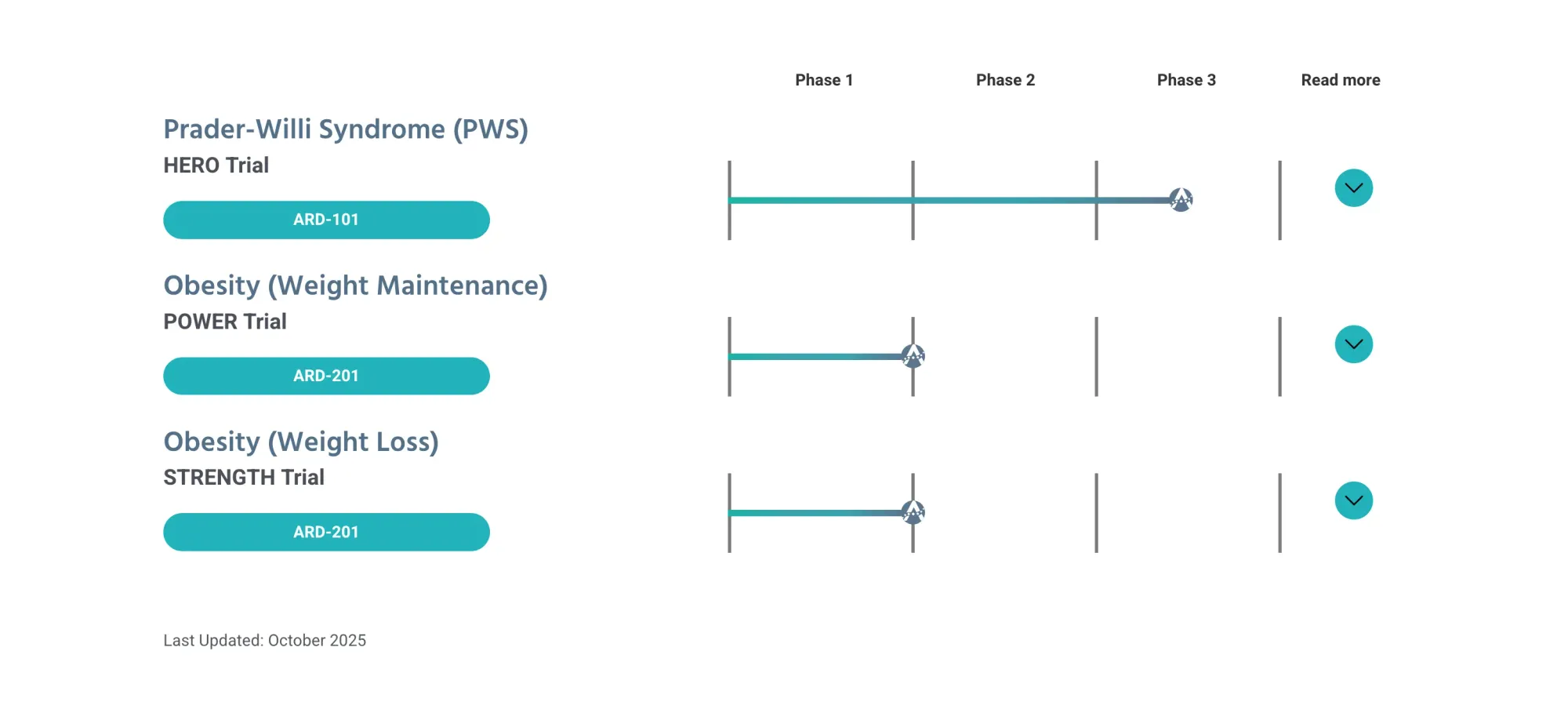
FDA pauses Aardvark’s PWS drug trials over safety concerns; studies stop, shares fall, talks with FDA ongoing.
FDA gave Fast Track to INmune Bio’s XPro for early Alzheimer’s, speeding trials of a drug targeting brain inflammation.

Regenxbio’s DMD gene therapy showed strong results, boosting muscle protein and safety, moving closer to FDA approval.

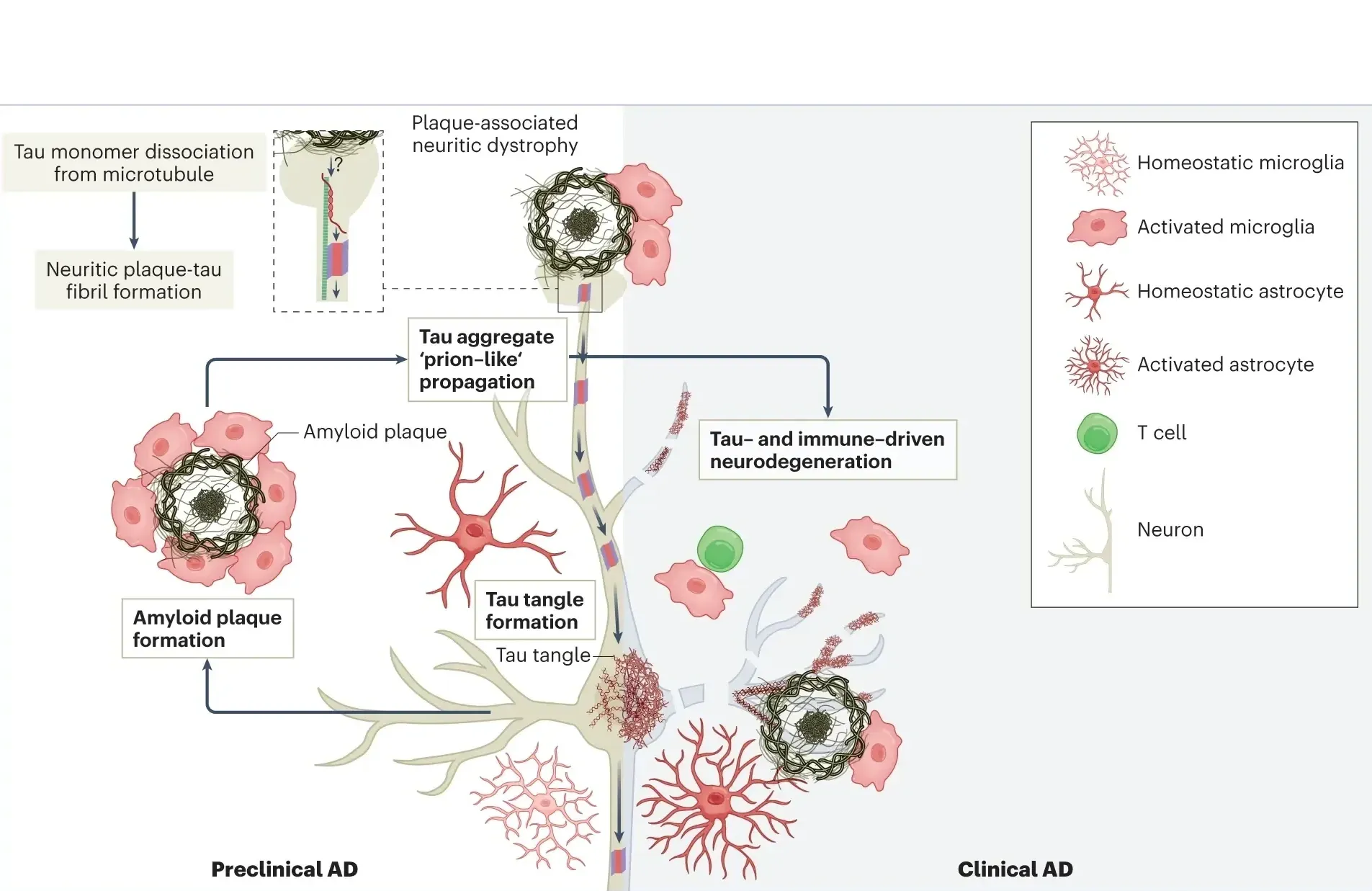
Biogen’s diranersen showed robust tau reduction and slowed cognitive decline in early Alzheimer’s Phase 2 trial. Moving to late-stage studies despite missing primary endpoint.

WW International rose after Galloway Capital built 8.42% stake, saying company is undervalued despite debt issues.

UBS upgraded CORT to Buy, raised target to $72 on strong Lifyorli cancer drug outlook and steady Korlym sales. Upside ~60–70%.

Zydus will buy Assertio for $166.4M, paying $23.50/share, giving shareholders more value and taking the company private.

Pulse Biosciences stock jumped after top executives bought $13M in shares, signaling strong confidence in future growth.
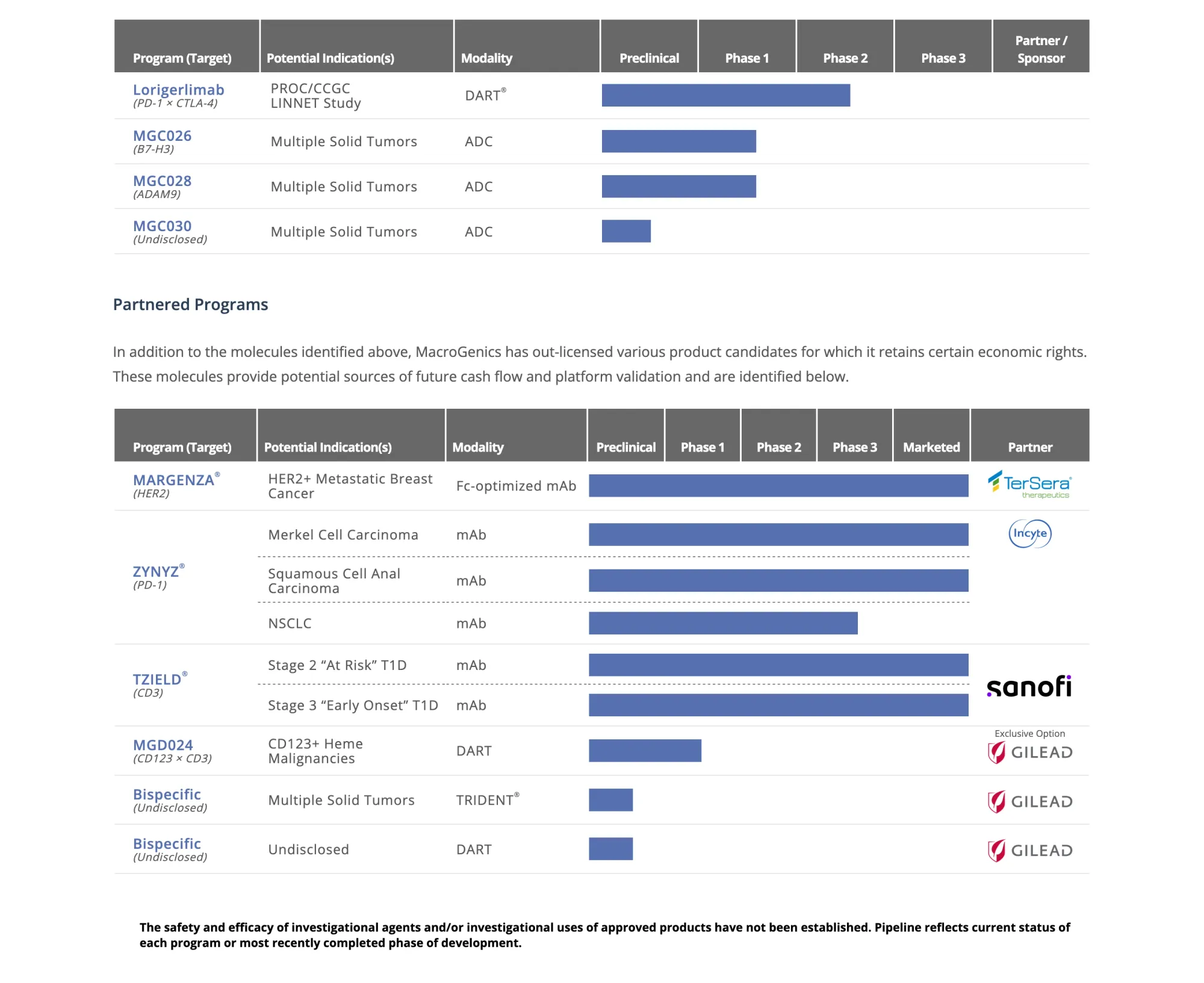
MacroGenics sells manufacturing site to Bora for $122.5M, keeps access, gains cash, and focuses on cancer drug development.
Arvinas & Pfizer partner with Rigel to launch Veppanu, a new oral breast cancer drug showing strong results and better survival.
Hims & Hers missed earnings, lost money, but grew users and expects stronger future growth with higher revenue guidance.
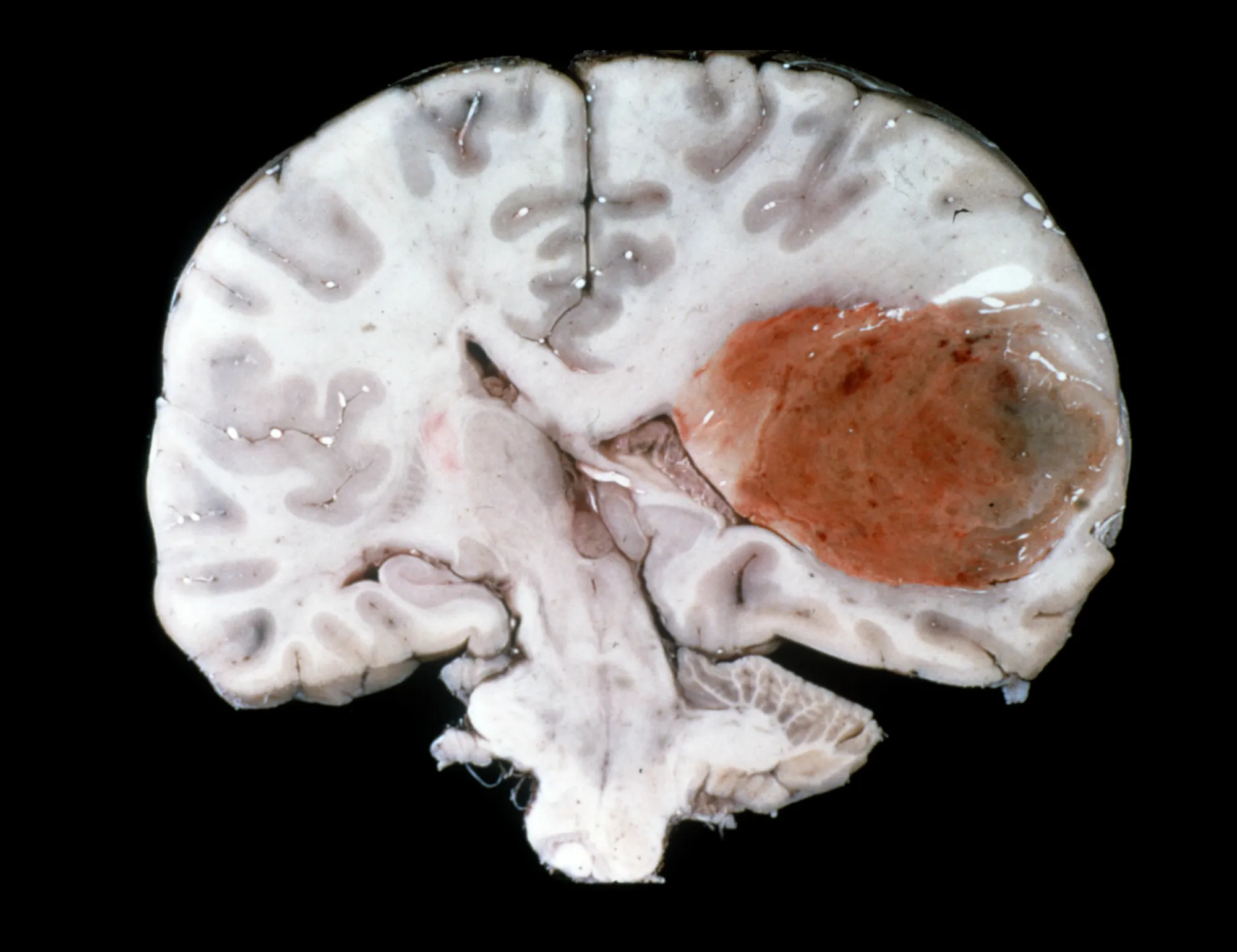
Alpha Tau’s brain cancer trial shows 2/3 glioblastoma patients’ tumors disappeared, all controlled, with good safety so far.
MBX drug showed early weight loss up to 7% in 8 weeks, was well tolerated, and may become once-monthly treatment.

Zai Lab’s drug Zoci got FDA Fast Track for rare cancer epNEC, showing 38% tumor response in early trials, speeding approval path.

Inhibrx drug INBRX-106 with pembrolizumab boosted tumor response in head/neck cancer, showing higher shrinkage and immune activation.

Guggenheim rates Coherus Oncology Buy with $12 target vs $1.75 now, seeing big upside in cancer drug pipeline but high risk.
Erasca rises after teaming with Merck to test ERAS-0015 + Keytruda in RAS cancers, showing early tumor shrink but past safety concern.
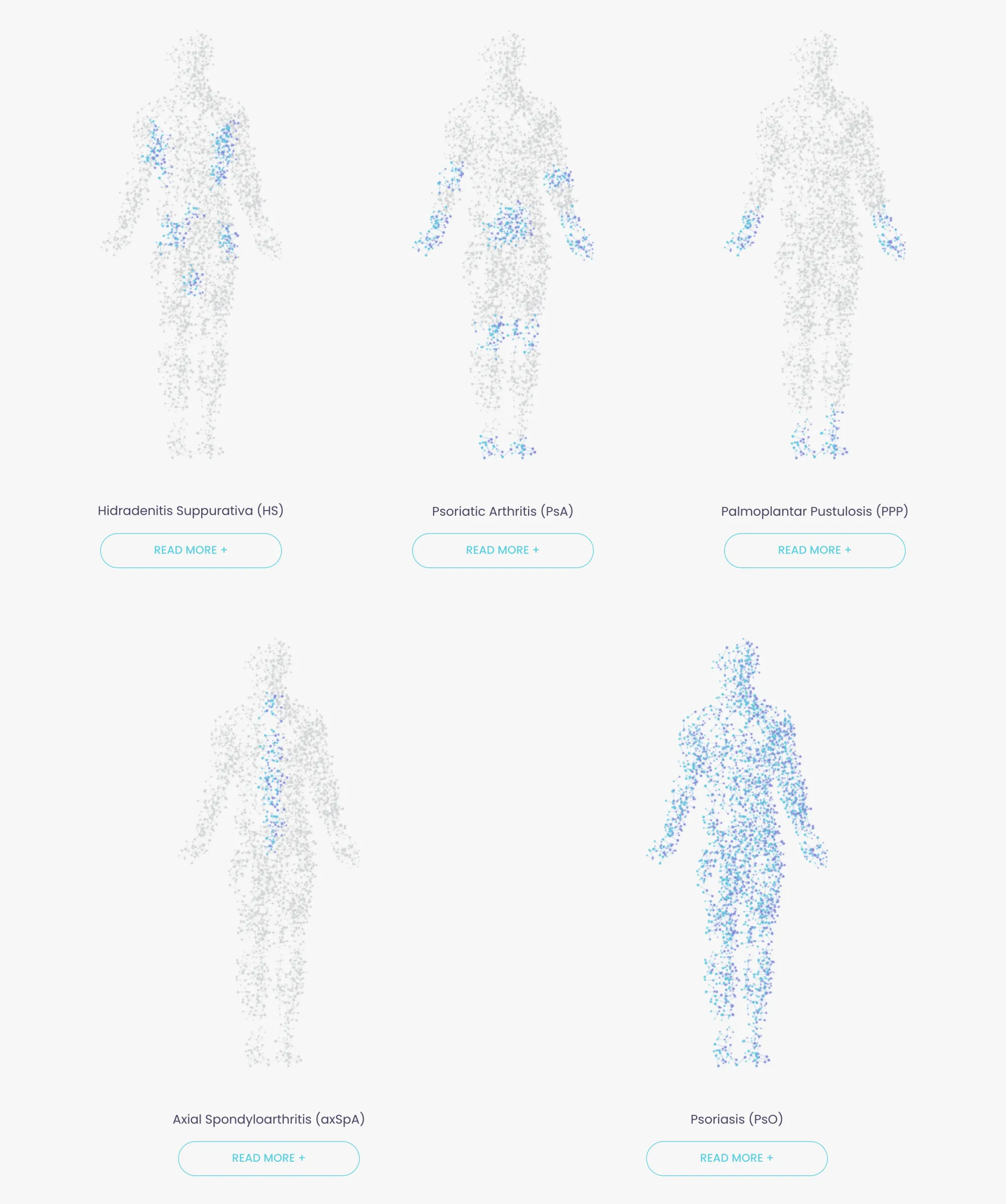
MoonLake gets FDA support for its HS drug, showing top results. Analysts stay bullish with strong growth potential ahead.
Moderna stock rises as it works on a hantavirus vaccine after a cruise outbreak, but the disease is rare and vaccines will take years.

Replimune stock jumped after news Trump may fire FDA chief, raising hopes of easier drug approvals despite past rejections.

PTC beat estimates, Sephience sales surged, losses small, outlook raised, and analysts upgraded stock on strong growth momentum.

Atara stock jumps 52% after FDA signals its cancer therapy may get approved, boosting hope for patients and investors.
Oppenheimer backs Aprea with $5 target. Its cancer drug shows early promise, and low valuation offers big upside potential.

Angelini is set to buy US-based Catalyst for $31.50 per share in a state-backed deal, expanding into rare neurological diseases and entering the US market.
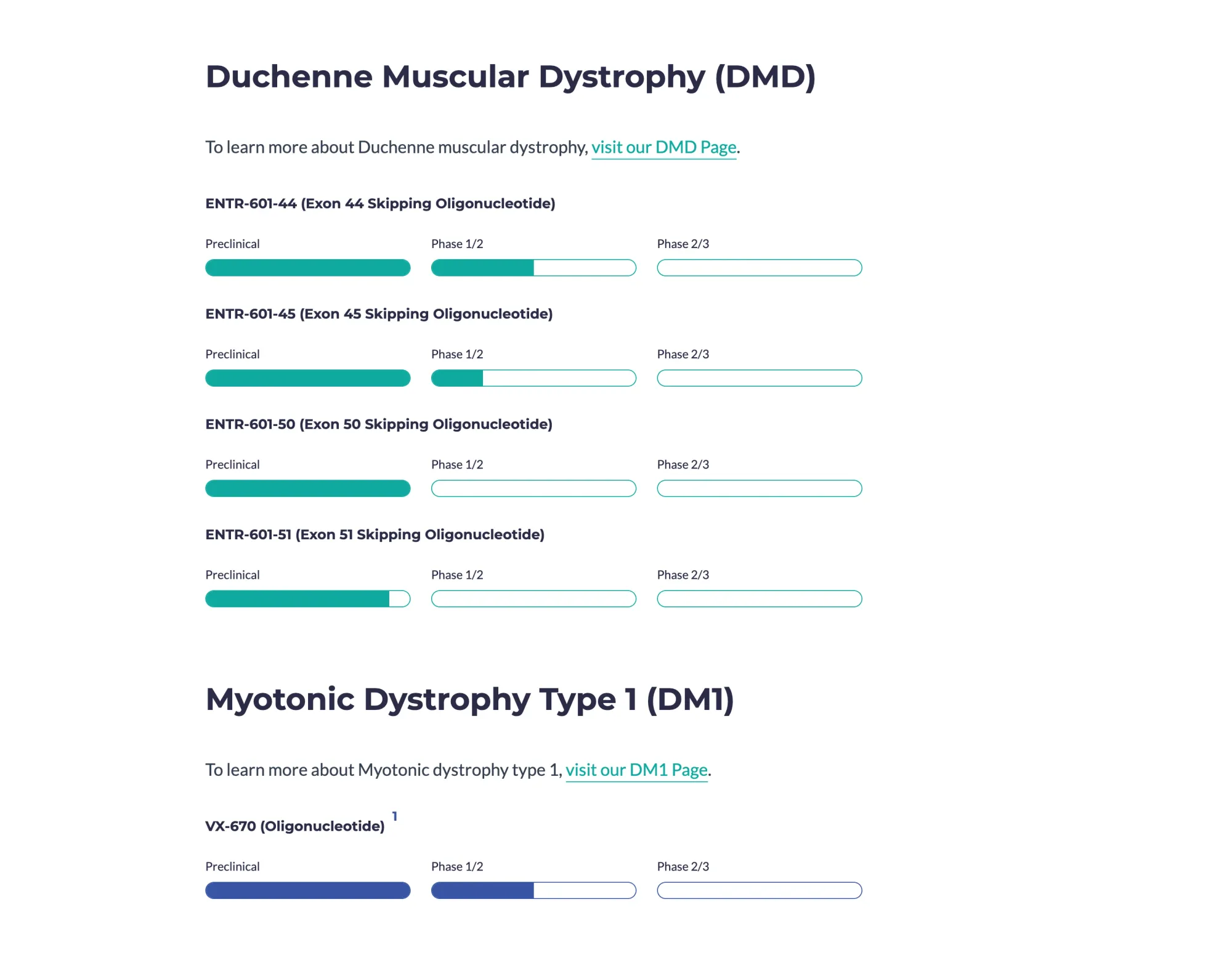
Entrada DMD drug was safe and helped movement a bit, but low muscle protein results disappointed investors, stock -60%.

enGene bladder cancer drug shows good safety and early response, but weak long-term results; stock drops ~66% after update.

Sarepta reported strong Q1 2026 earnings, with revenue of $730.8M, but shares fell as Elevidys sales dropped 46% due to label restrictions.

Oscar Health posted record profit, strong growth, and lower costs, showing a major turnaround and fast expansion.
Novavax beat forecasts with $140M revenue, boosted by vaccine tech deals, new partnerships, and strong cash position.

CVS had strong Q1: revenue $100.4B, earnings beat expectations, raised outlook, cash flow up. Stock rose.

Novo Nordisk beat expectations as its new weight-loss pill sold fast, boosting sales, prescriptions, and improving its 2026 outlook.

Tempus AI revenue rose 36% to $348M, but losses widened. Stock fell ~7% after weaker profit and cautious outlook.
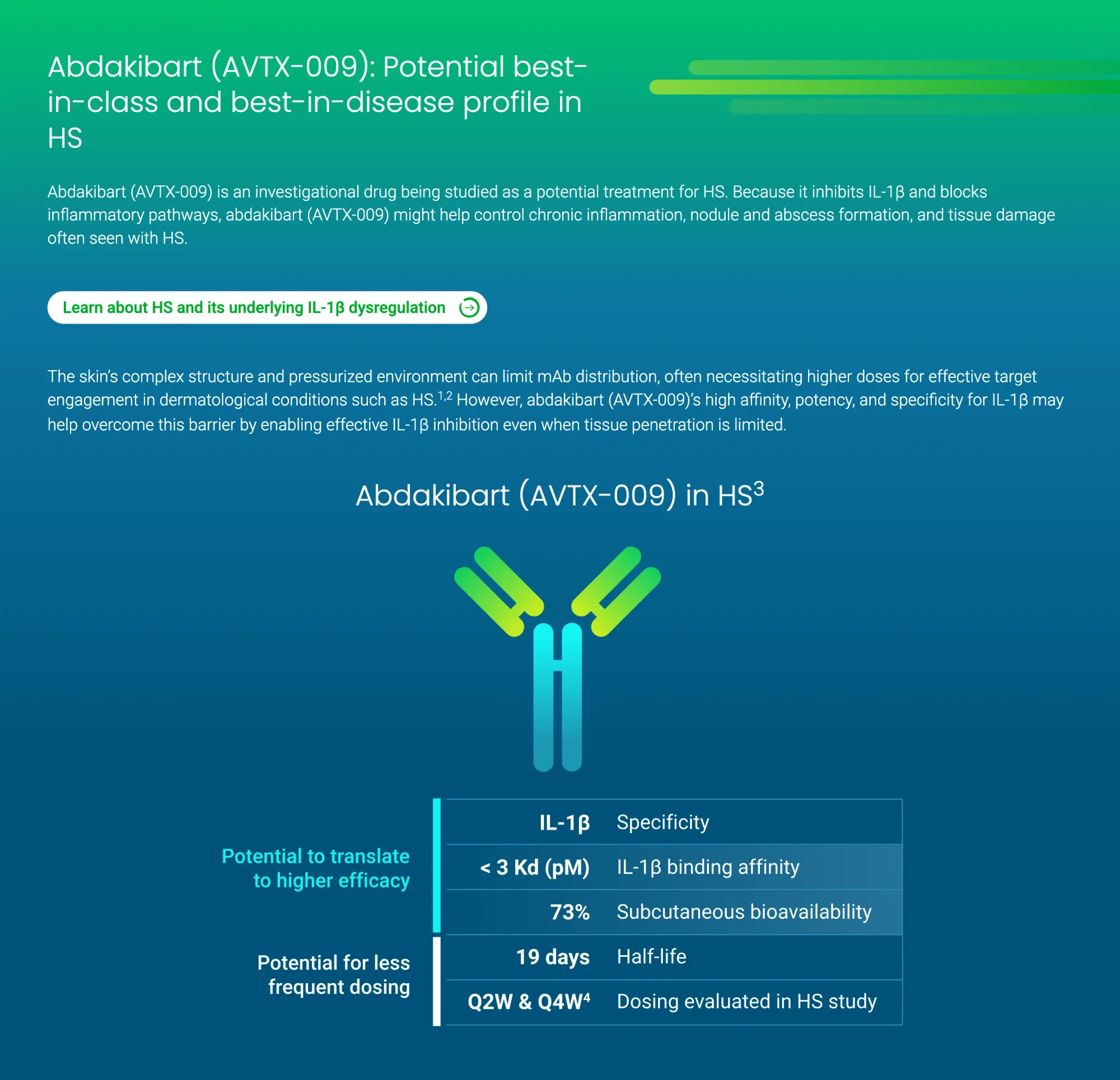
Avalo’s abdakibart showed strong results in reducing HS symptoms in a Phase 2 trial, with a favorable safety profile and better outcomes than placebo.
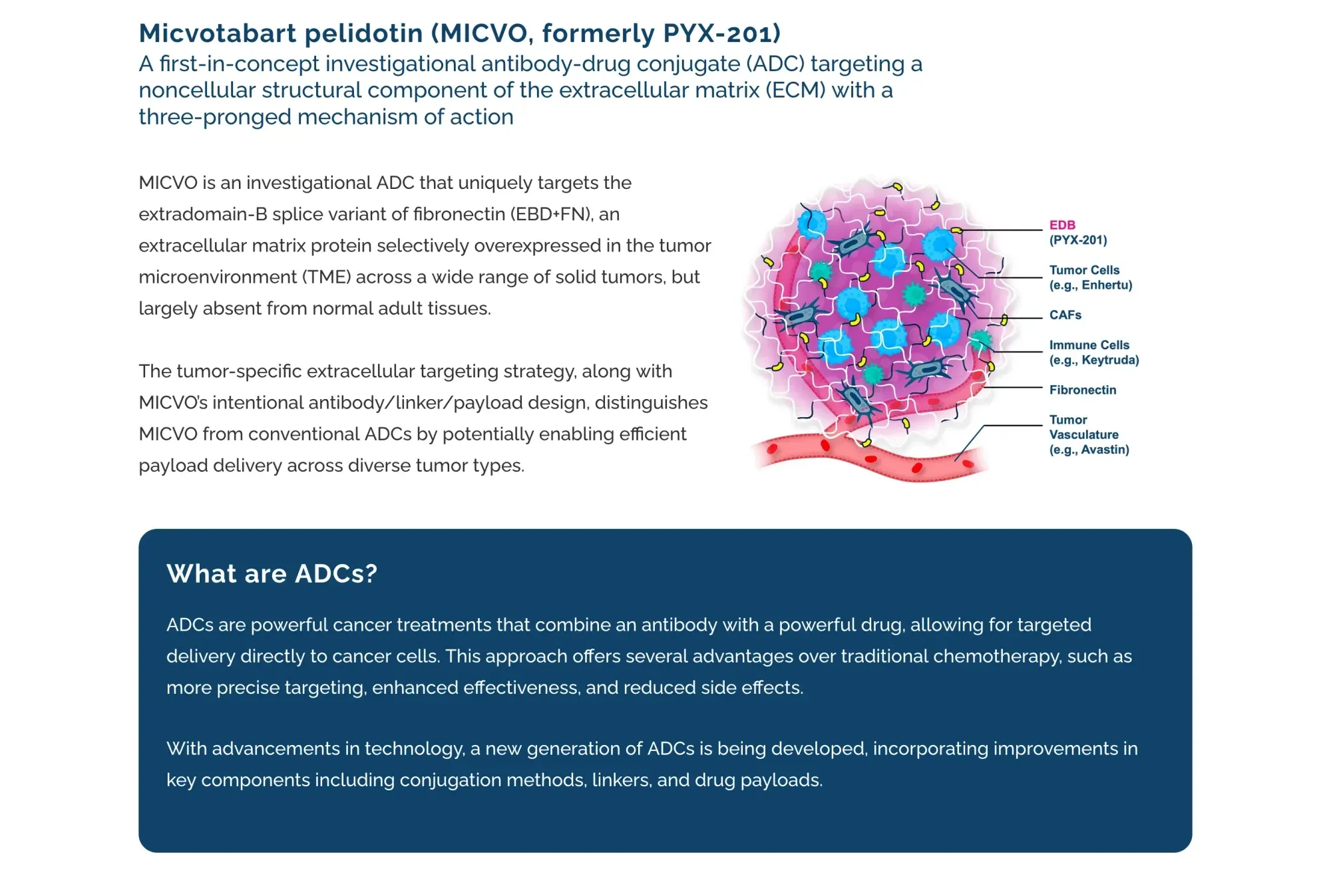
Wedbush rates Pyxis “Outperform” with $6 target vs $1.90. Early cancer drug data promising but risky biotech with funding needs.
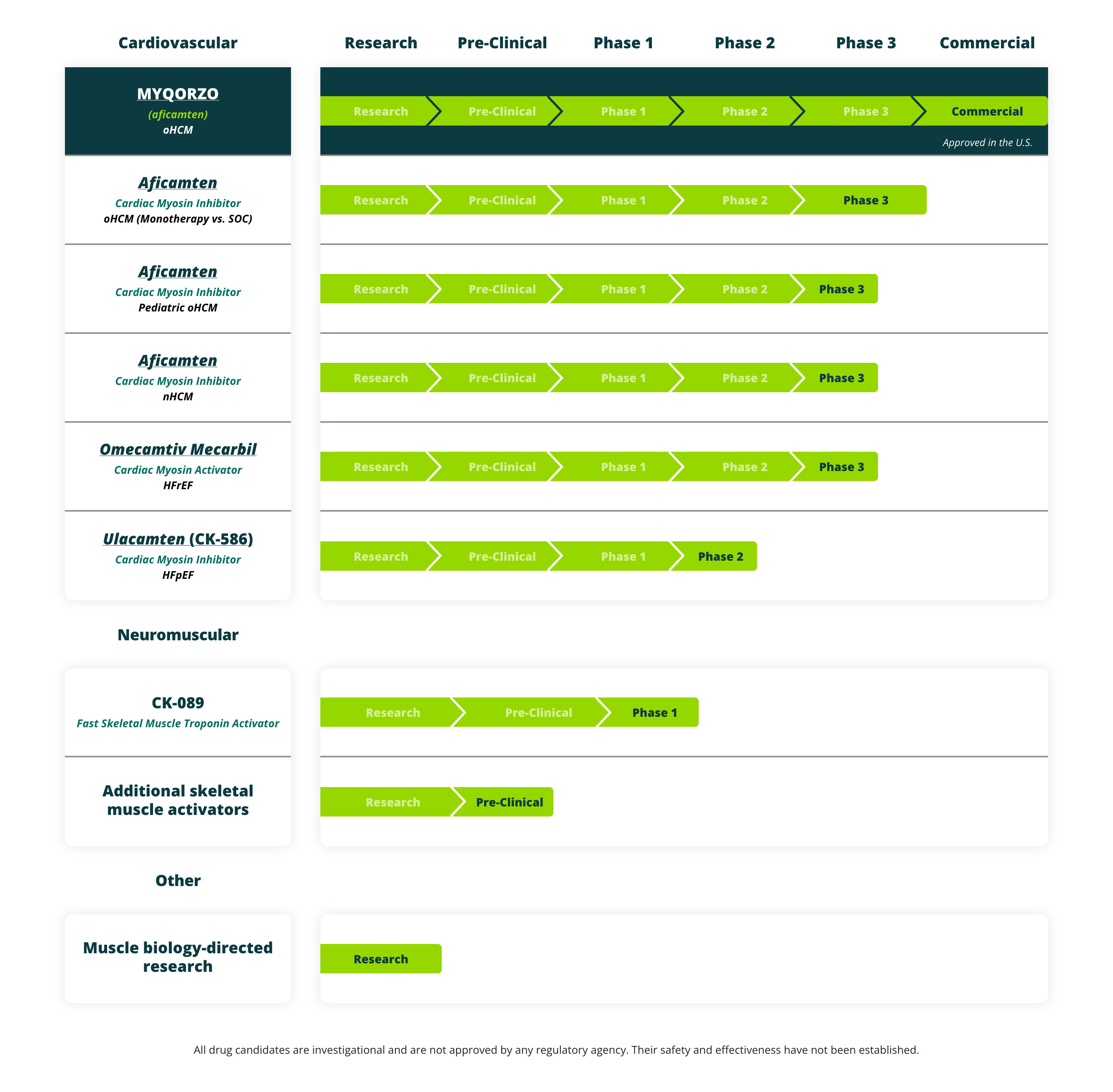
Cytokinetics’ (aficamten) met both primary endpoints in the Phase 3 ACACIA-HCM trial for non-obstructive hypertrophic cardiomyopathy (nHCM). Already approved for obstructive HCM, the drug could soon double its market.
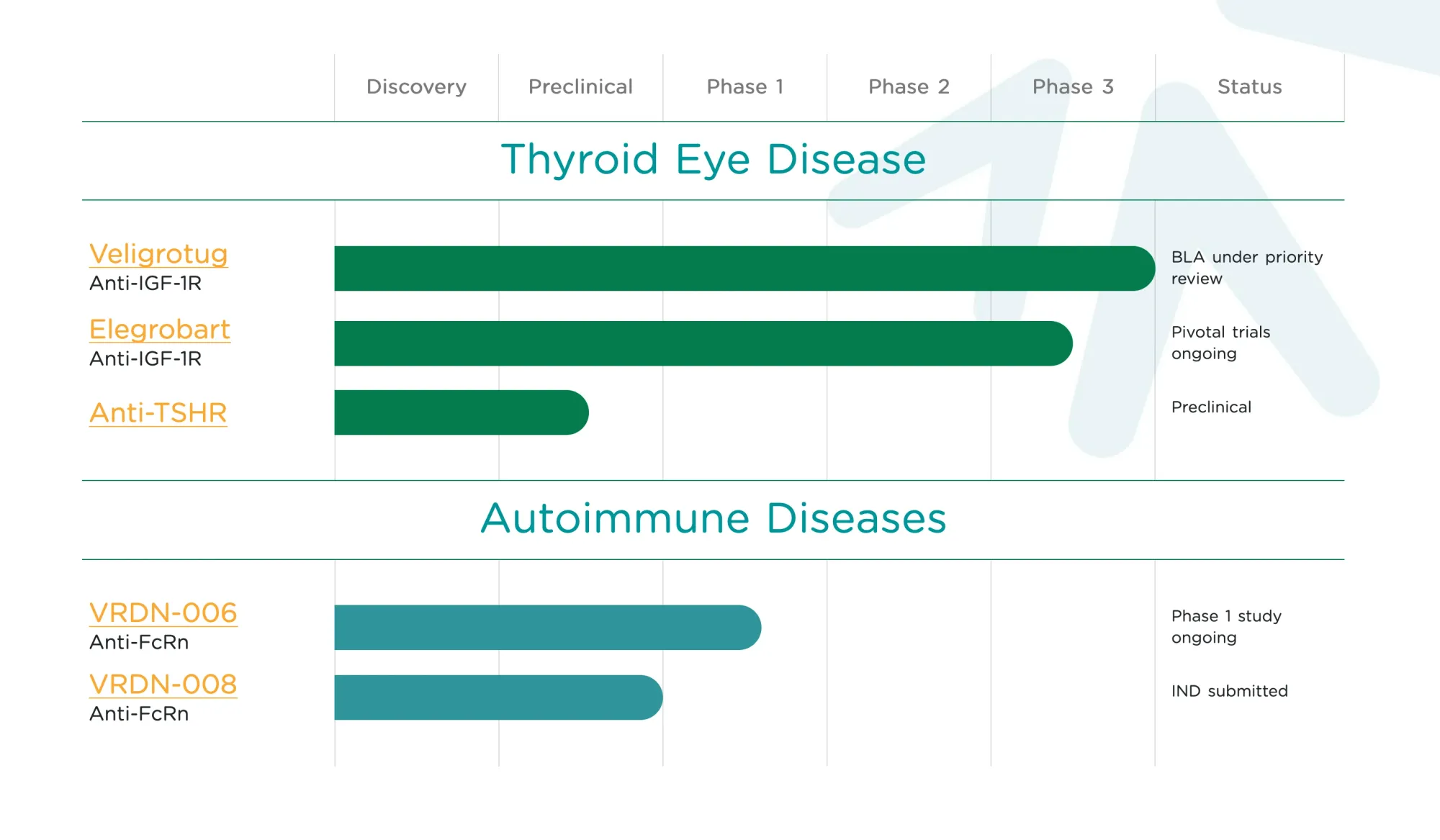
Viridian's drug for thyroid eye disease showed strong Phase 3 results, improving eye symptoms with simple injections and possible home use.
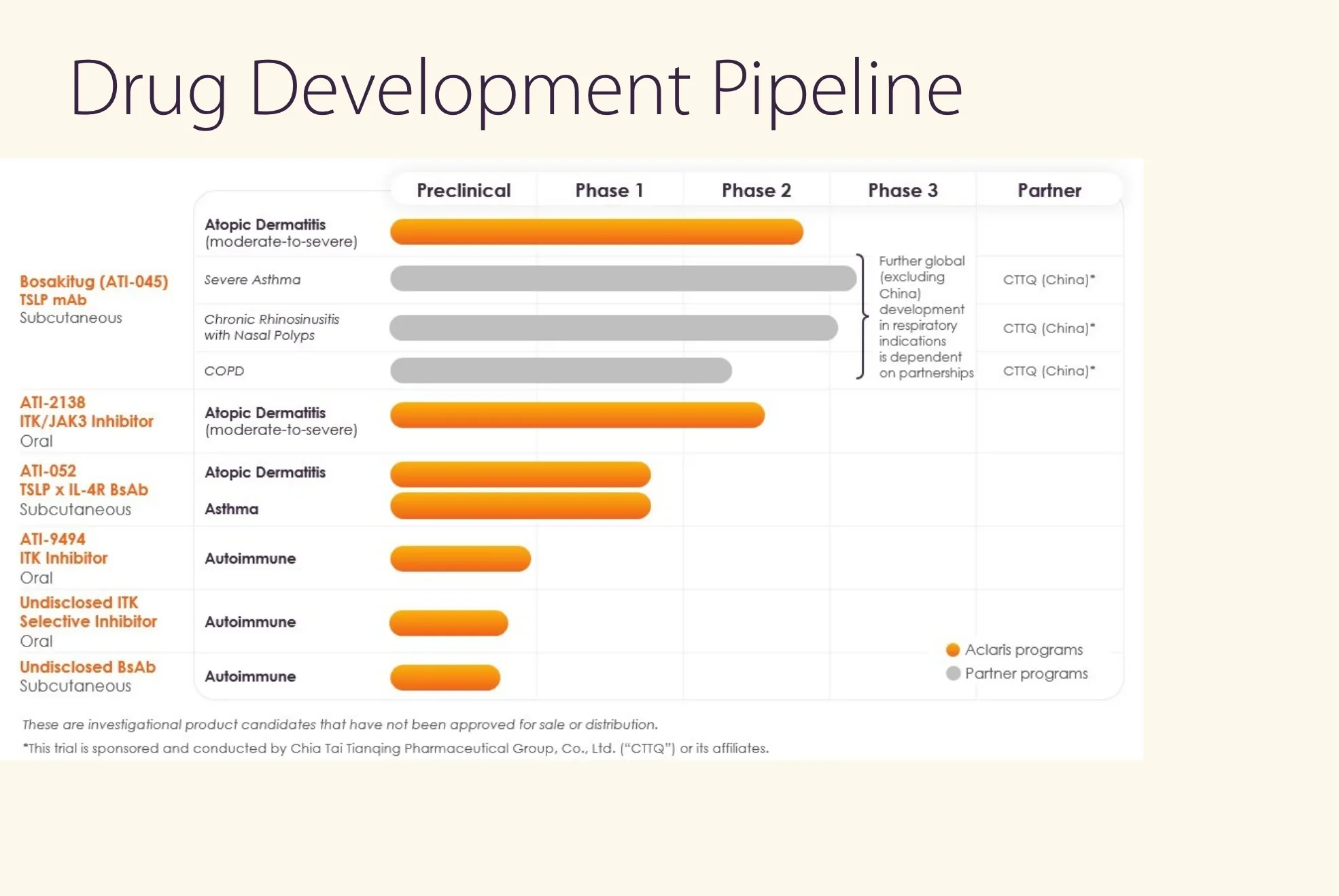
Guggenheim rates ACRS Buy, $12 target vs $4.50 now; skin/immune drugs, trials ahead, big upside if results are good.

Biomea is testing a new diabetes pill that may fix insulin cells. Early results look strong, with big growth potential but high risk.

UCB buys Candid Therapeutics for $2B+ to develop immune disease drug that may reset immune system; rivals’ stocks rise.
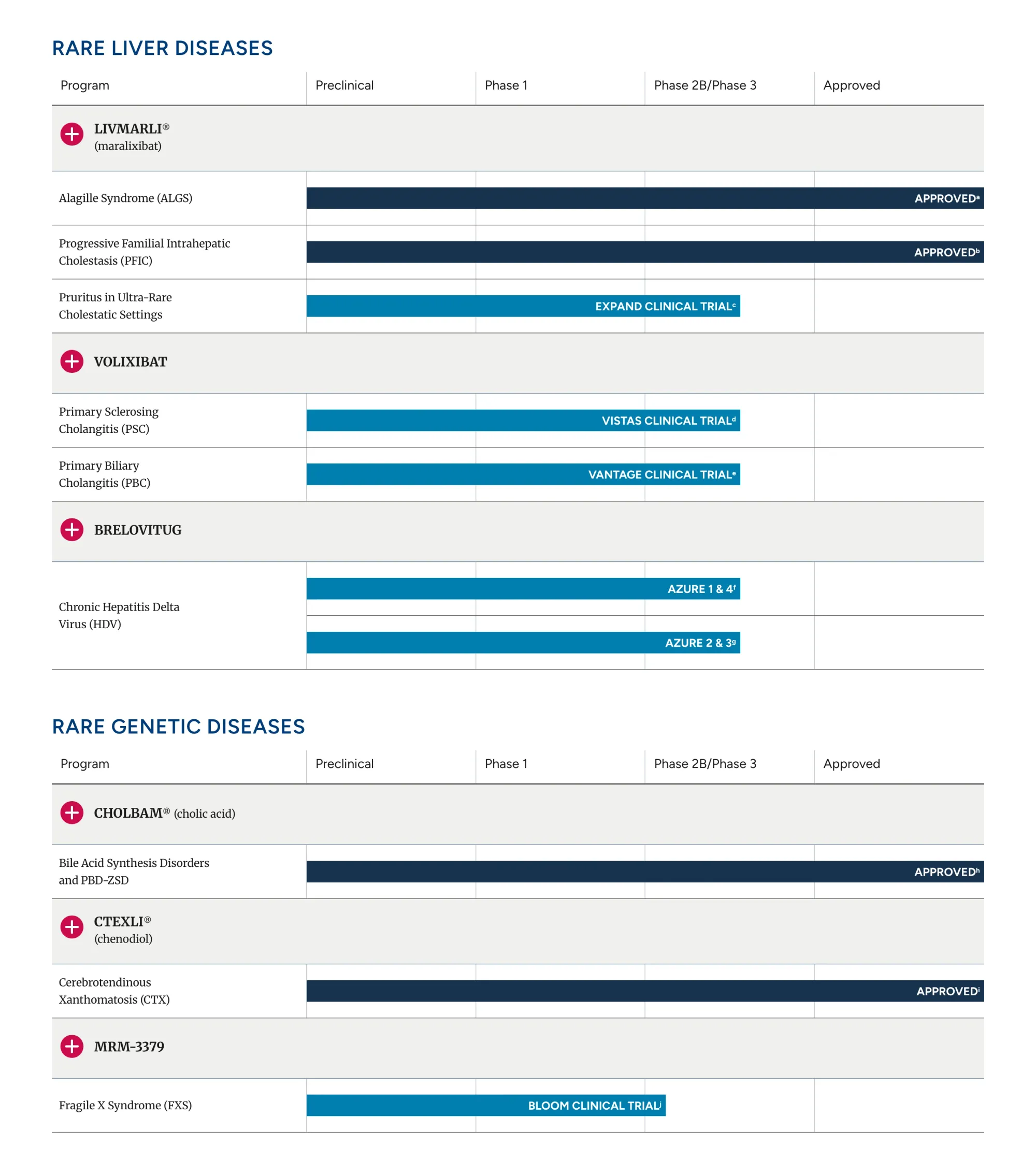
Mirum’s drug volixibat reduced severe itching in PSC patients in a Phase 2 trial; next step is FDA filing in 2026.

Garda raised bid to $21.80/share cash to buy Assertio, boosting stock ~17%. Deal gives shareholders higher, certain payout.
Eli Lilly states that a post-launch report of hepatic failure for Foundayo (orforglipron) is not plausibly linked to the drug. RBC Capital describes it as “statistical background noise.”
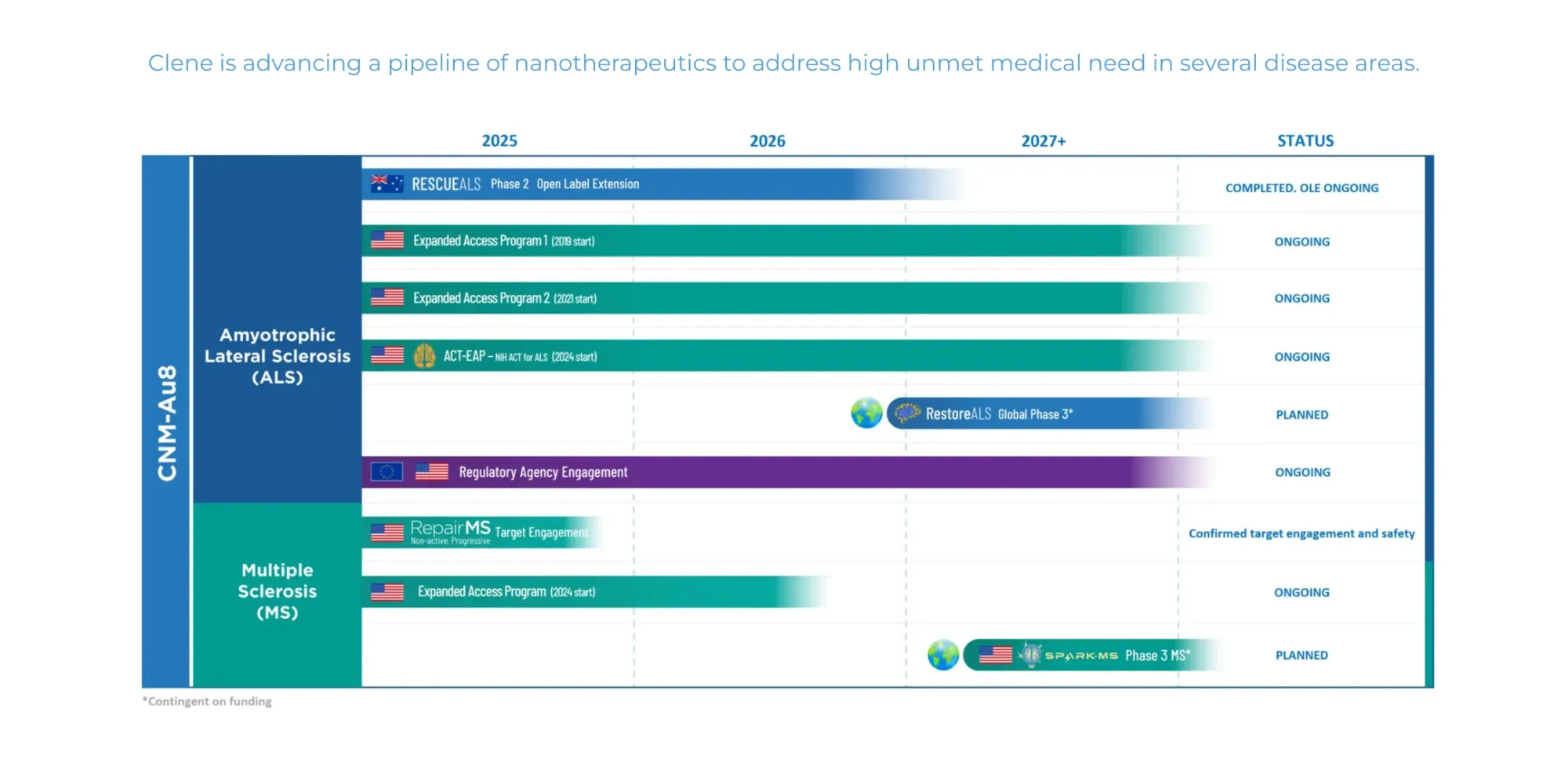
FDA may fast-track Clene’s ALS drug CNM-Au8 after strong results, offering hope for longer life and slower disease progression.
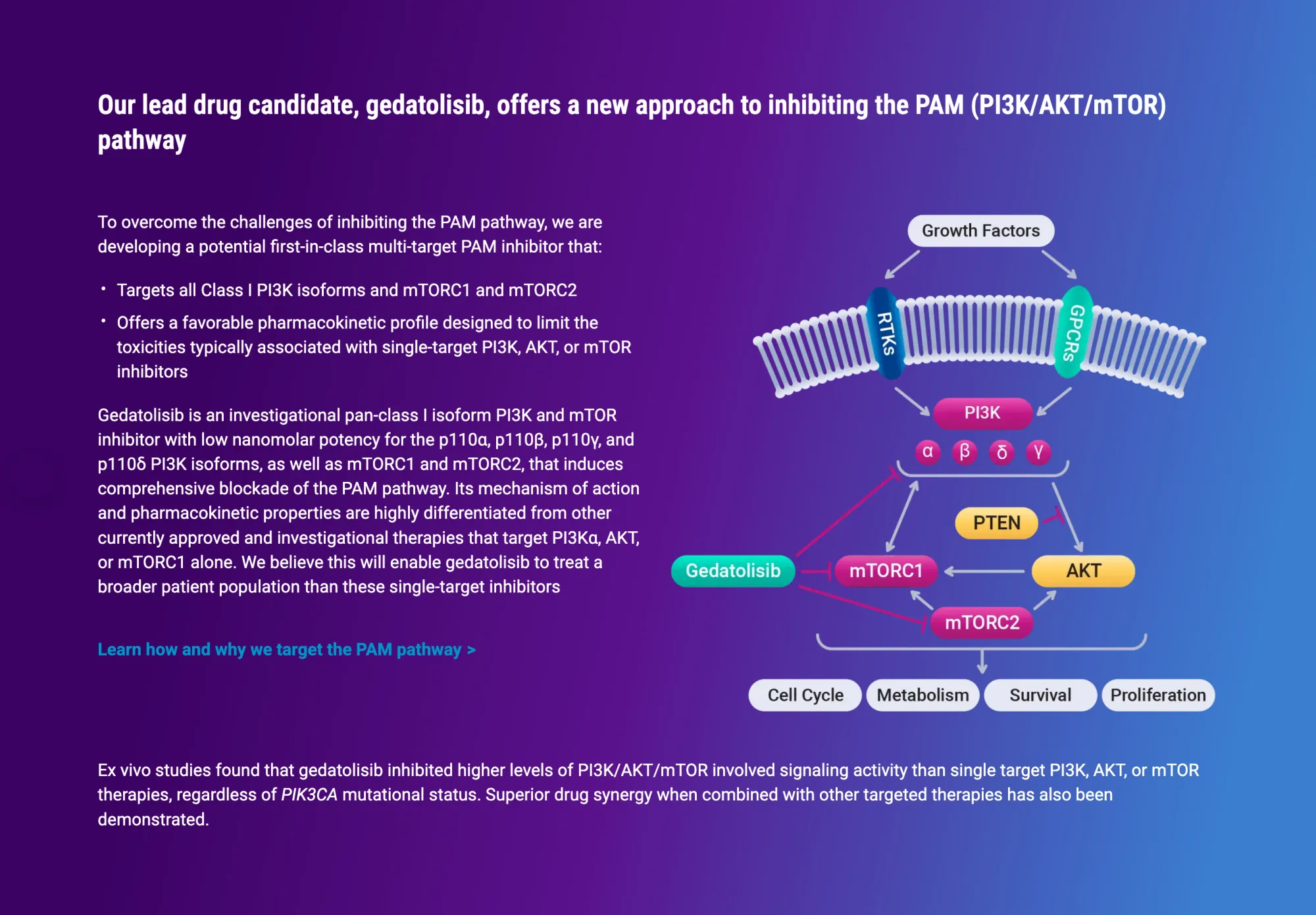
Celcuity’s drug showed strong results in breast cancer, boosting survival and raising hopes it could become a new standard treatment.

FDA approved Pfizer/Arvinas Veppanu pill for advanced breast cancer with ESR1 mutation, helping hormone-resistant cases.
Summit stock fell after its cancer drug update was just “keep going,” not the big win investors hoped for.

ARCHIMED will buy Esperion and take it private for $3.16/share (+58% premium) plus up to $100M extra payouts if targets are met.
Moderna beat Q1 revenue on strong overseas COVID vaccine sales, revenue $389M, smaller US demand, reported a per-share loss.
FDA approves AUVELITY to treat Alzheimer’s agitation, offering a safer, effective option to reduce distress and improve patient care.

Eli Lilly raised profit outlook thanks to strong Zepbound & Mounjaro sales; revenue and earnings beat expectations, stock rose.

Teva buys Emalex for $700M to add a new Tourette drug. Strong sales growth led by key medicines; generics weaker but outlook solid.

Chiesi will buy KalVista for $1.9B to get EKTERLY, a new oral rare disease drug, and grow its global rare disease business.

Humana beat Q1 2026 expectations, revenue rose, Medicare membership grew, outlook steady but GAAP cut due to lower ratings.
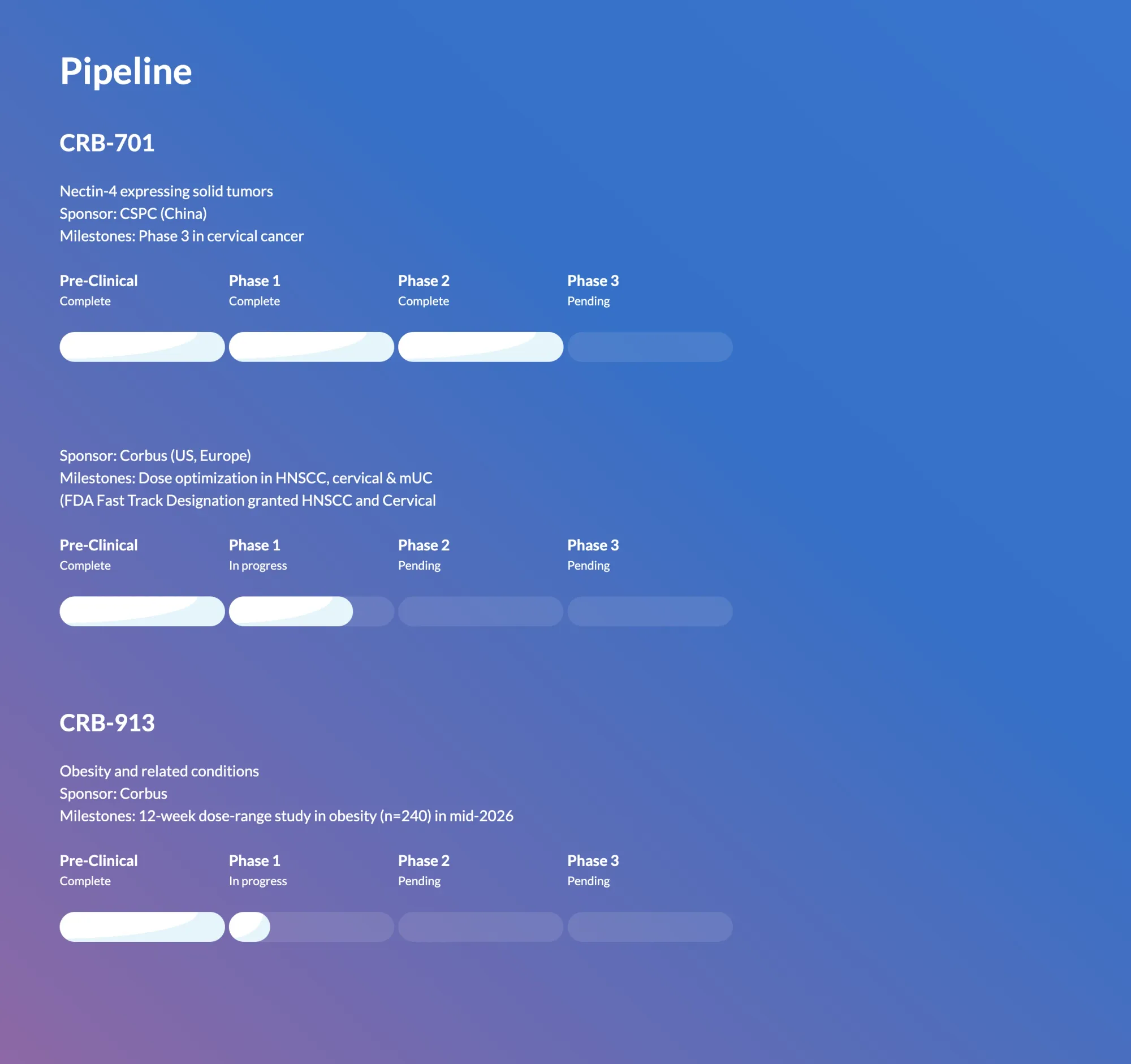
Corbus Pharmaceuticals gets Buy rating ($45 target). Drug CRB-701 shows strong cancer results, safer than rivals. Stock down 43% may be opportunity.

Guardant stock fell after FDA raised doubts on a key drug study. Delay fears hurt short-term, but long-term outlook still strong.

Pfizer extends Vyndamax patent to 2031, delaying generics and giving BridgeBio more time to grow its rival heart drug without pressure.
Rocket Pharma sold an FDA voucher for $180M cash, boosting funds without dilution and extending runway to 2028 for heart gene therapies.
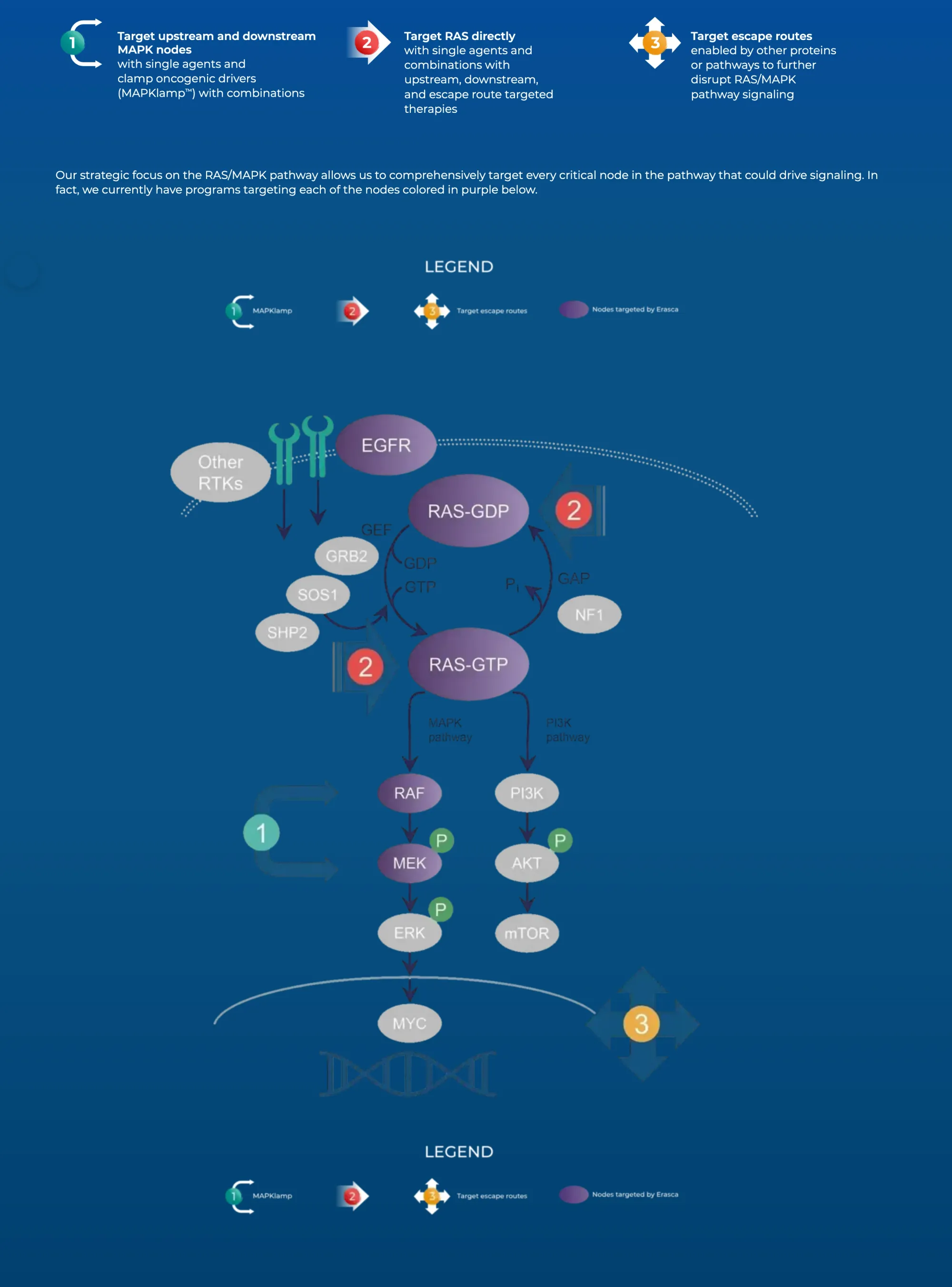
Erasca stock fell after a patient died in a cancer drug trial. Analysts say it’s likely a rare case, drug still shows promise.
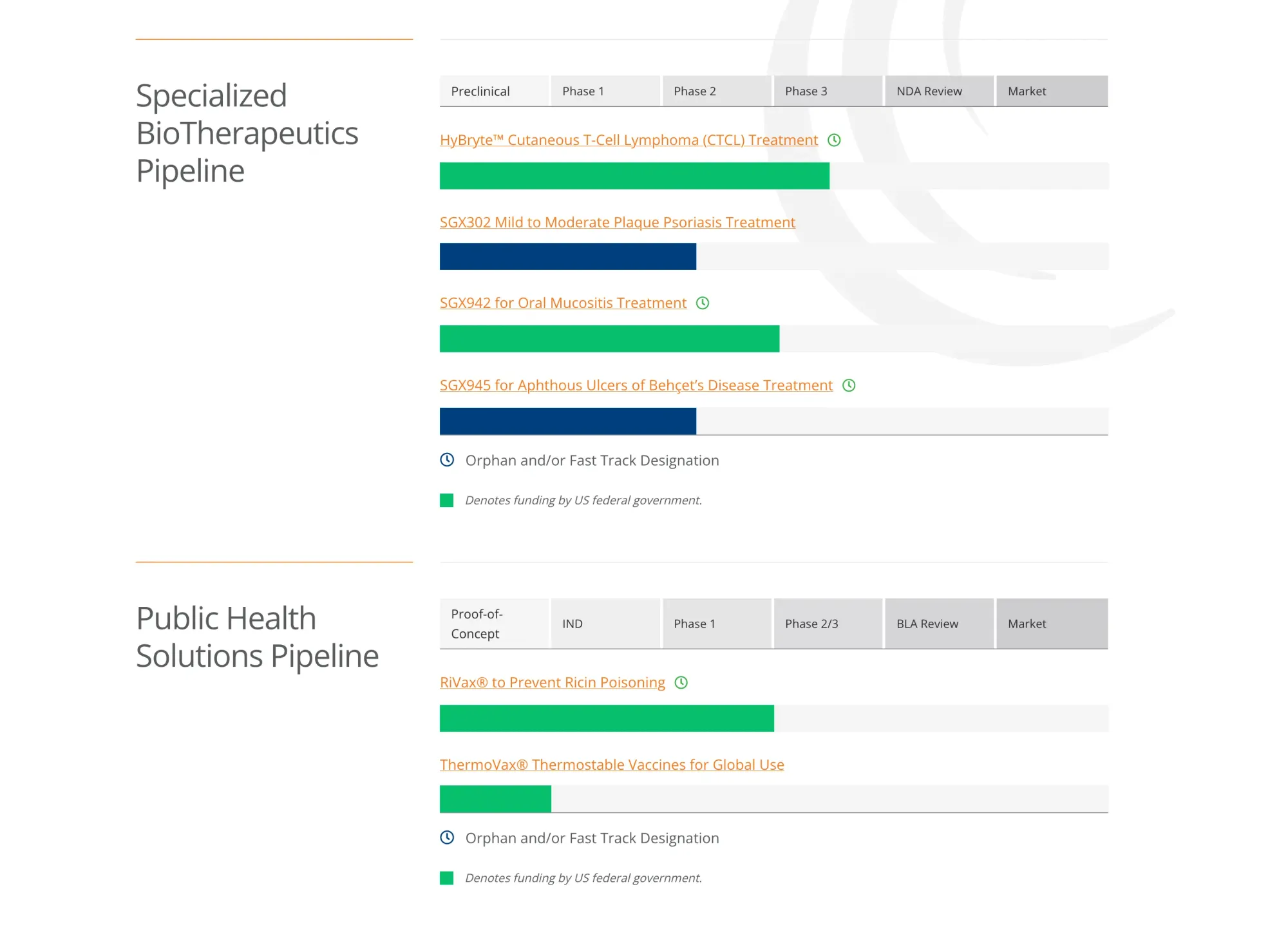
Soligenix stopped HyBryte CTCL trial early as it didn’t work well, though it was safe; company will review data and options.

Angelini Pharma may buy Catalyst Pharmaceuticals. Talks are early, but news pushed Catalyst’s stock up. No deal is certain yet.

Spruce Biosciences has a rare disease drug (TA-ERT) showing strong results, FDA path clear, big sales and upside potential.

Erasca denies patent claims by Revolution Medicines, will fight back; Stifel keeps Buy rating.
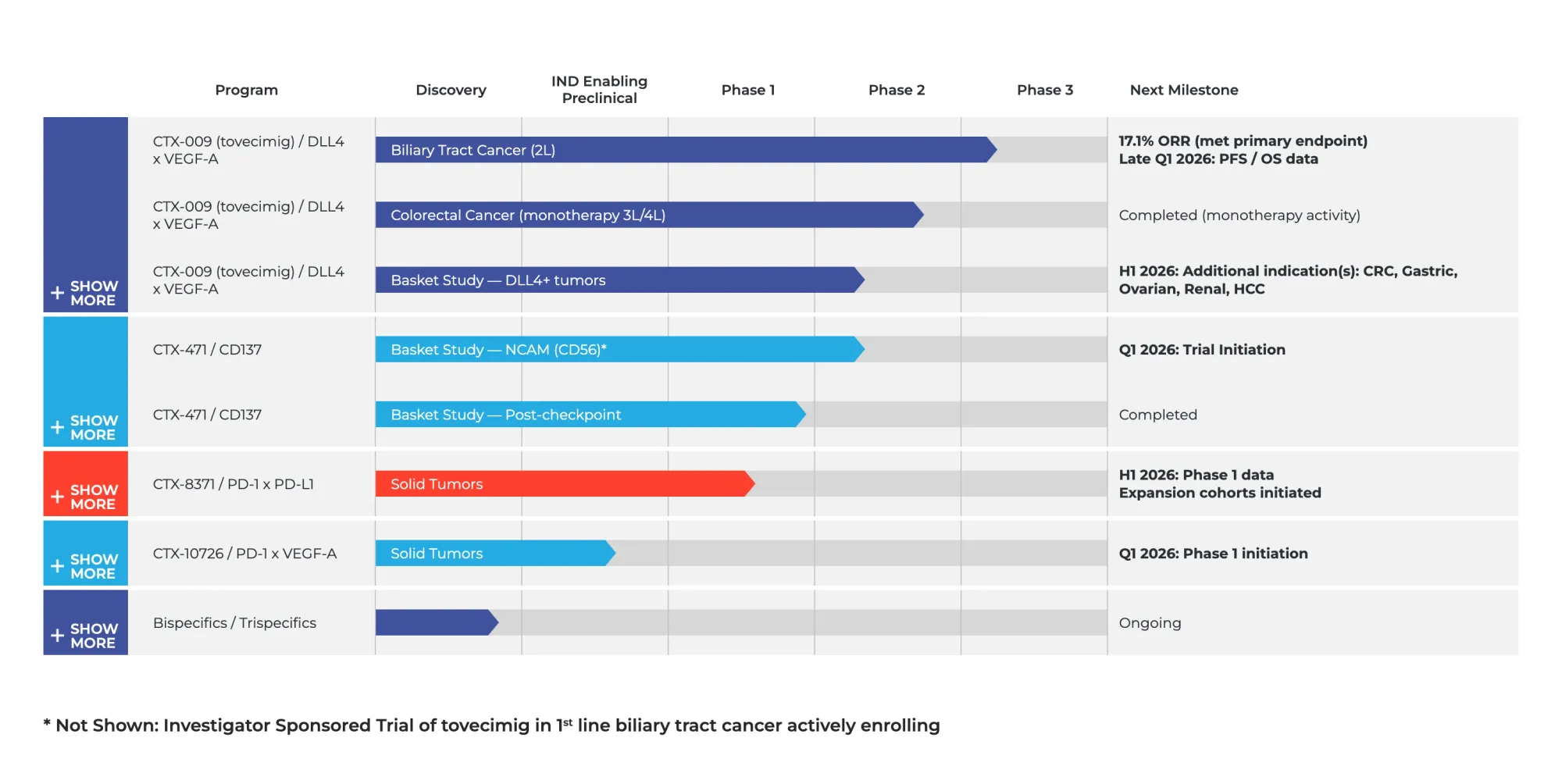
Compass Therapeutics fell after cancer drug improved outcomes but failed to show clear survival benefit due to patient crossover.
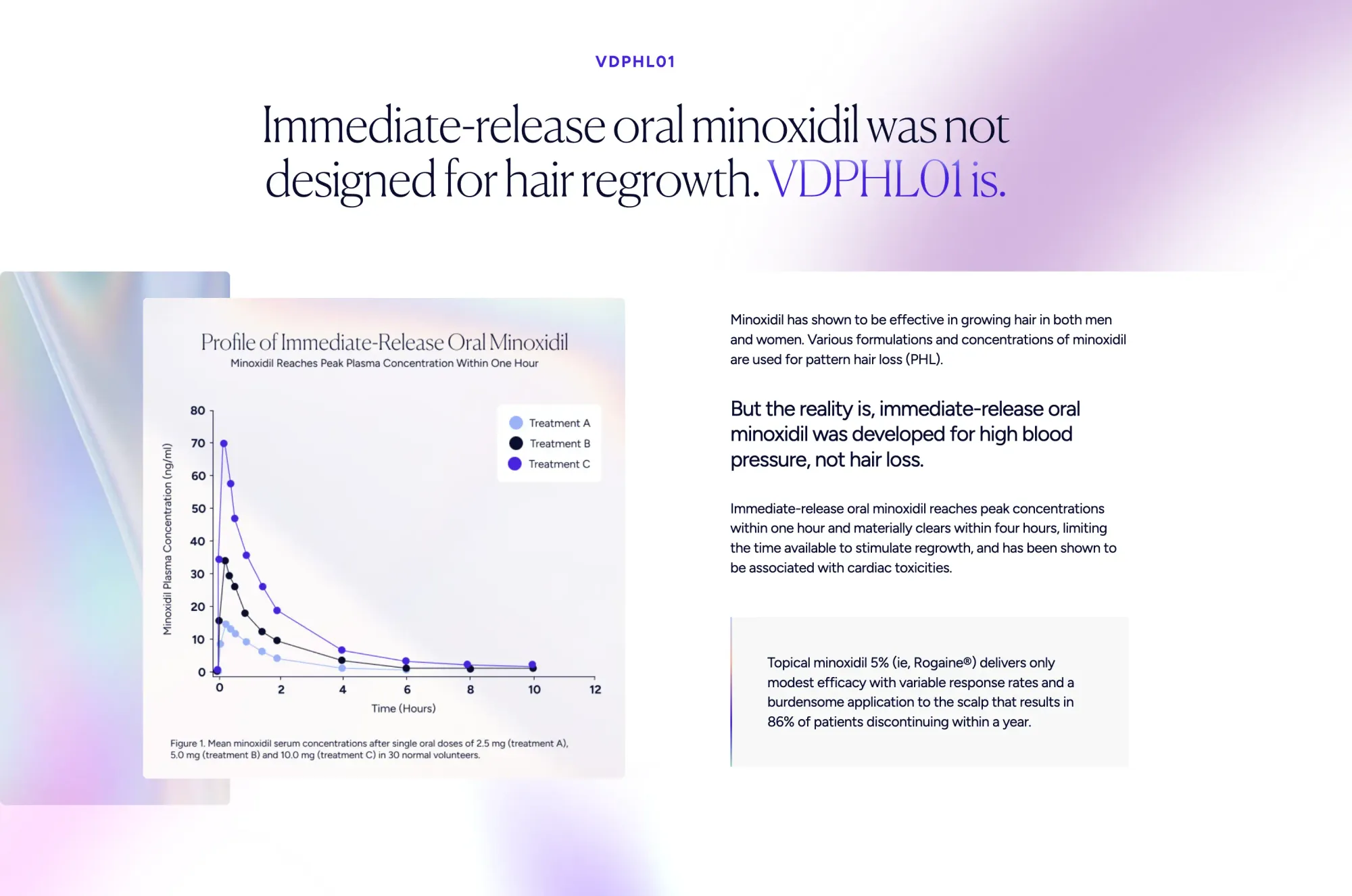
Veradermics’ hair pill VDPHL01 showed strong results, growing 30+ hairs/cm in trials, safe, and may become a new baldness treatment.

Oruka’s new psoriasis drug cleared skin in most patients with few side effects and may need only yearly injections.

Eli Lilly buys Ajax to develop a new pill for rare blood cancers that may work longer and better than current treatments.
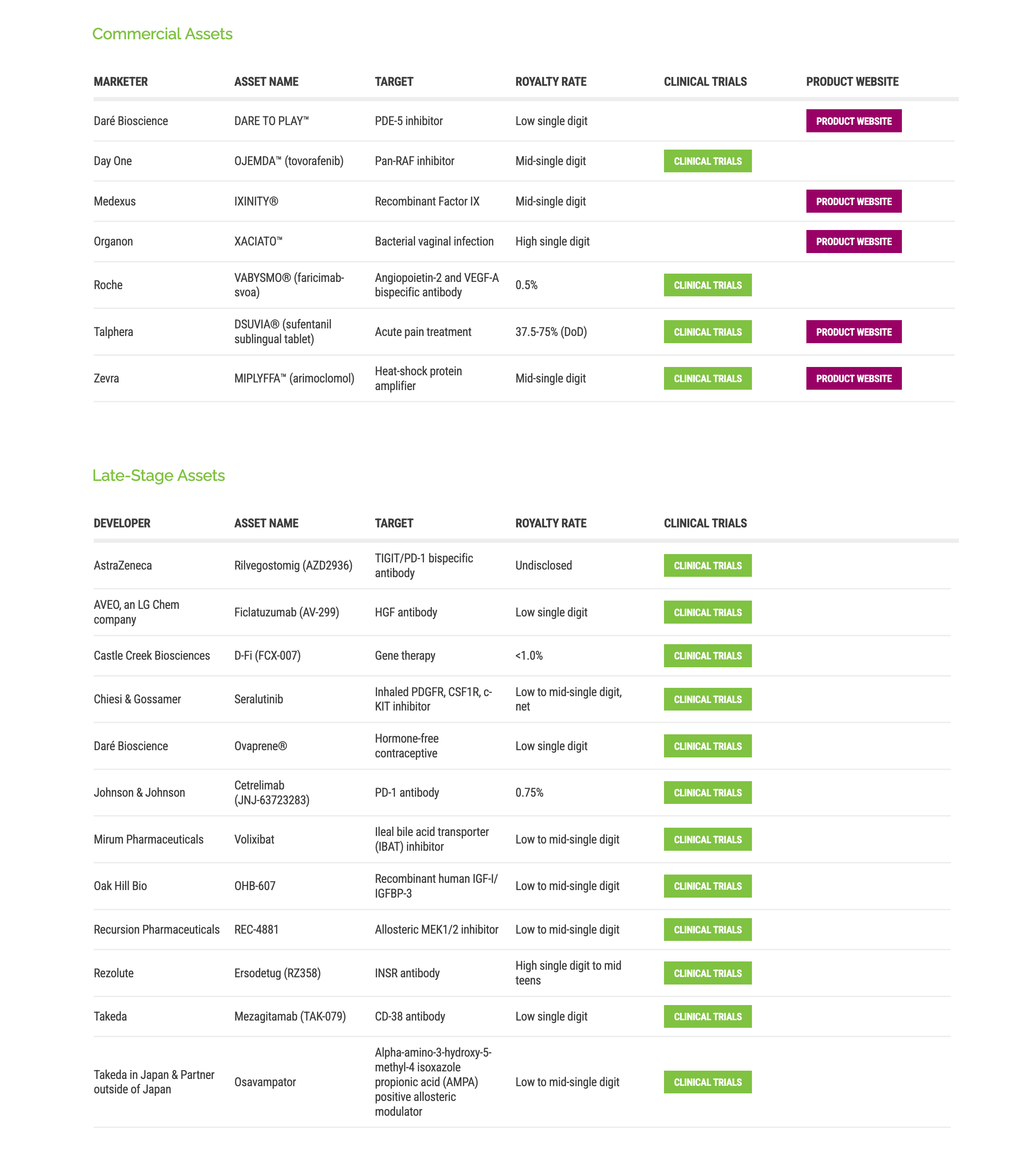
Ligand buys Xoma for $740M cash, boosting its drug royalty portfolio to 200+ medicines and spreading investment risk.

Sun Pharma will buy Organon for $11.75B, boosting global reach, women’s health, and specialty drugs, and creating a $12.4B annual revenue pharma giant.
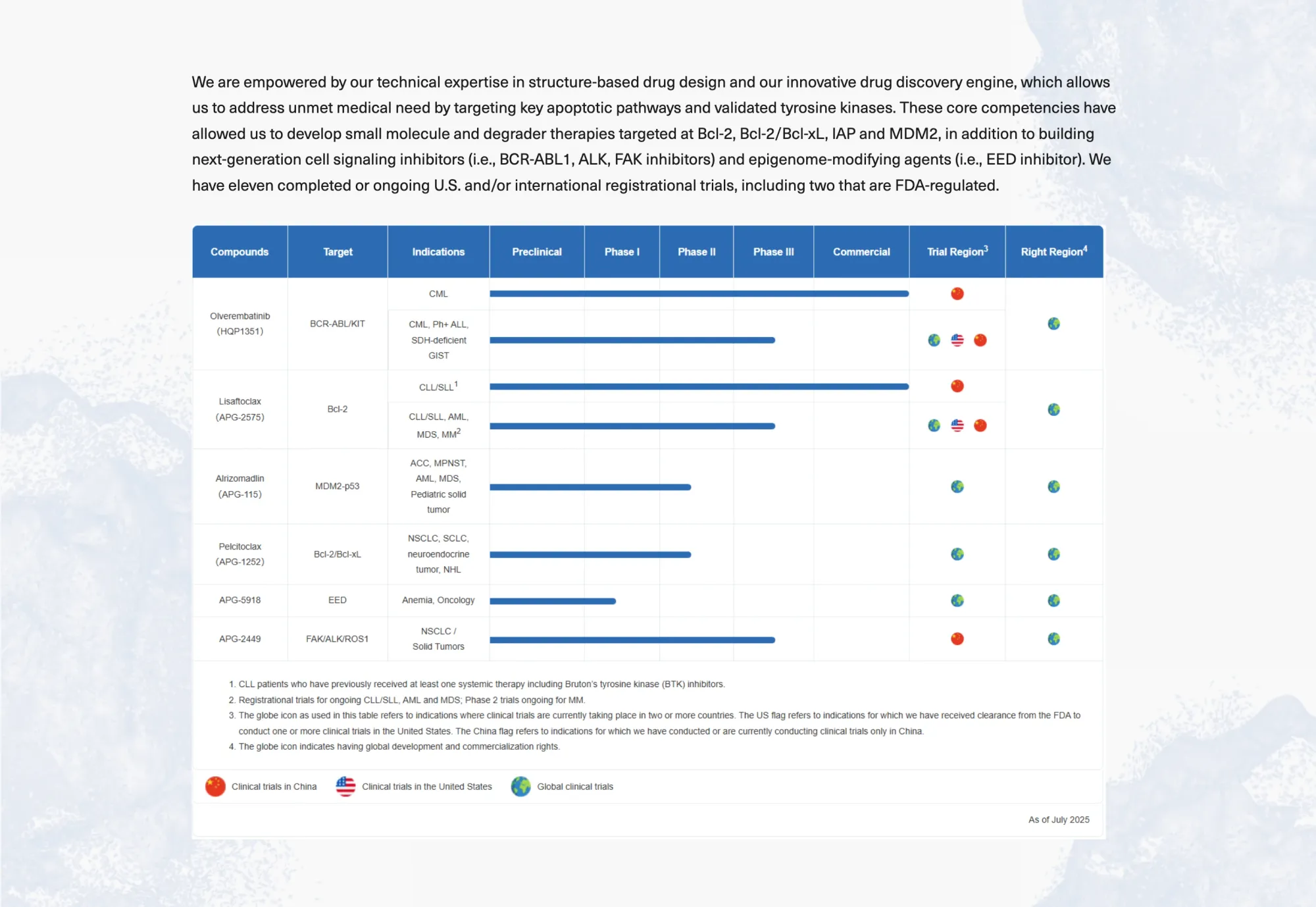
Ascentage Pharma gets Buy ratings; its cancer drugs show strong results, fast growth in China, and high future upside potential.
Investors offered to buy Seer for $2.35/share plus extra future payout; stock rose after takeover bid. Board responds by May 2.
Lilly’s new weight-loss pill started slow, far behind Novo’s, but it’s too early to tell which drug will win.

Sun Pharma offers $13B to buy Organon, aiming to expand globally in branded drugs, competing with EQT and Grünenthal.
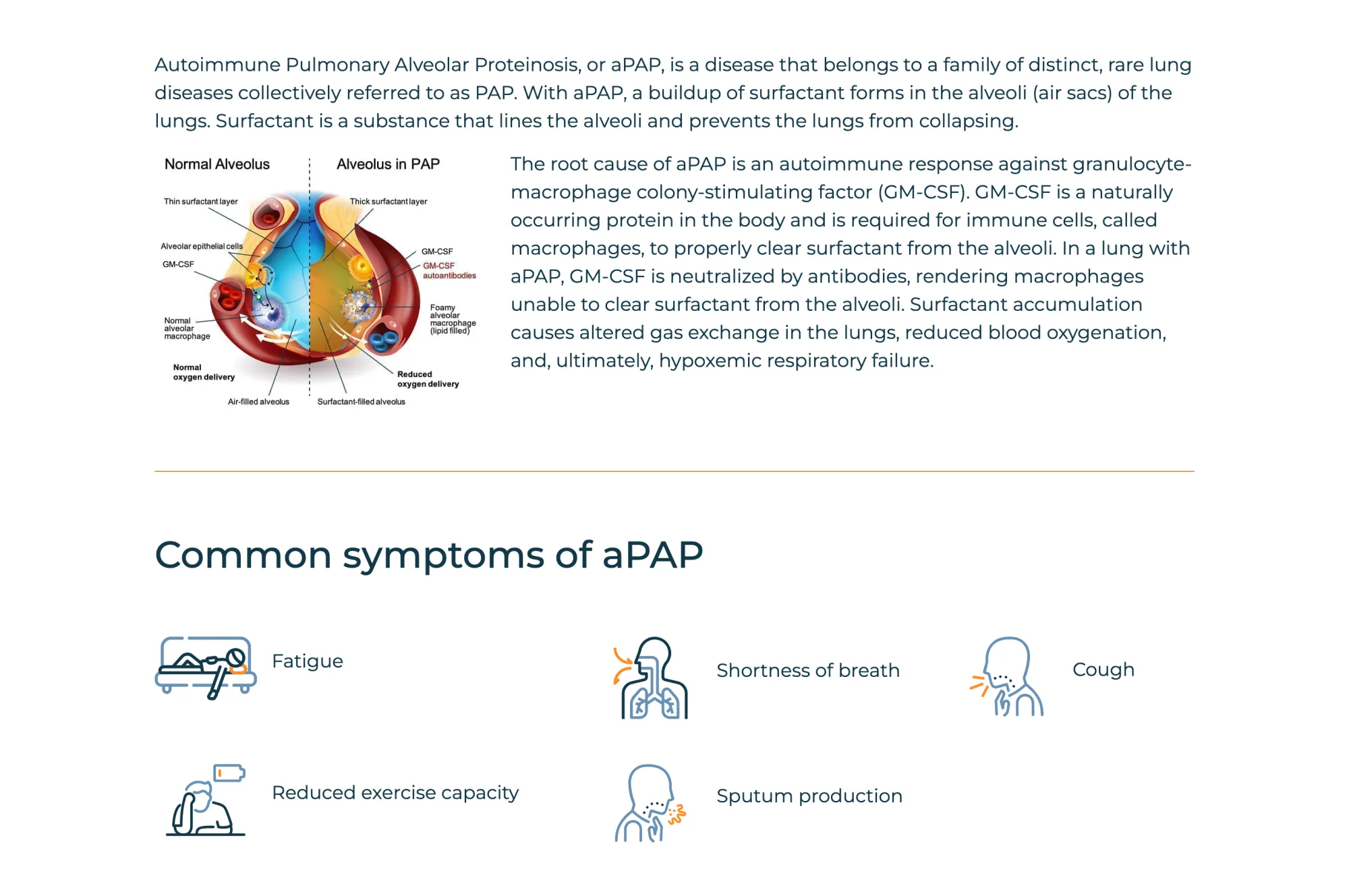
Savara gets FDA priority review for its drug, analyst raises target to $11, sees strong approval chances and upside.

JPMorgan backs Hims & Hers Health as a strong buy, boosted by GLP-1 drugs and big deals with Novo Nordisk and Eli Lilly.

Cannabis stocks fell after hype faded: US reform helps medical marijuana only, not full market, so investors took profits.

Regeneron will cut U.S. drug prices, offer free gene therapy for hearing loss, and align costs with other countries under a Trump deal.

FDA flagged fixable issues for GTx-104, not rejection. No new trials needed. IV drug may improve brain bleed ICU treatment.
OpenAI launched ChatGPT for doctors to help with notes and research. It’s free in the US, boosts AI use, and Doximity stock fell.

IKS Health is buying TruBridge to improve rural US healthcare using AI. Deal helps 2000 hospitals, closes 2026.

The Trump administration is expected to reclassify marijuana from Schedule I to Schedule III as soon as this week, easing research barriers after months of delay.

Amneal is buying Kashiv BioSciences for up to $1.1 billion to become a global leader in affordable biosimilar drugs. Strong Q1 results beat expectations and 2026 guidance raised.
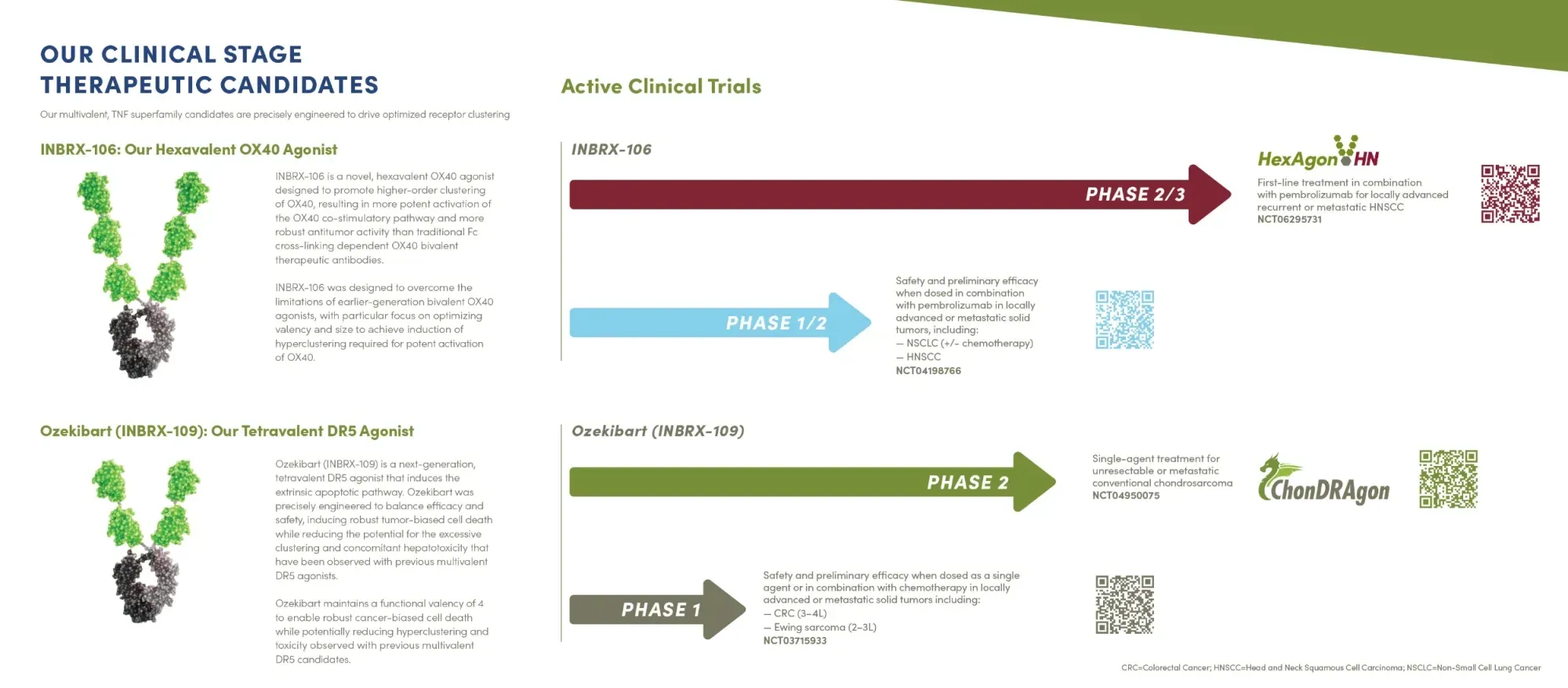
Inhibrx is drawing strong interest from Merck and other big pharma for its experimental cancer drug INBRX-106 that could boost Keytruda. Potential $9B spin-off value.
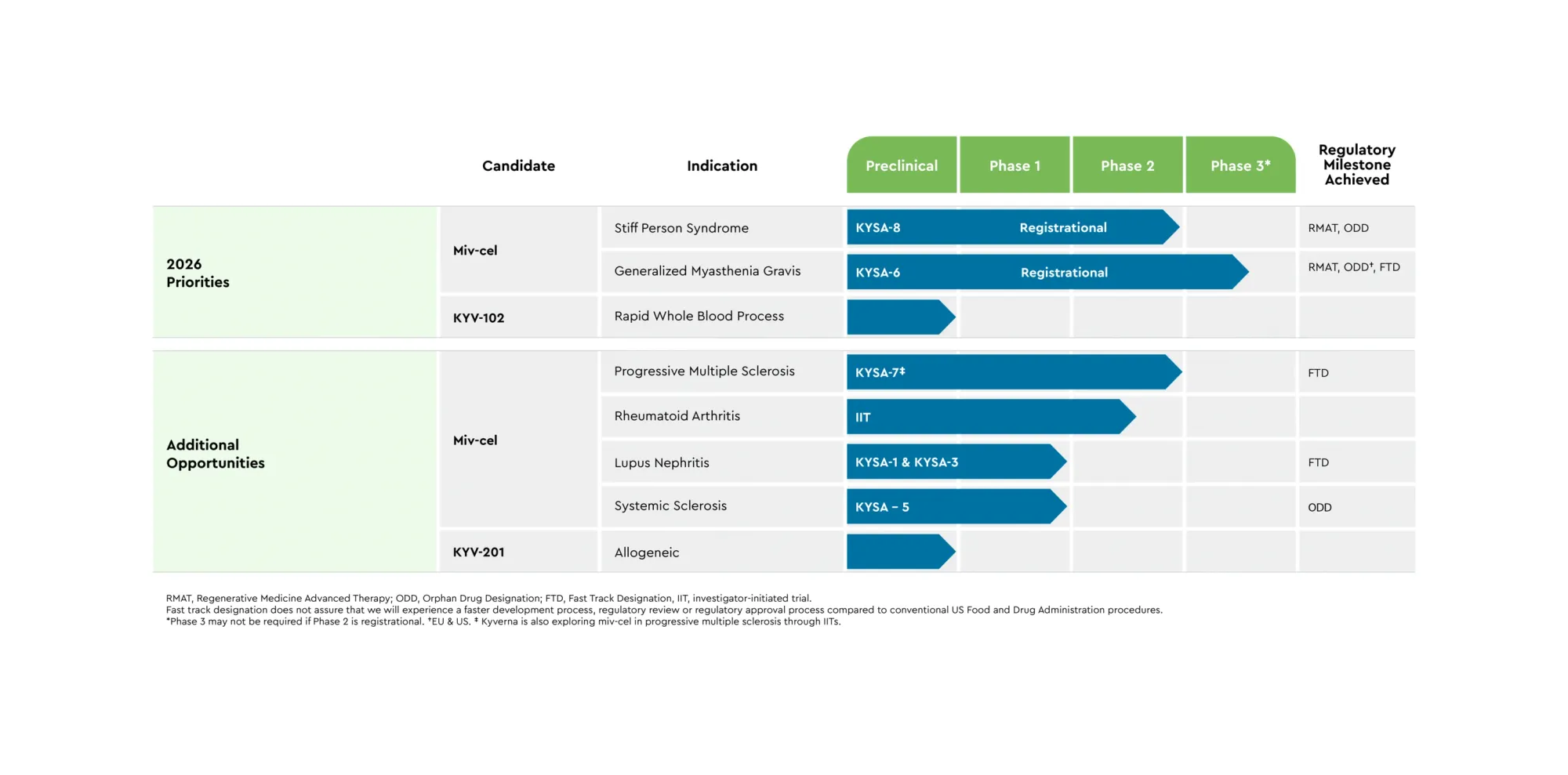
Kyverna’s CAR-T drug greatly improved rare SPS: patients walked 46% faster, many stopped meds; FDA filing planned 2026.
Private equity firms CVC Capital Partners and GTCR have submitted a joint offer to take medical device company Teleflex (TFX) private. Shares jumped on the news.

Merck’s new kidney cancer combos failed in Phase 3, missing key goals; analysts see mixed impact for Arcus and its drug.

Citi analysts say Amazon One Medical’s new $29 on-demand GLP-1 prescription renewals could pressure Hims & Hers, but they are not a full replacement for the telehealth weight-loss giant.

Harmony Biosciences wins key patent lawsuit, protecting Wakix from generics until ~2030; analyst stays positive with $50 target.

Ionis drug zilganersen showed promise for rare brain disease (Alexander disease), improving symptoms; FDA decision due Sept 2026.

BTIG raises CMPS to Buy, $14 target; stock jumps 42% after Trump boosts psychedelic research funding and faster FDA paths.

UnitedHealth Group beat expectations, raised outlook, cut costs, and grew steadily—showing a solid turnaround and boosting investor confidence.

Achieve Life Sciences gets Buy rating as its quit-smoking drug cytisinicline shows strong results; FDA decision due June 2026.

BBOT receives U.S. FDA Fast Track Designation for BBO-11818, its pan-KRAS inhibitor for advanced KRAS-mutant pancreatic cancer. Phase 1 data expected H2 2026.
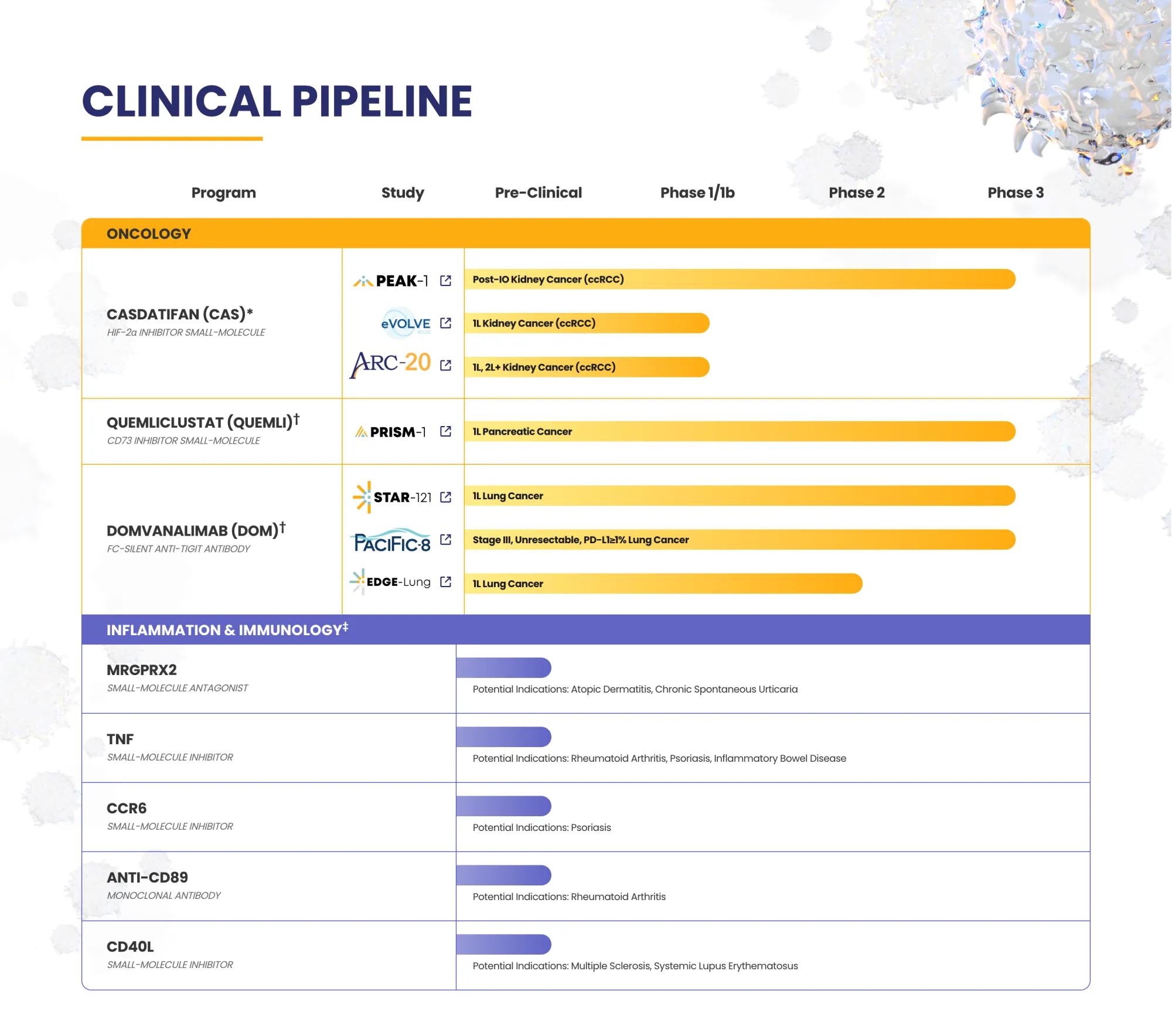
Arcus stops a lung cancer trial early after poor outlook, ends a partner study, and gains more control as Gilead scales back the deal.

Cantor is very bullish on CADL, sees big undervaluation and CAN-2409 as a major drug with huge growth potential.
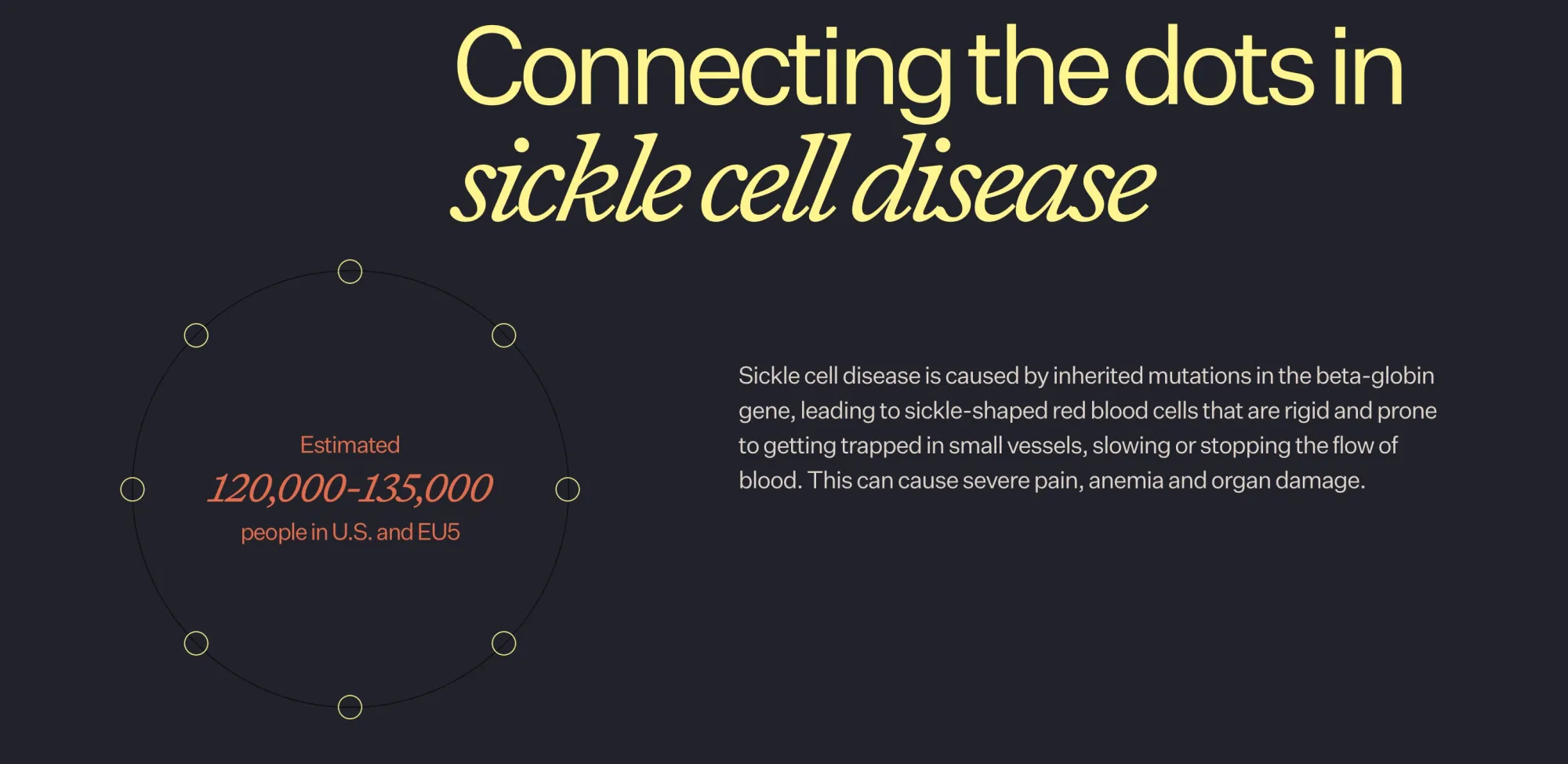
Novo Nordisk’s new pill cuts sickle cell pain crises, boosting outcomes and sending Agios Pharma shares down after strong trial results.

Nektar drug showed promising hair regrowth in severe alopecia areata after 52 weeks, with good safety and twice-monthly shots.
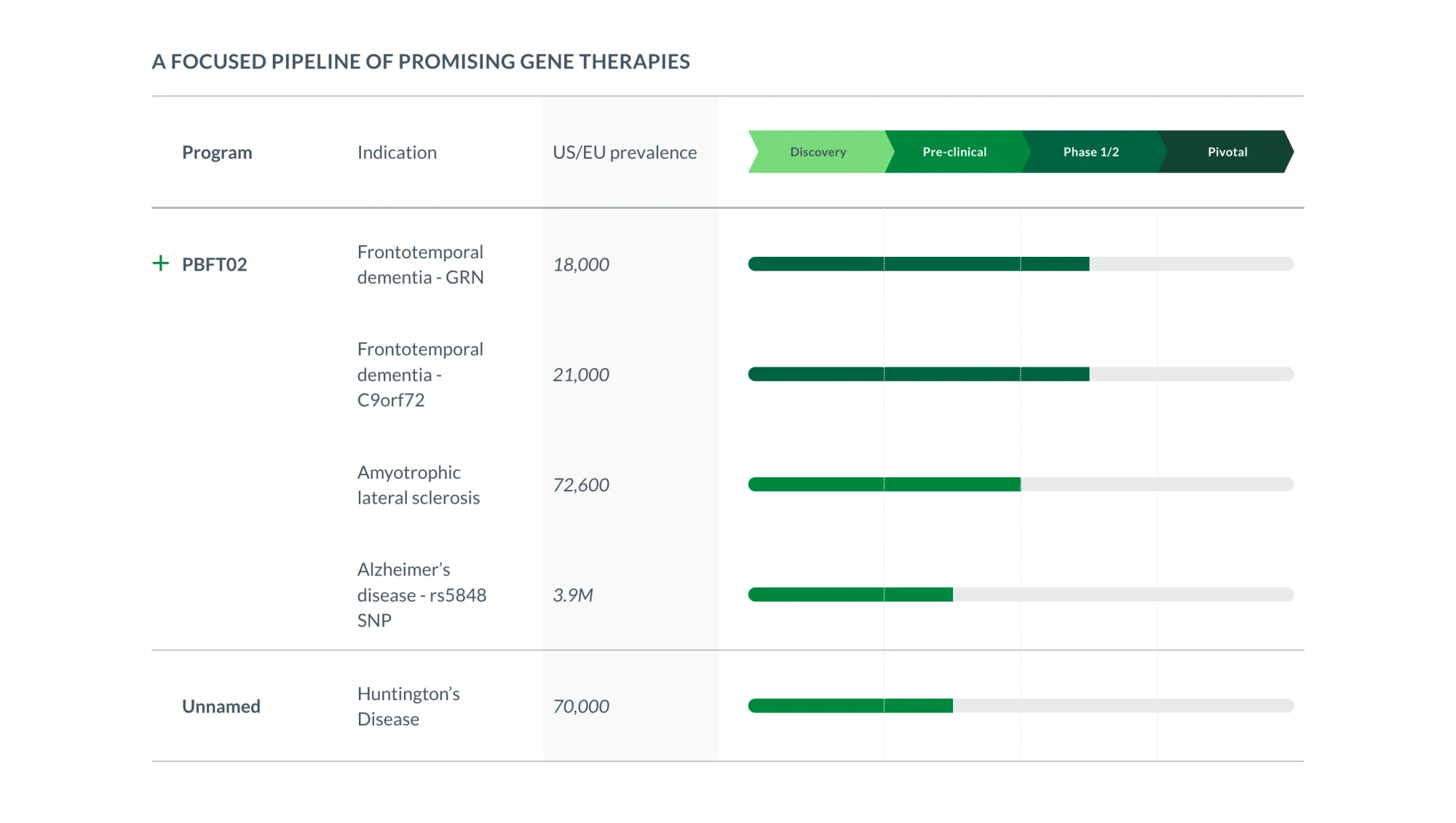
Passage Bio fell after the FDA demanded a larger trial; the drug shows promise for brain disease, but its future is now costlier and slower.
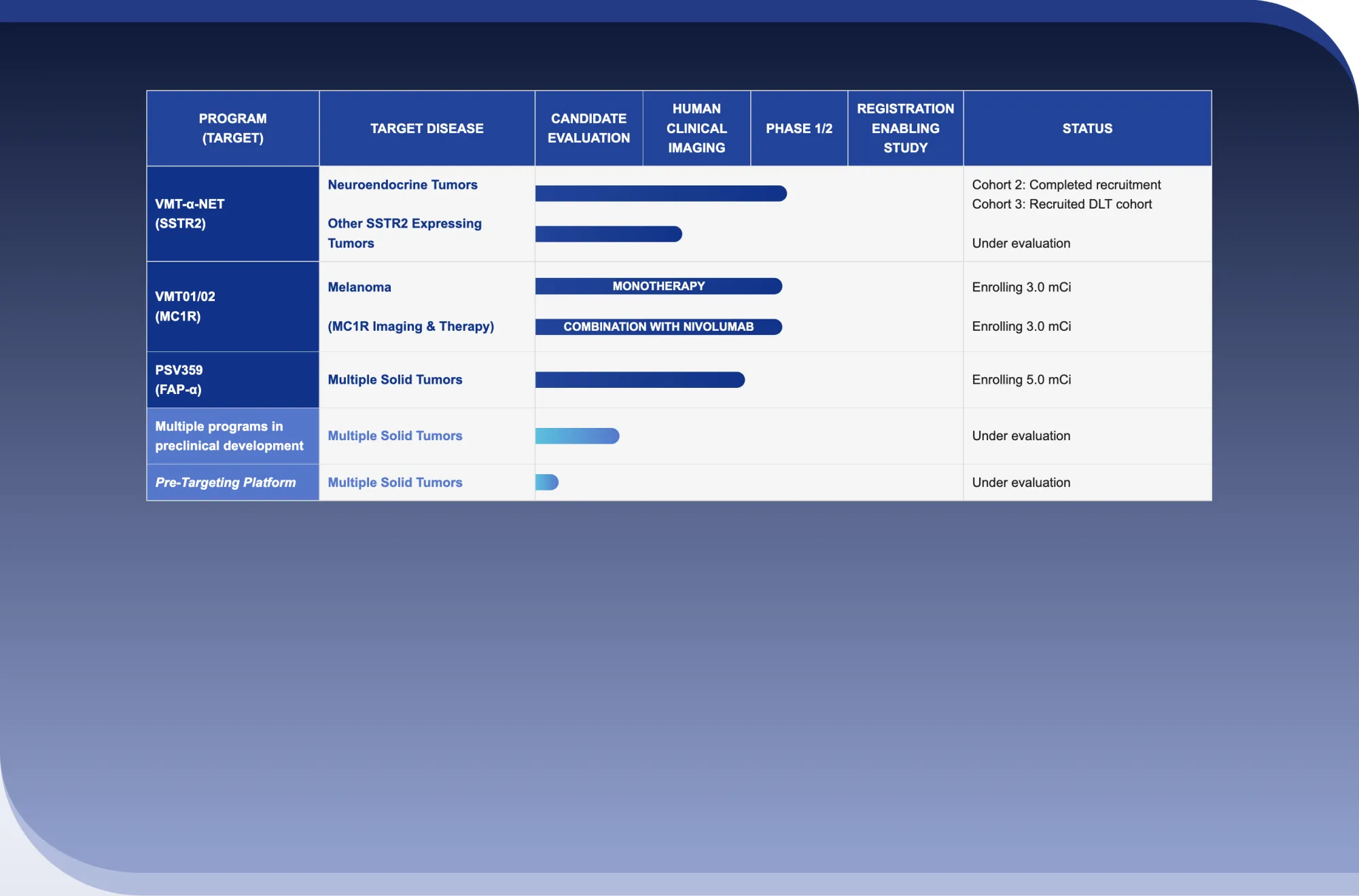
New alpha therapy for advanced NETs shows good safety and early tumor shrinkage in ~43% patients, with encouraging results so far.
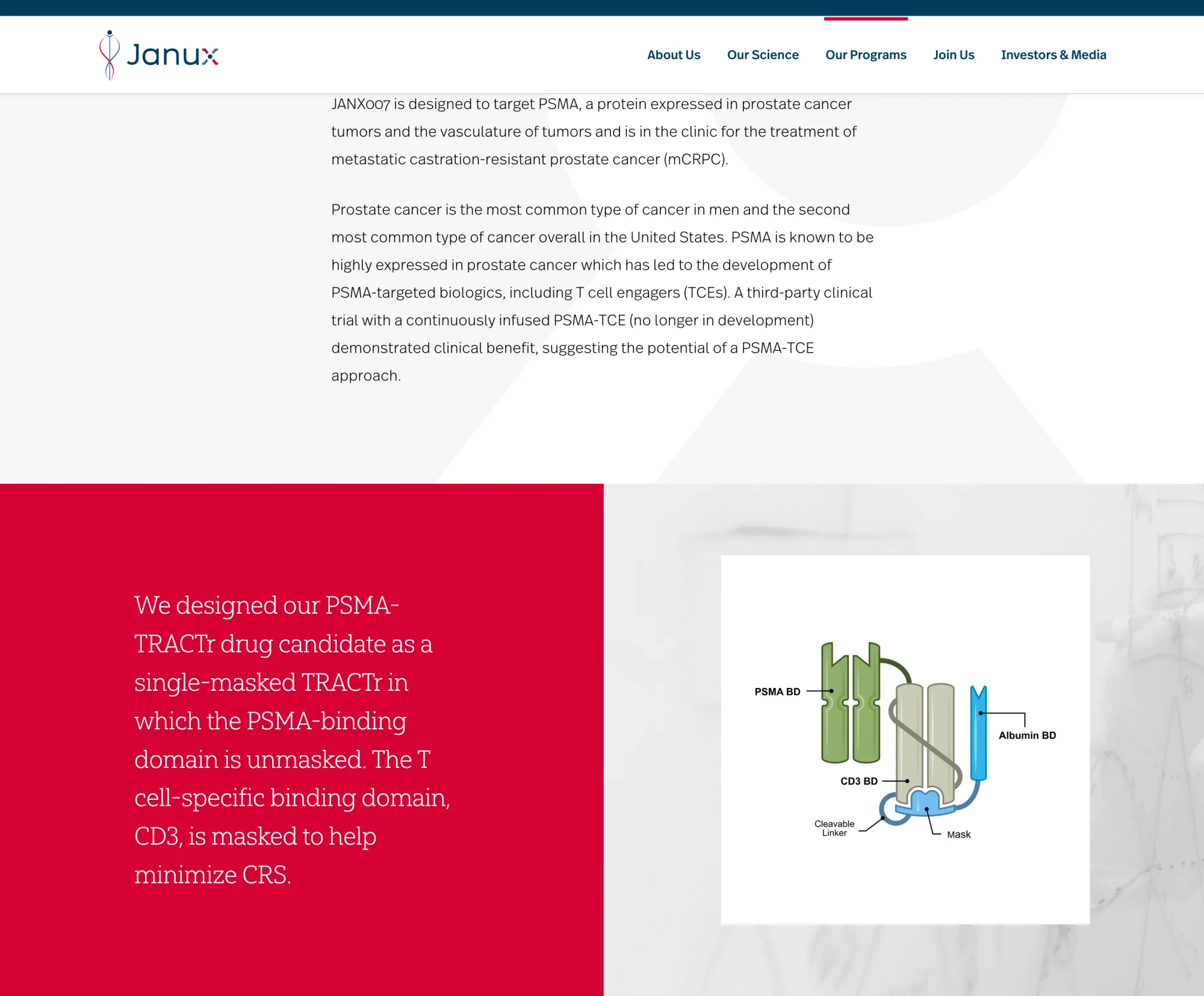
Barclays cut Janux Therapeutics to Underweight, citing doubts on JANX007 and limited near-term upside despite long-term potential.