Scholar Rock Shares Jump as Apitegromab BLA Resubmitted to FDA for SMA Treatment
Scholar Rock resubmits its BLA for apitegromab to treat SMA. FDA acceptance expected in 30 days, PDUFA target late September 2026.

Scholar Rock resubmits its BLA for apitegromab to treat SMA. FDA acceptance expected in 30 days, PDUFA target late September 2026.
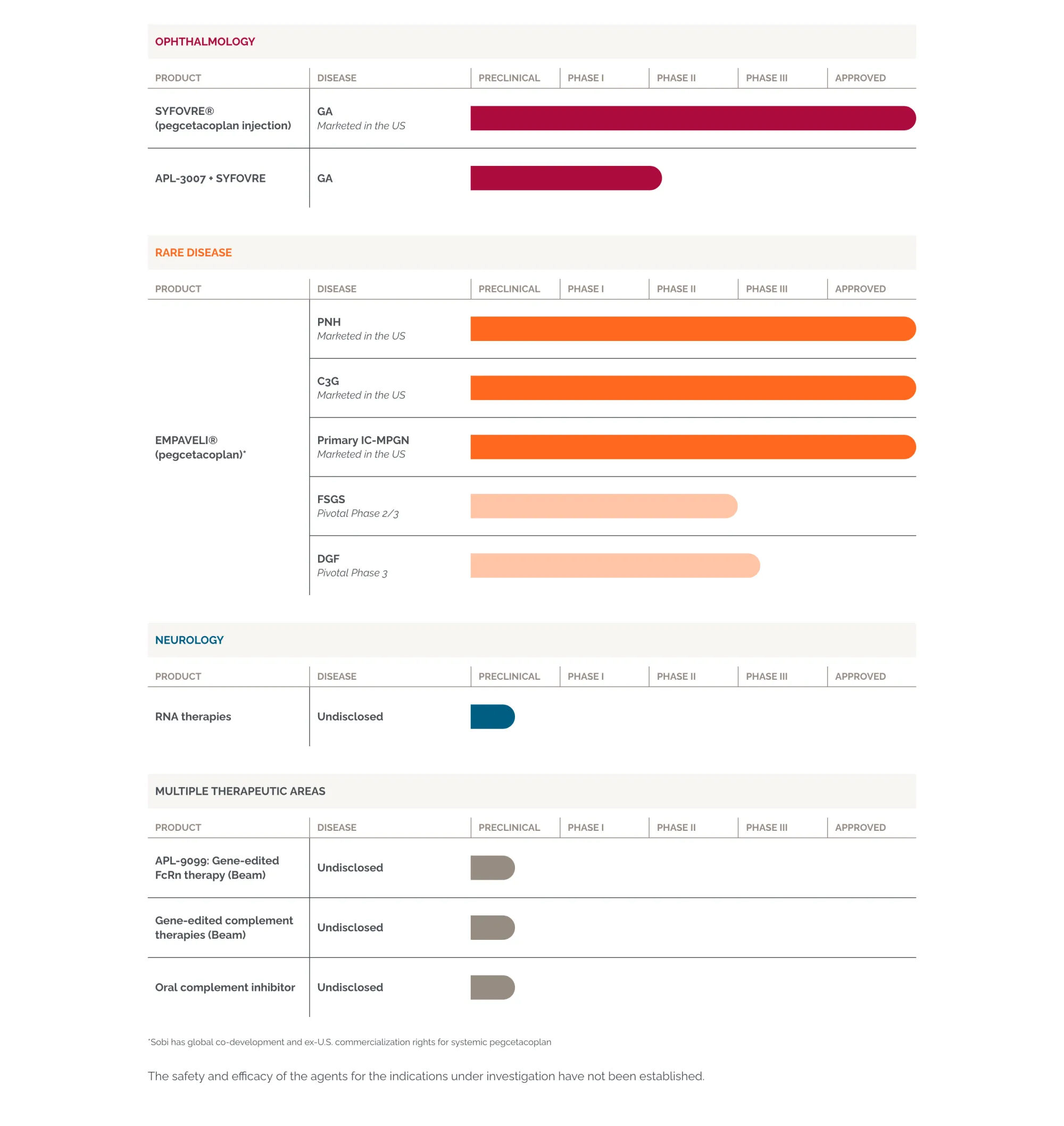
Biogen is acquiring Apellis for $5.6 billion, adding two approved drugs (Syfovre & Empaveli) for eye disease and rare kidney conditions. Boosts growth and nephrology pipeline.

Eli Lilly is acquiring Centessa in a $7.8 billion deal to develop breakthrough OX2R drugs for narcolepsy, idiopathic hypersomnia, and excessive daytime sleepiness.

PepGen’s latest low-dose (5 mg/kg) DM1 data shows minimal splicing correction and almost no functional improvement over placebo.
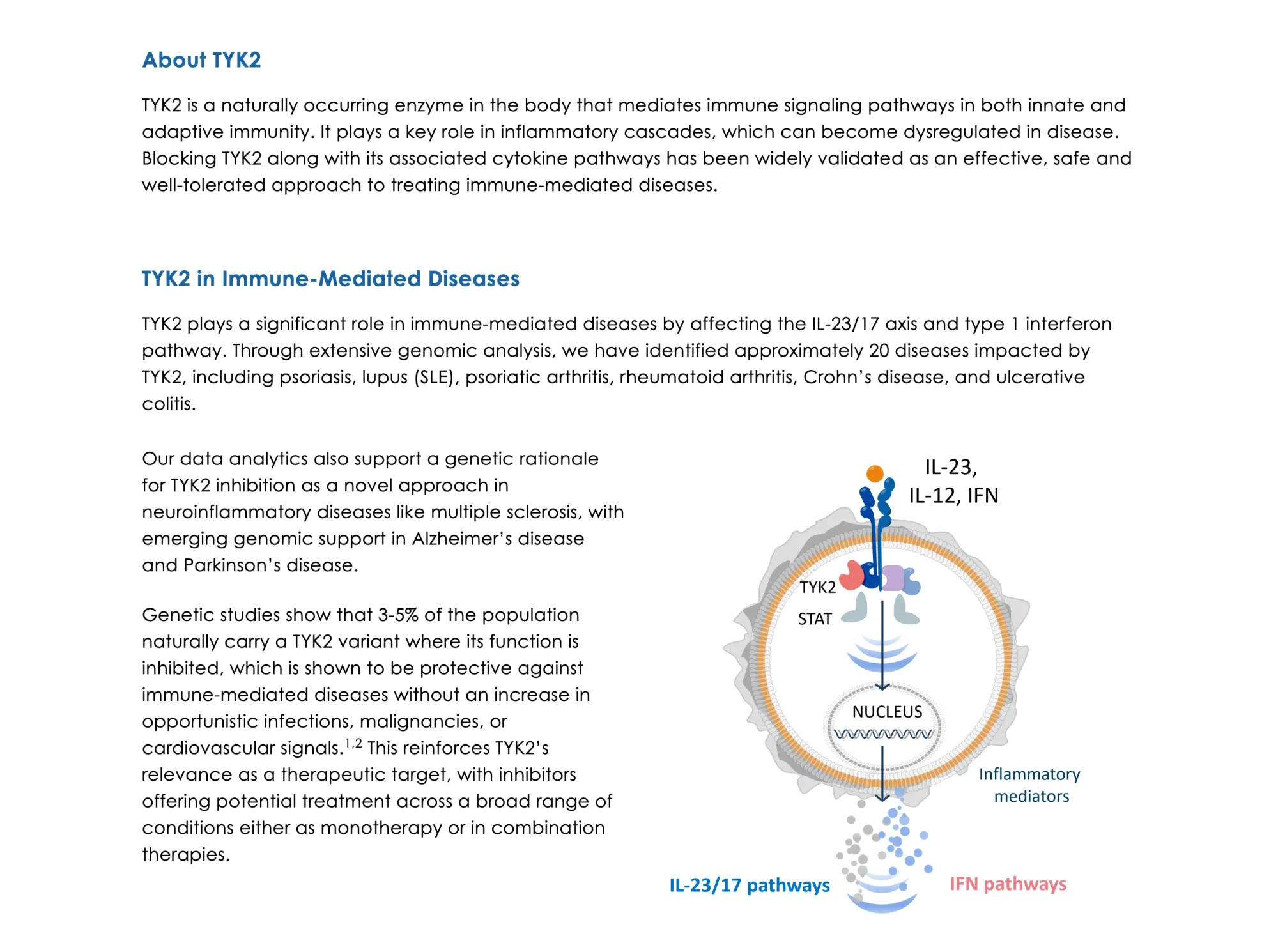
Alumis shares plunged after strong Phase 3 psoriasis data for envudeucitinib. HC Wainwright cuts price target to $25 citing rising competition.
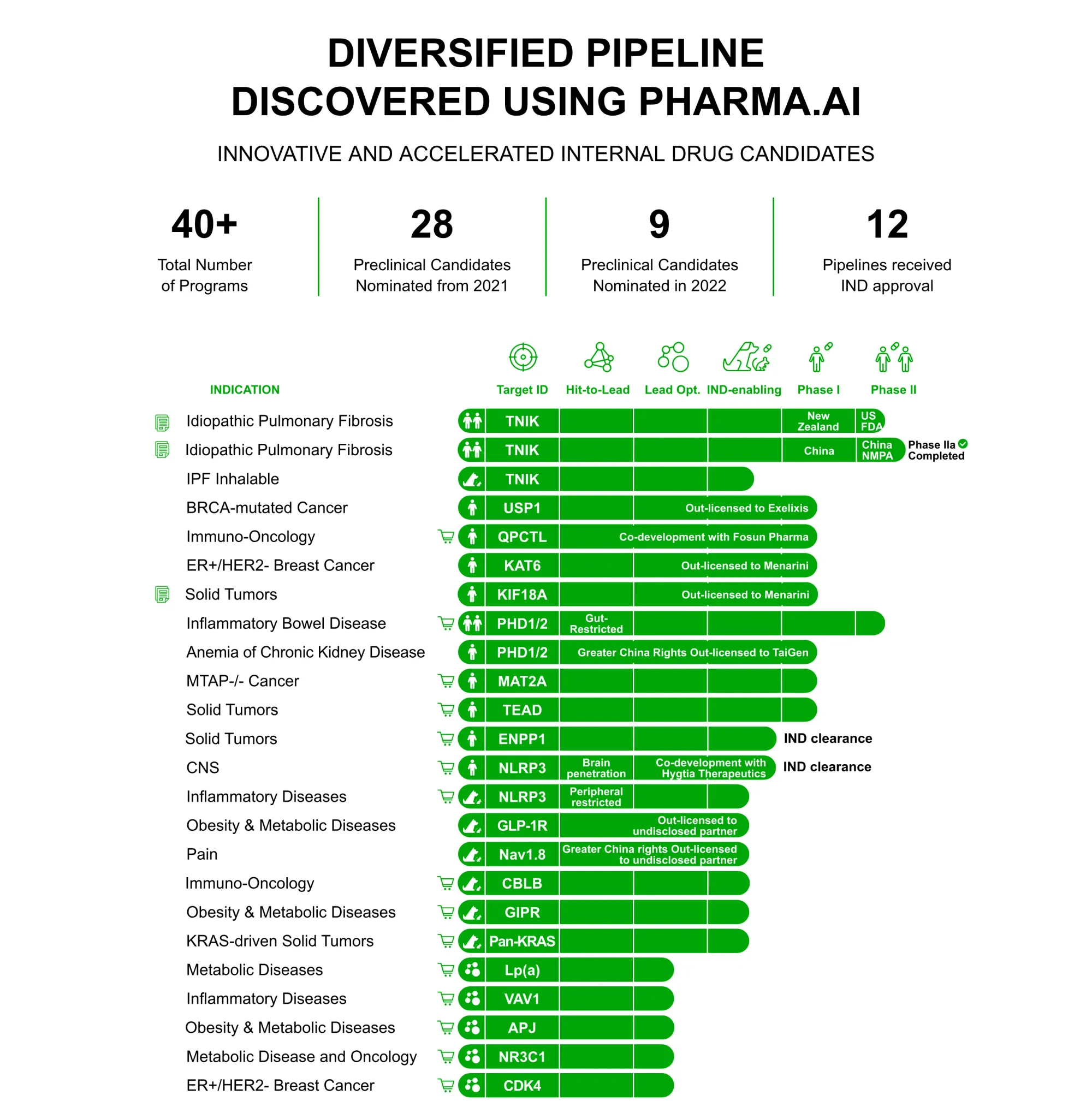
Eli Lilly has signed a $2.75 billion deal with Hong Kong-listed AI biotech Insilico Medicine for a GLP-1 diabetes drug.

Wells Fargo says Boston Scientific’s CHAMPION-AF trial met all endpoints for WATCHMAN FLX but calls results “good, not great” due to slightly higher stroke rate vs blood thinners.
Morgan Stanley upgrades Insmed to Overweight and raises price target to $212.

Aurinia Pharmaceuticals has agreed to acquire Kezar Life Sciences for $6.955 in cash per share plus a Contingent Value Right (CVR).

The United Therapeutics TETON-1 trial shows that inhaled Tyvaso preserves lung function by 130 mL versus placebo and reduces disease progression. Positive results were seen across all patient groups, with an FDA filing planned for this summer.
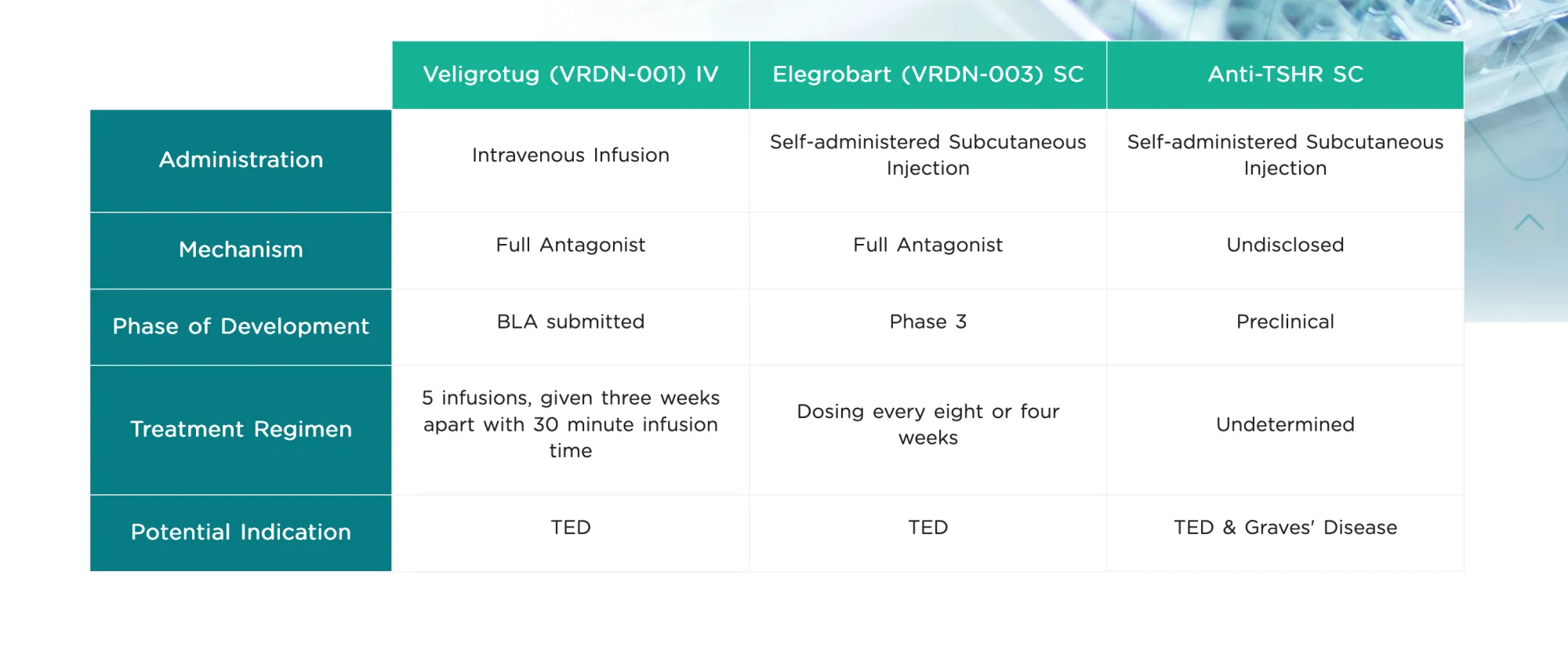
Viridian highlights 54% proptosis response for Elegrobart in TED, but secondary endpoints like inflammation and diplopia showed limited gains.

Zymeworks receives U.S. FDA Fast Track Designation for ZW191, its FRα-targeting antibody-drug conjugate for advanced platinum-resistant ovarian cancer.

Rocket Pharmaceuticals won FDA approval for Kresladi and a valuable Priority Review Voucher, yet the stock dropped. Analysts explain the "sell the news" reaction, dilution concerns, and why they remain bullish with Outperform ratings.
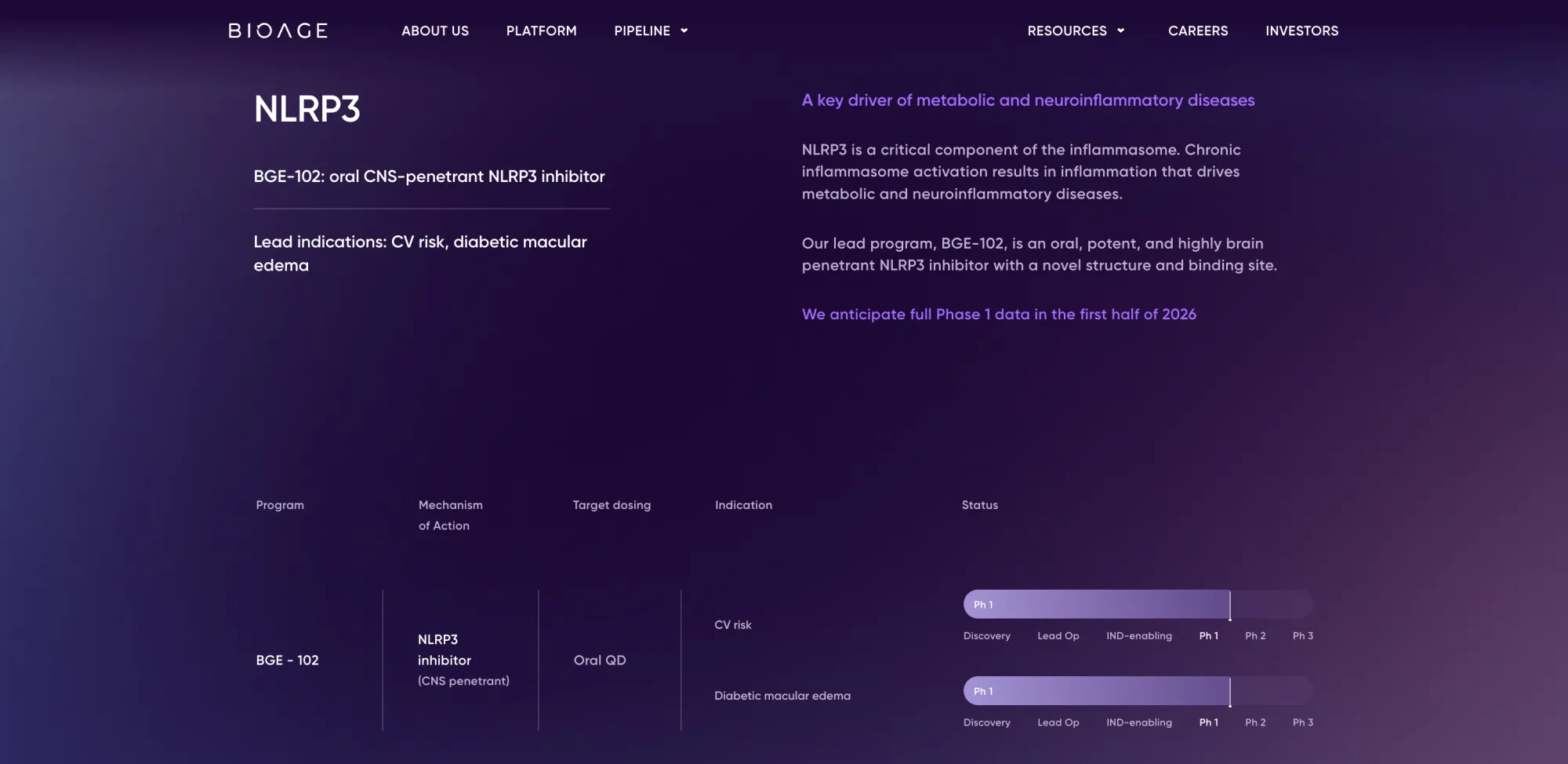
Analysts are bullish on lead asset BGE-102, an oral NLRP3 inhibitor for cardiovascular risk in obese patients.

AstraZeneca’s experimental drug tozorakimab significantly reduced COPD flare-ups in two Phase 3 trials. Promising new hope for millions of patients with chronic lung disease.

The FDA has approved KRESLADI, Rocket Pharma’s one-time gene therapy for children with severe LAD-I — a rare, life-threatening immune disorder.
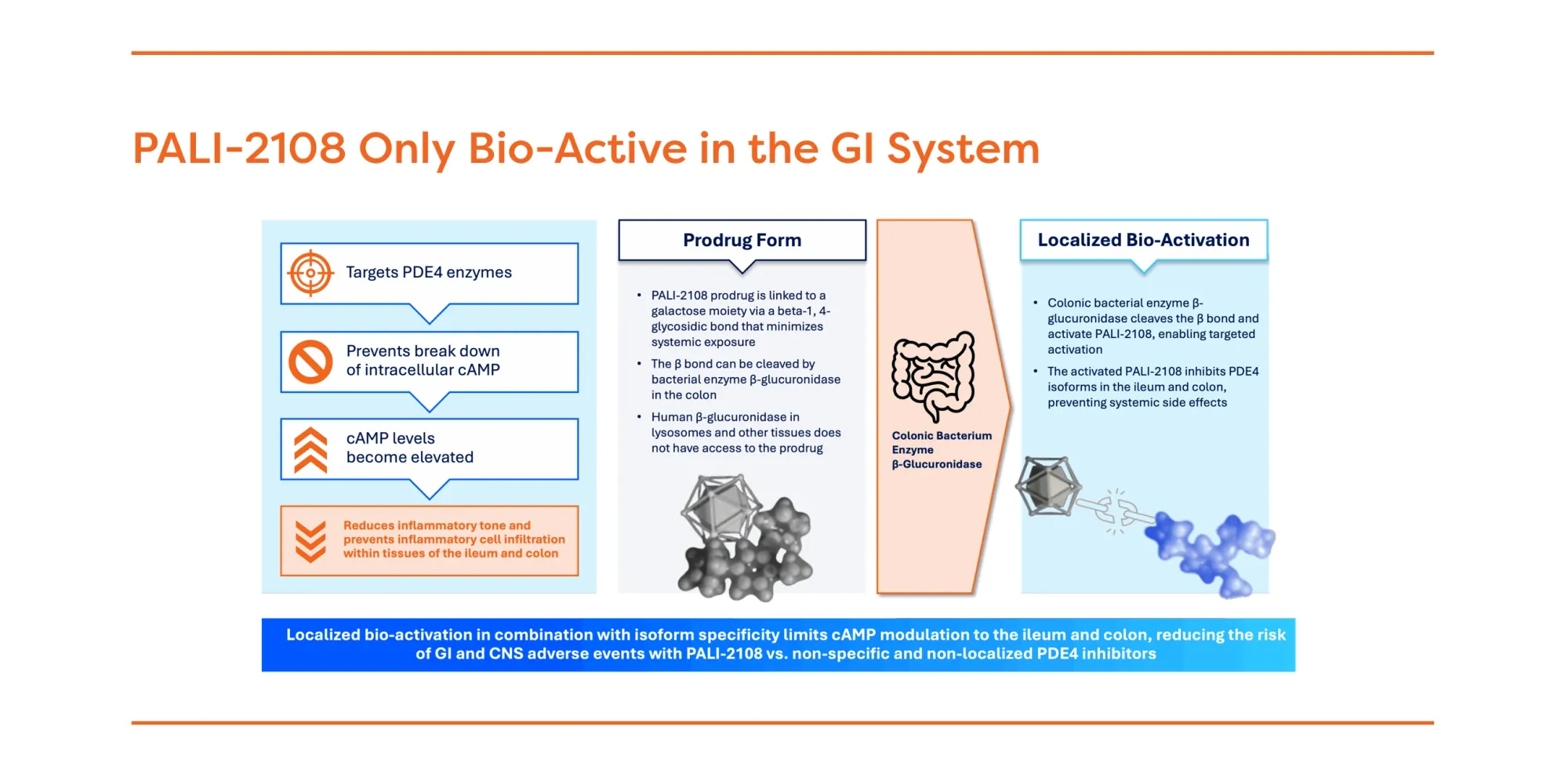
Analysts say PALI-2108 offers better safety and efficacy for ulcerative colitis compared to older PDE4 inhibitors.

Kodiak Sciences shares surged after Zenkuda hit its primary endpoint in the GLOW2 Phase 3 study for diabetic retinopathy.
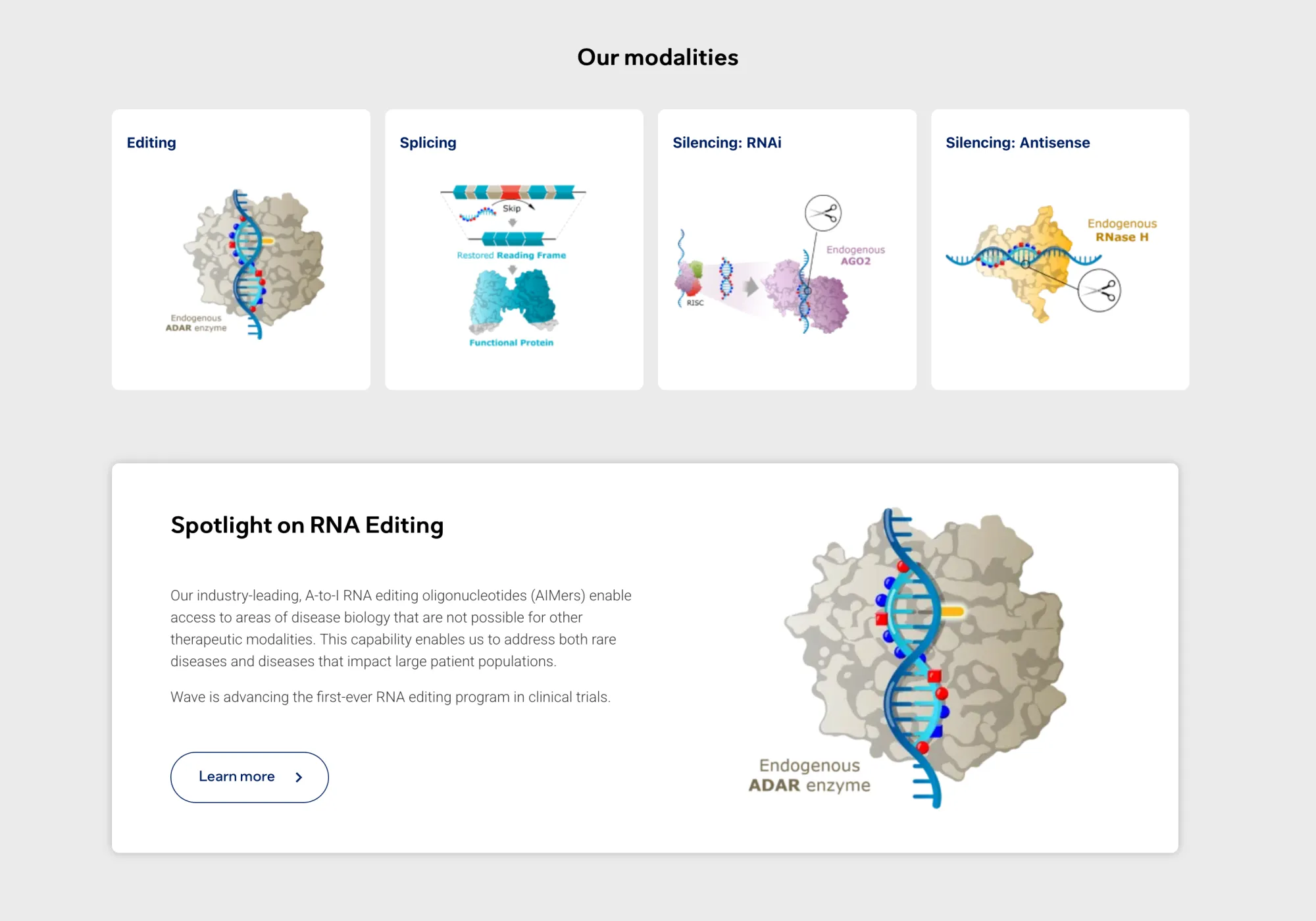
Wave Life Sciences' WVE-007 (INHBE-targeted) showed ~5% total fat reduction and 14% visceral fat drop with muscle preserved after one dose. Early results are modest (1% weight loss), but support infrequent dosing as potential GLP-1 complement.

Emergent BioSolutions wins $54M U.S. government contract for VIGIV plus $6.6M international orders for ACAM2000 smallpox vaccine.

Merck’s $6.7B acquisition of Terns Pharmaceuticals highlights huge potential in CML therapies. Why analysts see Enliven’s ELVN-001 as a best-in-class winner with strong takeover appeal.

The FDA has approved Denali’s AVLAYAH, the first brain-penetrating enzyme therapy for Hunter syndrome (MPS II) in nearly 20 years.
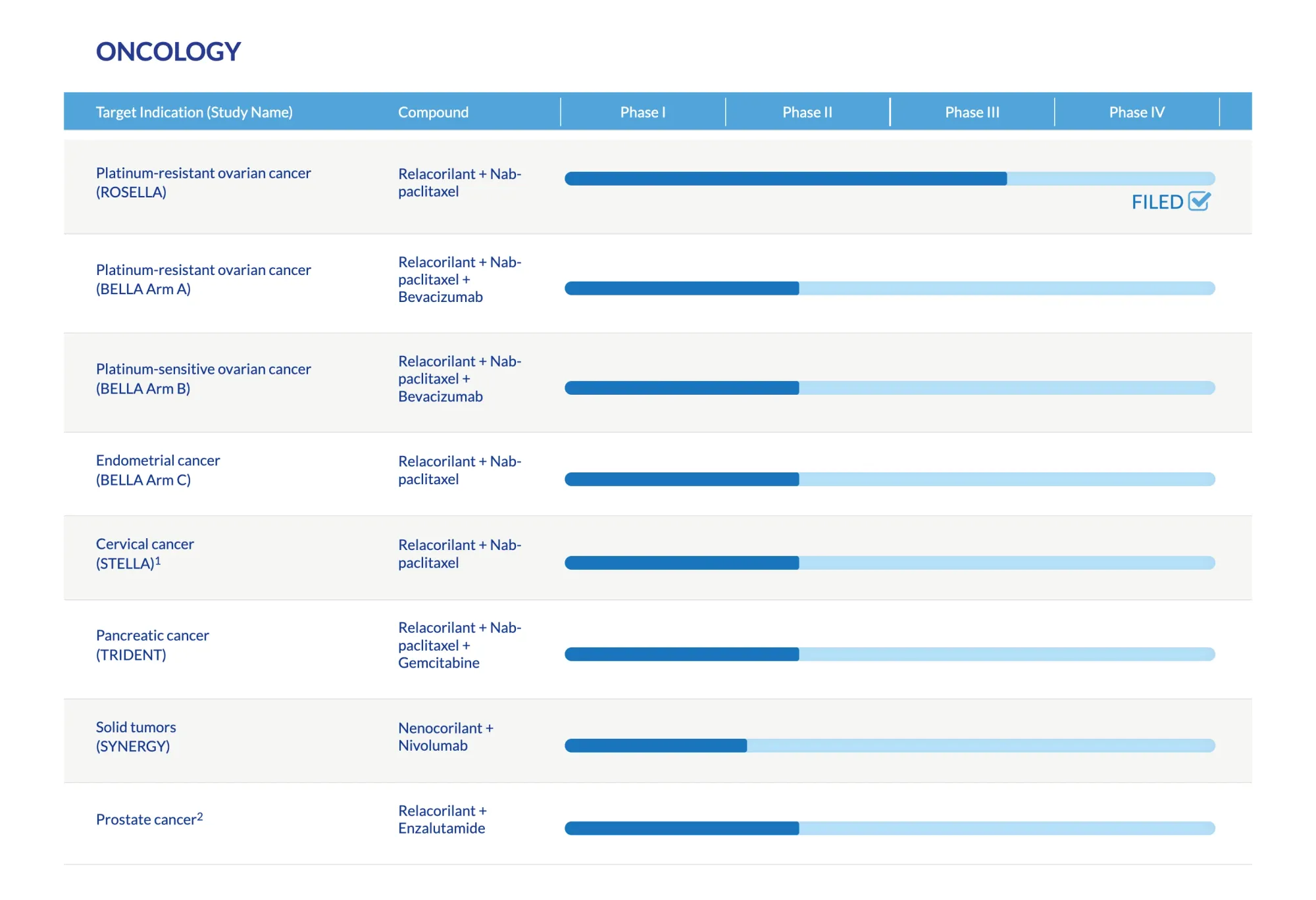
The FDA has approved Corcept's relacorilant (Lifyorli) in combination with nab-paclitaxel for platinum-resistant ovarian, fallopian tube, or peritoneal cancer, showing improved survival by several months.

BofA starts coverage on Bicara Therapeutics with a Buy rating and $35 price target. Ficerafusp alfa shows strong potential in head and neck cancer with clear differentiation over standard care.

Sarepta just shared first test results for two new medicines aimed at rare muscle diseases FSHD1 and DM1. Early data show the drugs reach high levels in muscle safely, with no serious side effects. Shares jumped.
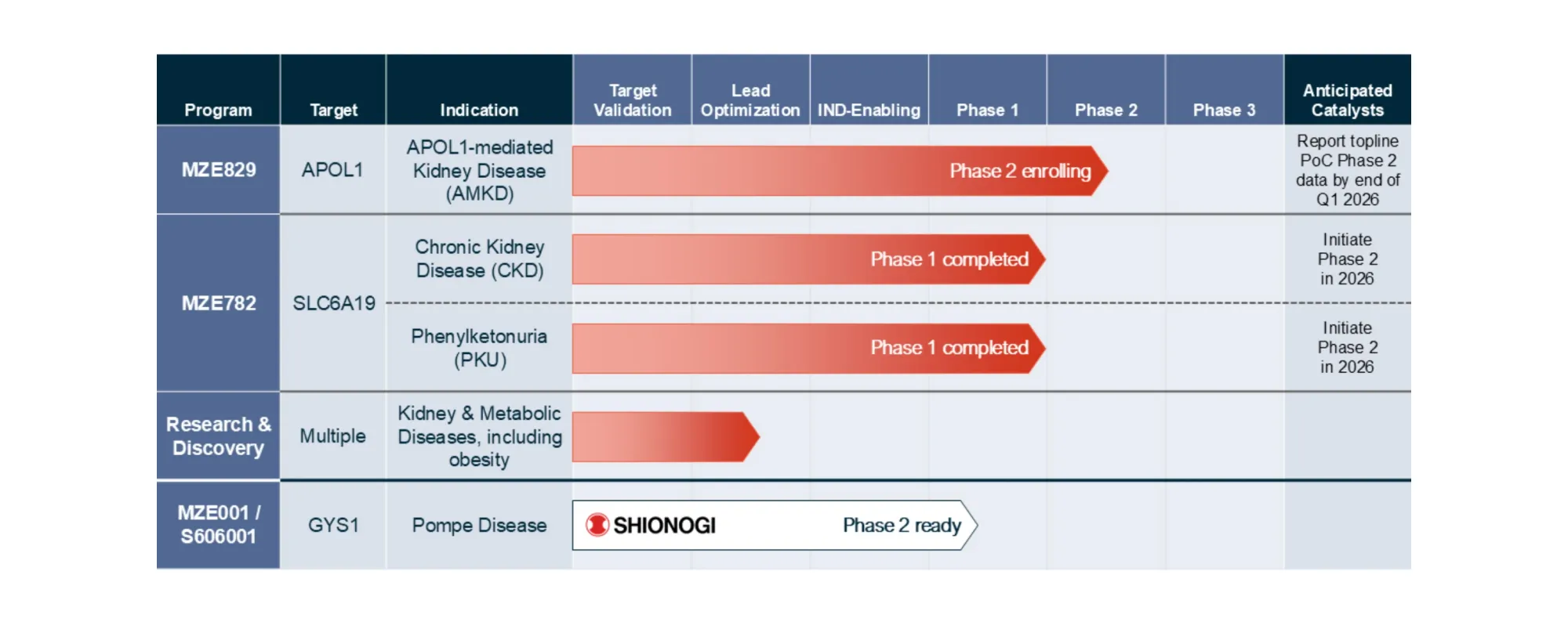
Maze’s new kidney pill MZE829 cut harmful protein in urine by 35.6% in its Phase 2 trial — including patients with diabetes. Stock still fell 30% as analysts say “don’t compare it to Vertex” but remain very bullish.

Anavex Life Sciences has withdrawn its EU marketing application for blarcamesine as an add-on therapy for early Alzheimer’s. Shares fell sharply after EMA committee feedback.

Morgan Stanley initiated coverage on Immix Biopharma with an Overweight rating and $20 price target.

BofA upgrades Acadia Pharmaceuticals (ACAD) to Buy from Neutral with a $29 price target.

Merck is buying Terns Pharmaceuticals for $6.7 billion to add TERN-701, a promising new oral drug for chronic myeloid leukemia (CML). The move strengthens its cancer pipeline as Keytruda patent expiry nears in 2028.

Grifols has approved a U.S. IPO of a minority stake in its Biopharma business.
Gilead’s up to $2.2B acquisition of Ouro Medicines highlights strong potential for Cullinan Therapeutics’ velinotamig. Clear Street maintains Buy rating with $33 PT.
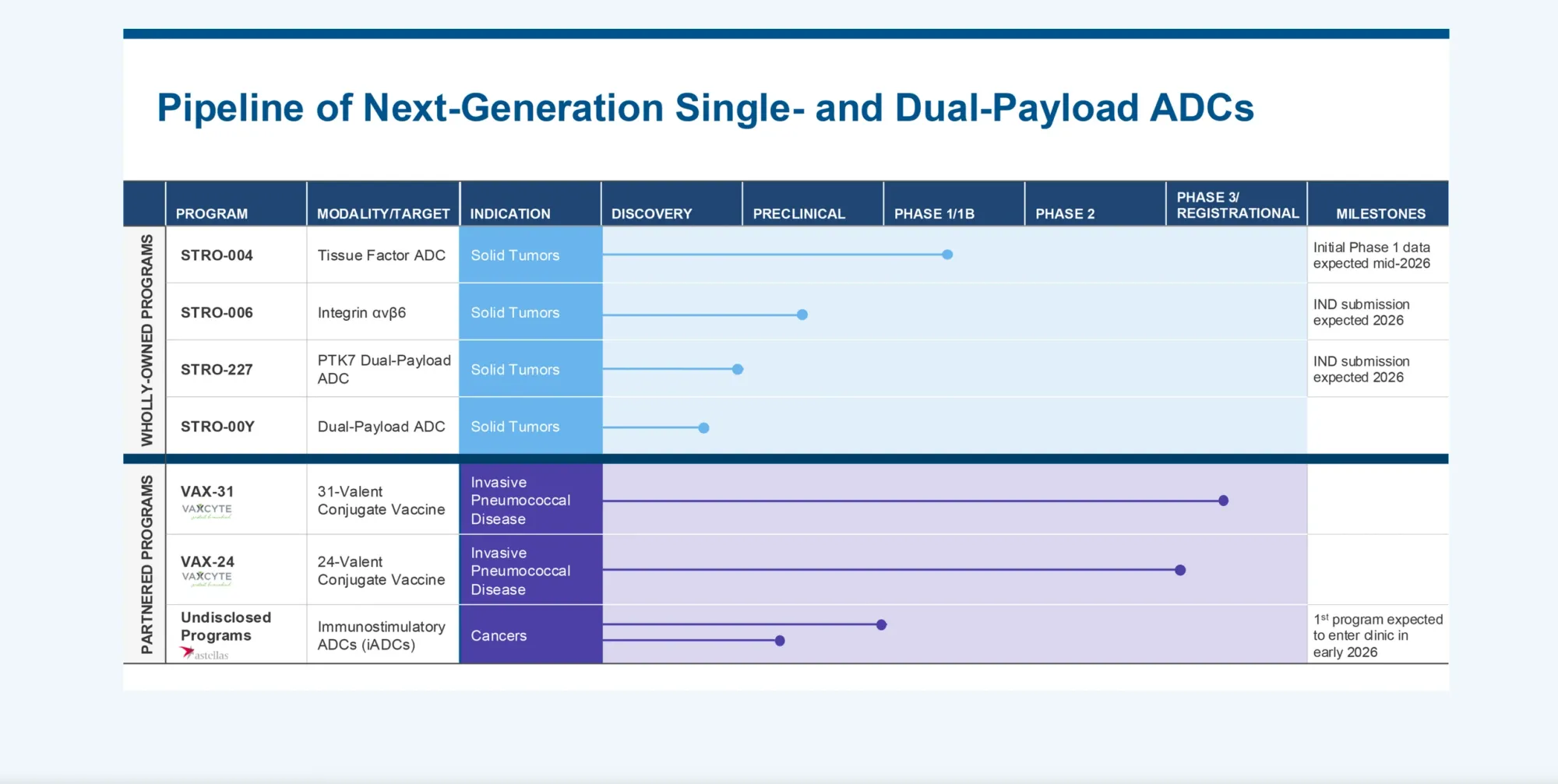
Wells Fargo upgrades to Overweight with $27 PT and H.C. Wainwright raises to Buy with $28 target on strong STRO-004 progress.
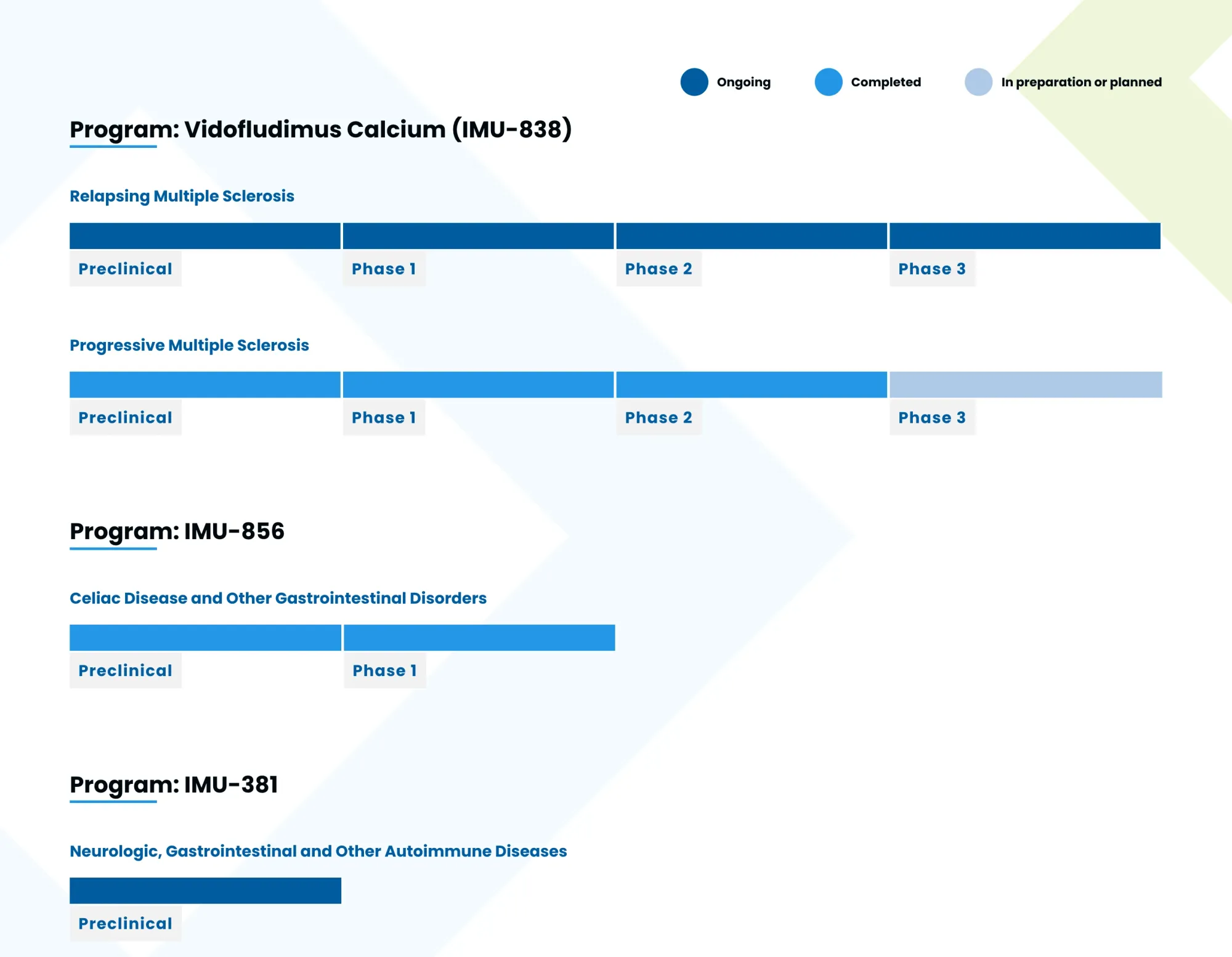
Guggenheim launches coverage on Immunic at Buy with a $7 target. The dual-action MS drug vidofludimus calcium targets both inflammation and nerve protection – Phase 3 data could drive major gains.
ImmunityBio shares dropped 20% after the FDA issued a warning letter accusing the company of false claims that ANKTIVA cures all cancers.

Goldman Sachs downgraded Ultragenyx from Buy to Neutral and slashed its price target to $25 after setrusumab Phase 3 miss in osteogenesis imperfecta. GTX-102 in Angelman syndrome now key focus, but risks remain.

Karyopharm’s (KPTI) SENTRY trial delivers strong spleen reduction and promising survival data with selinexor + ruxolitinib — though symptom improvement was not superior. $30M cash boost extends runway into late 2026.

Gilead to acquire Ouro Medicines for $1.675B upfront + $500M milestones, adding clinical-stage BCMAxCD3 therapy gamgertamig for severe autoimmune conditions. Partnership with Galapagos announced.
Gilead Sciences eyes up to $2B deal for autoimmune biotech Ouro Medicines, with $1.5B upfront + milestones, amid strong stock & deal spree.

Damora has appointed biotech leader Jennifer Jarrett as President and Chief Executive Officer, effective March 30, 2026. Two industry veterans also join the Board of Directors.

Wolfe Research upgrades Celldex Therapeutics to Outperform from Peer Perform, citing stronger 2026 catalysts and depressed valuation for rebound potential.

Pfizer and Valneva reported their Lyme disease vaccine demonstrated 73.2% efficacy in the Phase 3 VALOR trial. While it missed the primary statistical target due to fewer cases, strong results support upcoming regulatory submissions.

Insmed announced positive topline results from the Phase 3b ENCORE study of Arikayce in newly diagnosed MAC lung disease. The drug showed statistically significant improvements in respiratory symptoms and culture conversion rates vs standard therapy.
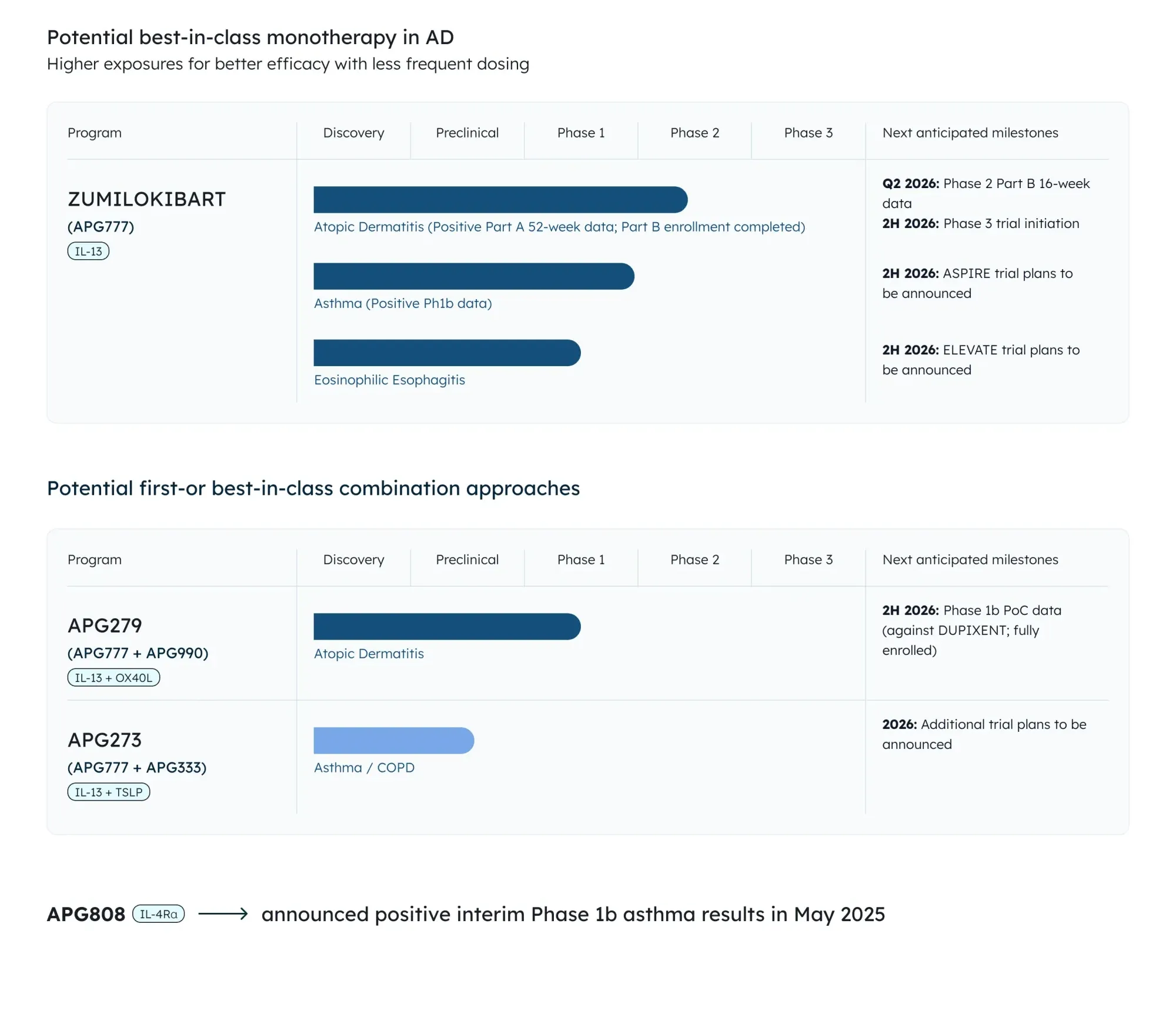
Apogee Therapeutics’ Phase 2 data show zumilokibart maintains strong skin clearance and itch relief in moderate-to-severe eczema for 52 weeks with dosing every 3 or 6 months.

Guggenheim starts SLDB coverage with Buy rating and $26 price target. SGT-003 gene therapy shows safer profile vs Sarepta's Elevidys, eyes accelerated FDA approval.

Oppenheimer upgrades FIGS stock to Outperform from Perform with $22 target, citing sustained recovery, improved ops, and undervalued long-term growth in healthcare apparel.

Rhythm Pharmaceuticals receives FDA approval for IMCIVREE (setmelanotide) — the first and only treatment for acquired hypothalamic obesity.
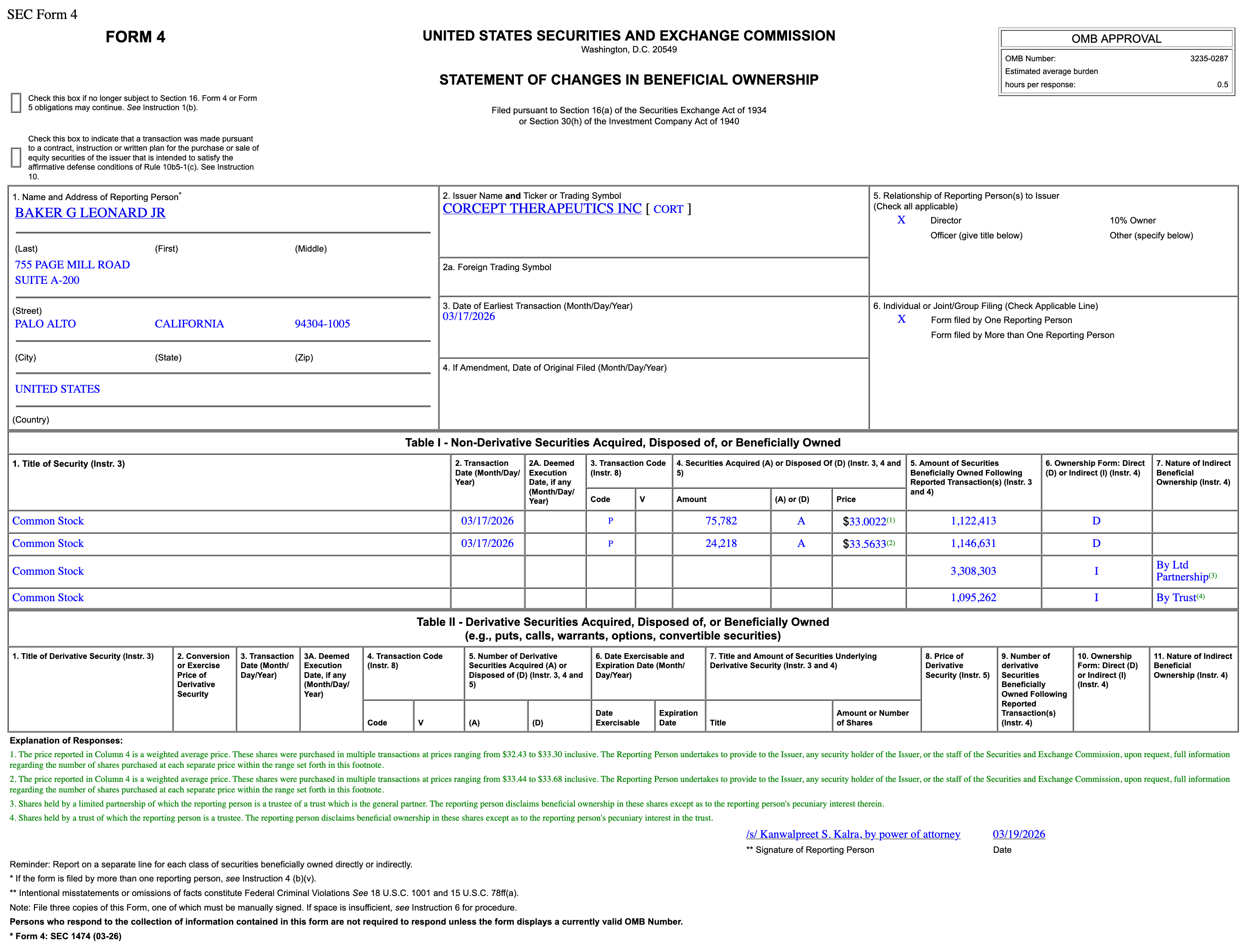
Corcept Therapeutics director Leonard Baker Jr. purchased 100,000 common shares at ~$33 each in open-market buys.

Roche halts emugrobart development, clearing path for Scholar Rock's apitegromab to dominate SMA muscle therapy market. Cantor sees multi-billion potential.

Eli Lilly’s retatrutide delivered up to 2.0% A1C reduction and 16.8% weight loss (36.6 lbs) in its first Phase 3 trial for type 2 diabetes. Analysts call it a key growth driver for Lilly with strong results vs Mounjaro.

Canaccord Genuity starts coverage on MapLight Therapeutics (MPLT) with Buy rating and $35 price target.
Activist investor Elliott Investment Management takes major stake in Align Technology, Invisalign maker, to push for stock price gains amid post-pandemic slump.
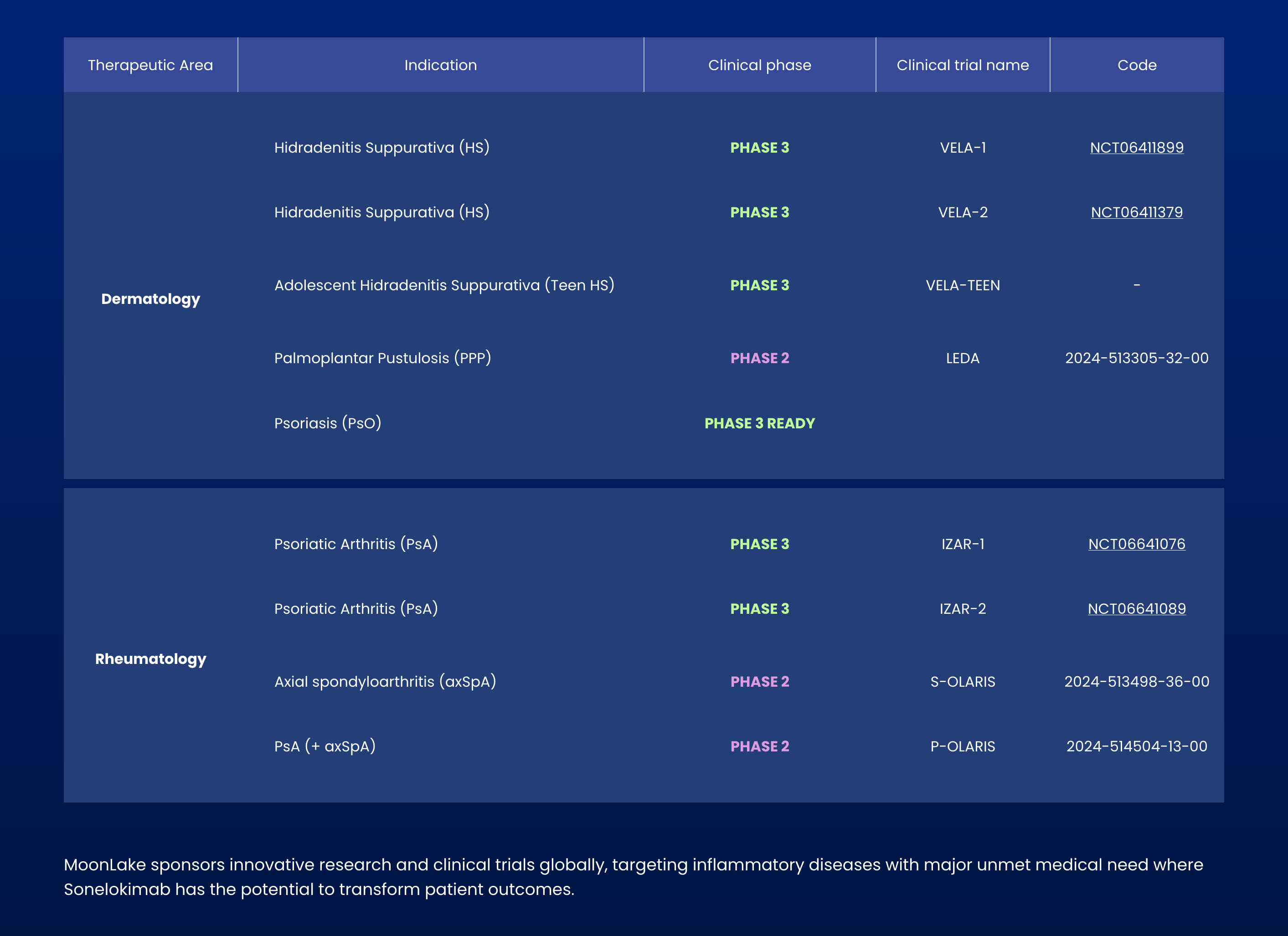
Rothschild Redburn upgrades MoonLake to Buy from Neutral. Sonelokimab's FDA path for HS clears major hurdle; seen as best-in-class contender attractive to big pharma.

Bernstein initiates Underperform on Novo Nordisk, citing endless earnings downgrades, weak catalysts, U.S. price deflation, and 2032 patent cliff risks.
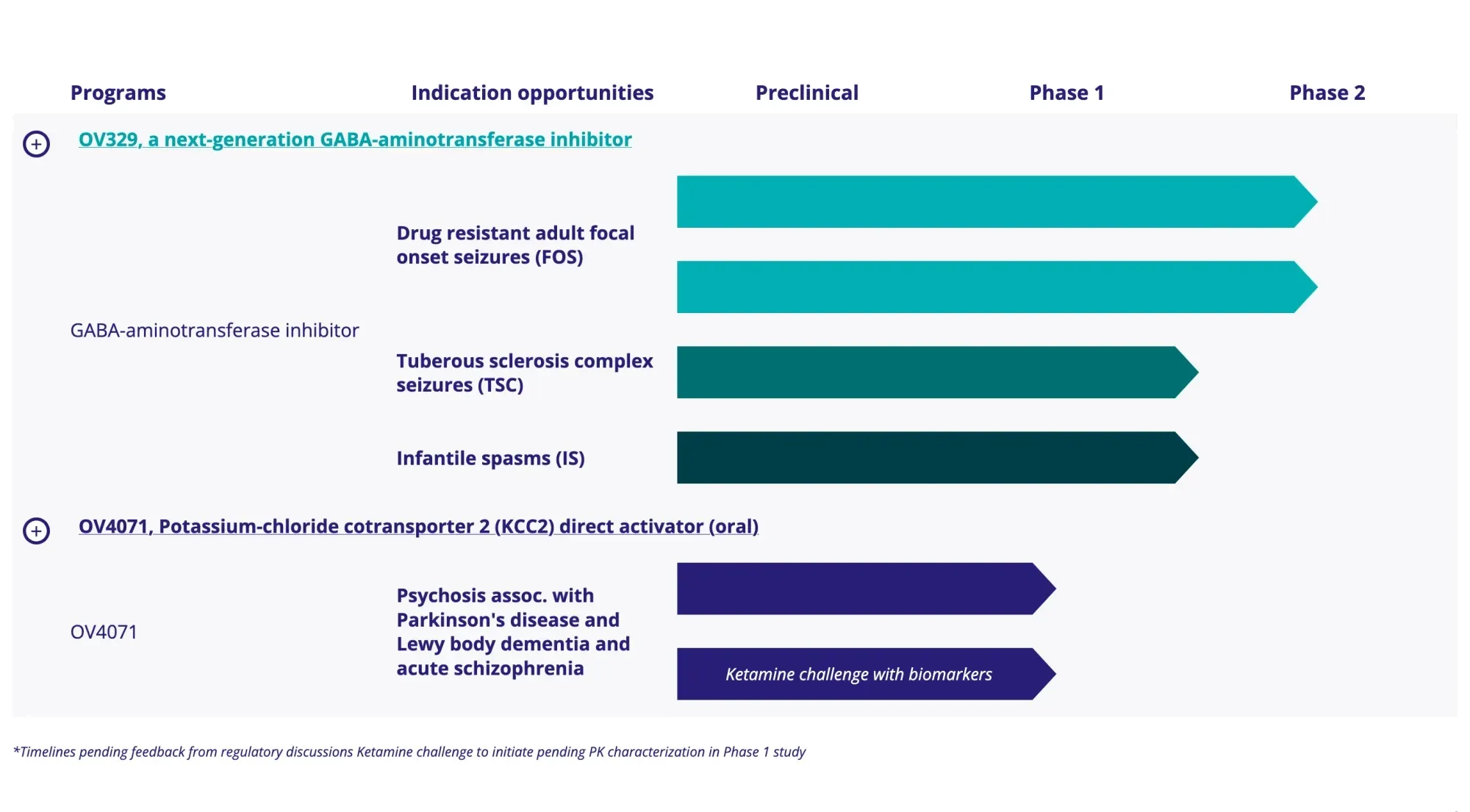
Ovid Therapeutics shares climbed after swinging to a surprise Q4 profit beat and raising $60 million via PIPE financing. Funds will expand OV329 development into tuberous sclerosis complex and infantile spasms.

TD Cowen upgrades Grail to Buy from Hold with $65 target after 60% selloff. Analyst sees attractive entry in multi-cancer detection despite NHS study miss.

B. Riley starts coverage on Arcturus Therapeutics with Buy, praising its standout lipid-enabled mRNA platform for cystic fibrosis (ARCT-032) and OTC deficiency (ARCT-810) programs.

Myriad Genetics' MyChoice CDx gains FDA approval as the only CDx to identify HRD-positive patients for Zejula maintenance in advanced ovarian cancer, boosting precision therapy.
China’s NMPA has granted priority review to Gyre Therapeutics’ NDA for Hydronidone (F351) for chronic hepatitis B liver fibrosis.
JPMorgan boosts Edgewise Therapeutics price target to $45 from $34, maintains Overweight rating. Bullish on EDG-7500 catalysts and strong 2026 setup.
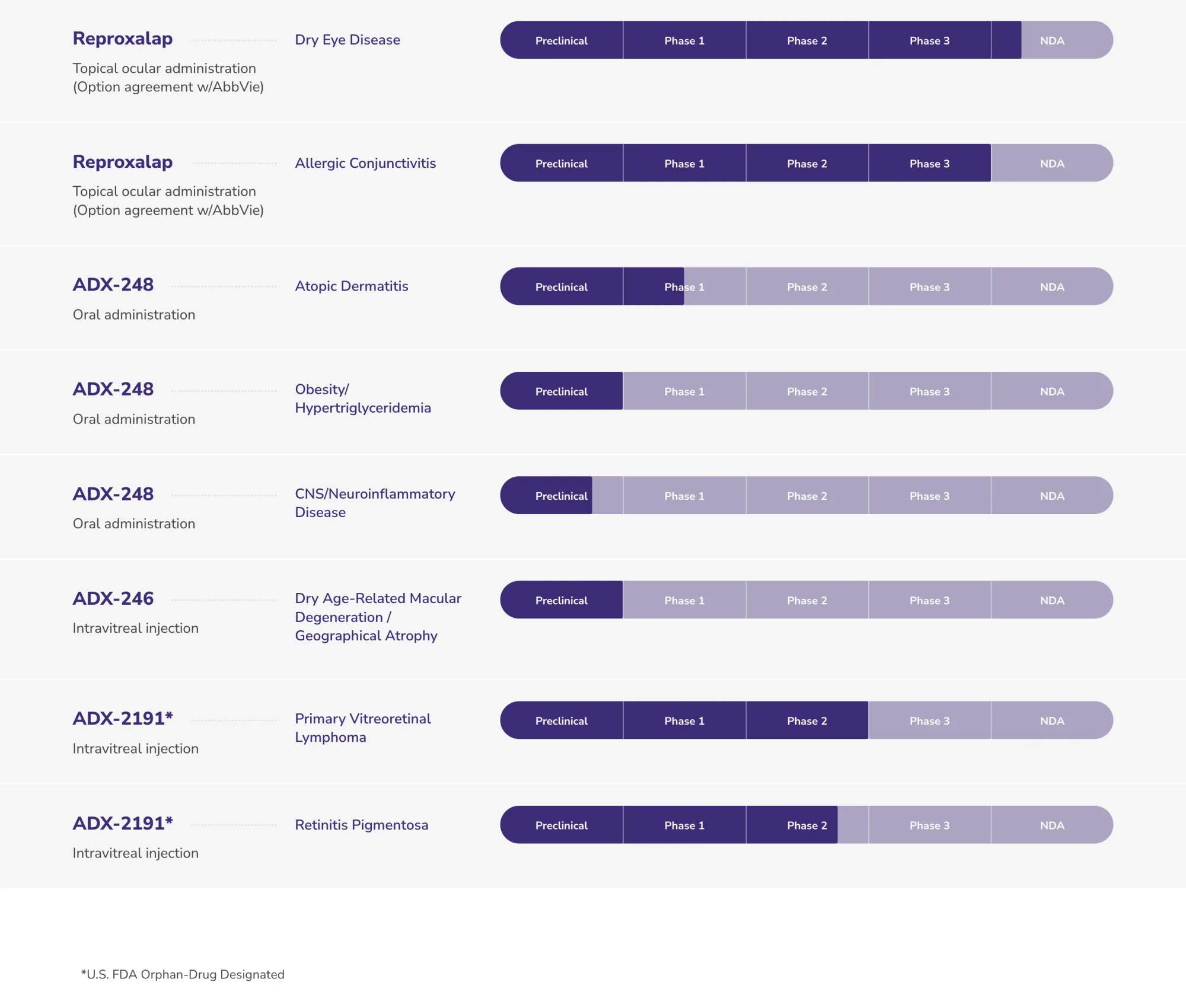
Aldeyra Therapeutics receives FDA Complete Response Letter rejecting reproxalap for dry eye disease due to insufficient efficacy. No safety issues. ALDX shares crash.

Cantor Fitzgerald initiates Overweight on Tenax Therapeutics with $35 target. Under-the-radar biotech nears Q3 2026 Phase 3 readout for potential first PH-HFpEF therapy — high-risk, high-reward setup.

LENSAR and Alcon have mutually terminated their merger after the Federal Trade Commission threatened to block the deal.

Rhythm Pharmaceuticals announced that setmelanotide missed its primary endpoints in the Phase 3 EMANATE trial. Post-hoc analyses showed statistically significant BMI reductions in POMC/PCSK1 and SRC1 patients.

HSBC downgraded Eli Lilly to Reduce from Hold, cutting its price target to $850 from $1,070. The analyst cites an overhyped $150B+ obesity market, rising price competition, and expected cuts in 2026.
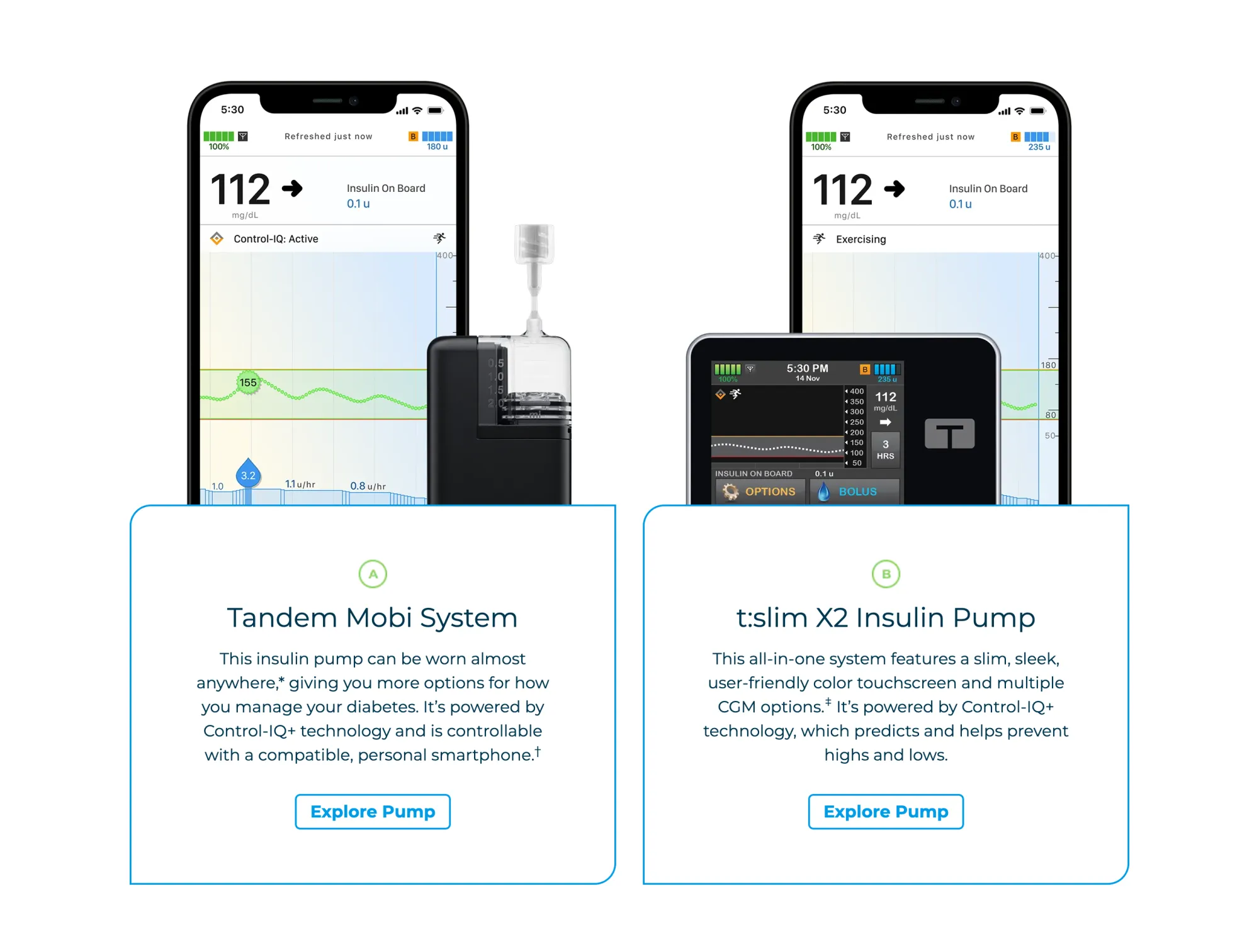
Piper Sandler upgrades Tandem Diabetes to Overweight from Neutral, raises PT to $33 from $21. Mobi pump, pharmacy pricing, and international growth drive revenue upside.
BioMarin is discontinuing Phase 2 Voxzogo trials for Turner Syndrome, SHOX-deficiency and ACAN-deficiency after SCFE events in investigator-sponsored studies. Noonan syndrome and most ISS trials continue as planned.
Jefferies initiates Buy on Design Therapeutics ($15 PT) & assumes coverage on Revolution Medicines ($140), Kymera ($110), Immatics ($18), Insmed ($228).
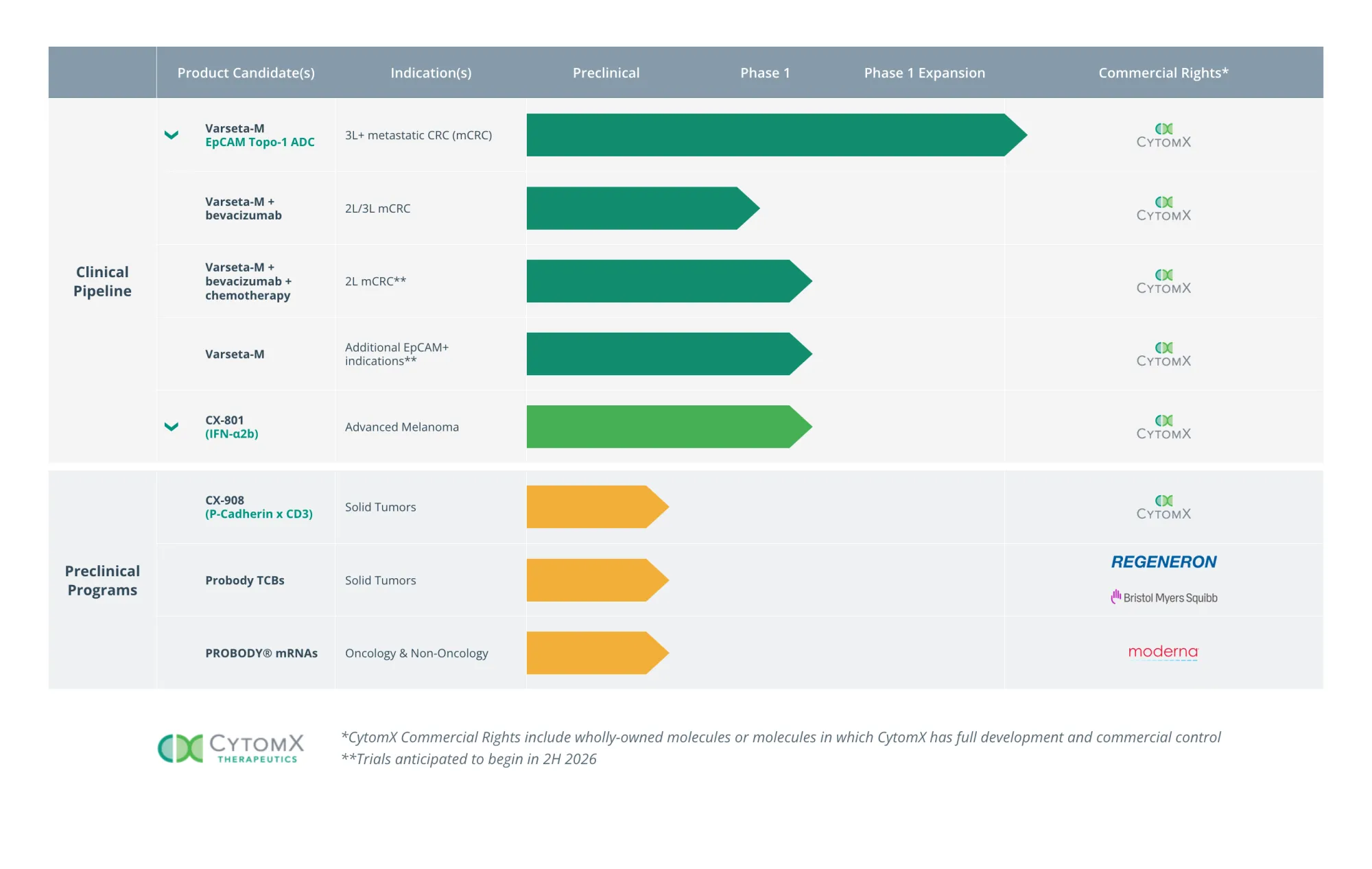
Shares of CytomX Therapeutics surged after strong Phase 1 trial data for Varsetatug Masetecan showed tumor shrinkage in late-stage colorectal cancer patients with mostly mild side effects.

WW International reported Q4 and full-year 2025 results beating guidance, with strong growth in clinical subscribers thanks to GLP-1 integration.
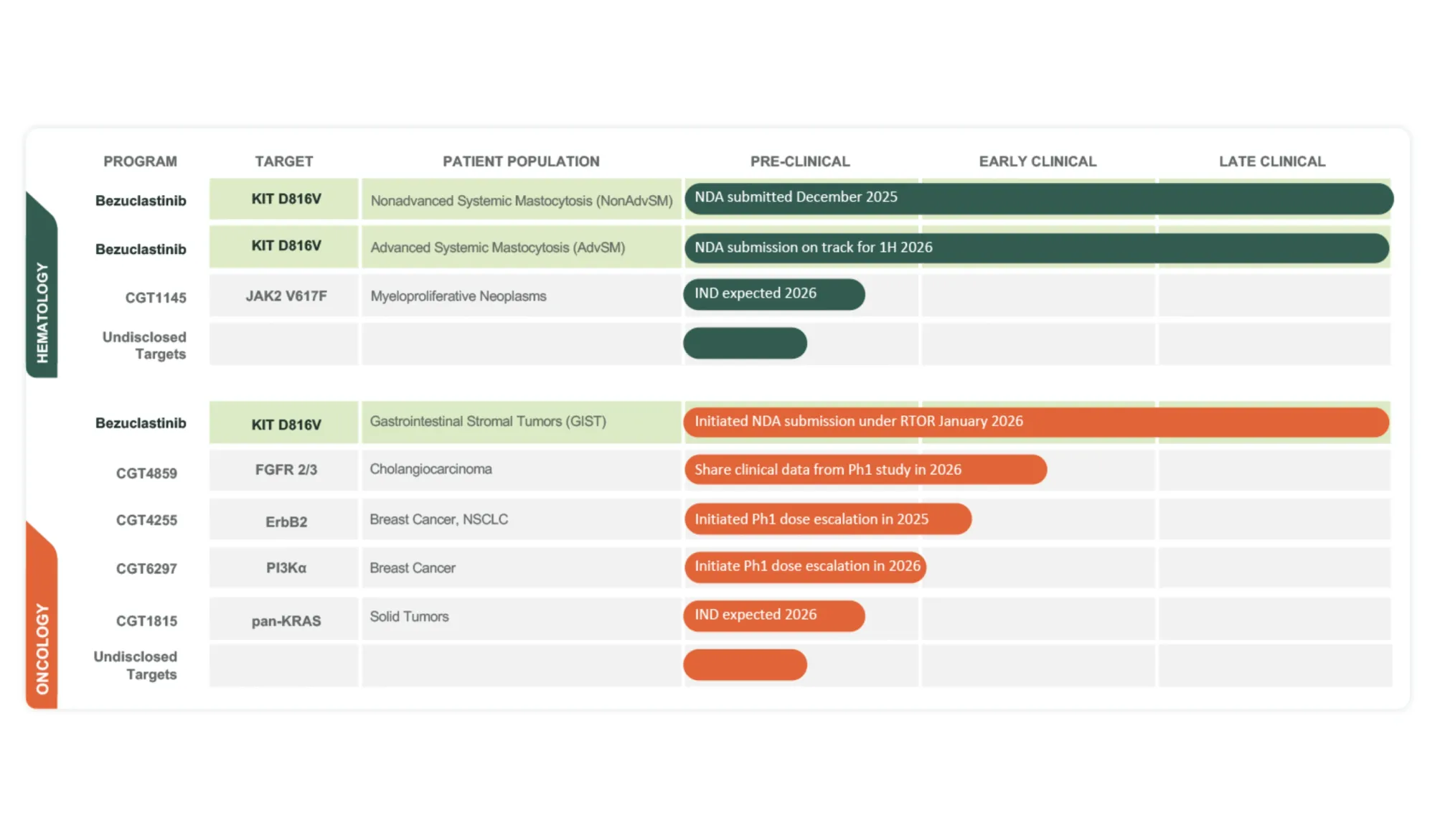
Jefferies analyst started coverage on Cogent Biosciences with a Buy rating and $55 price target after the FDA accepted the NDA for bezuclastinib in Non-Advanced Systemic Mastocytosis. PDUFA date set for December 30, 2026.

Structure Therapeutics' oral pill aleniglipron achieved 15–16% body weight loss (up to 39 lbs) after 44 weeks in Phase 2, rivaling injectables with better tolerability and no plateau. Phase 3 is planned for 2026—a potential game changer for obesity treatment.
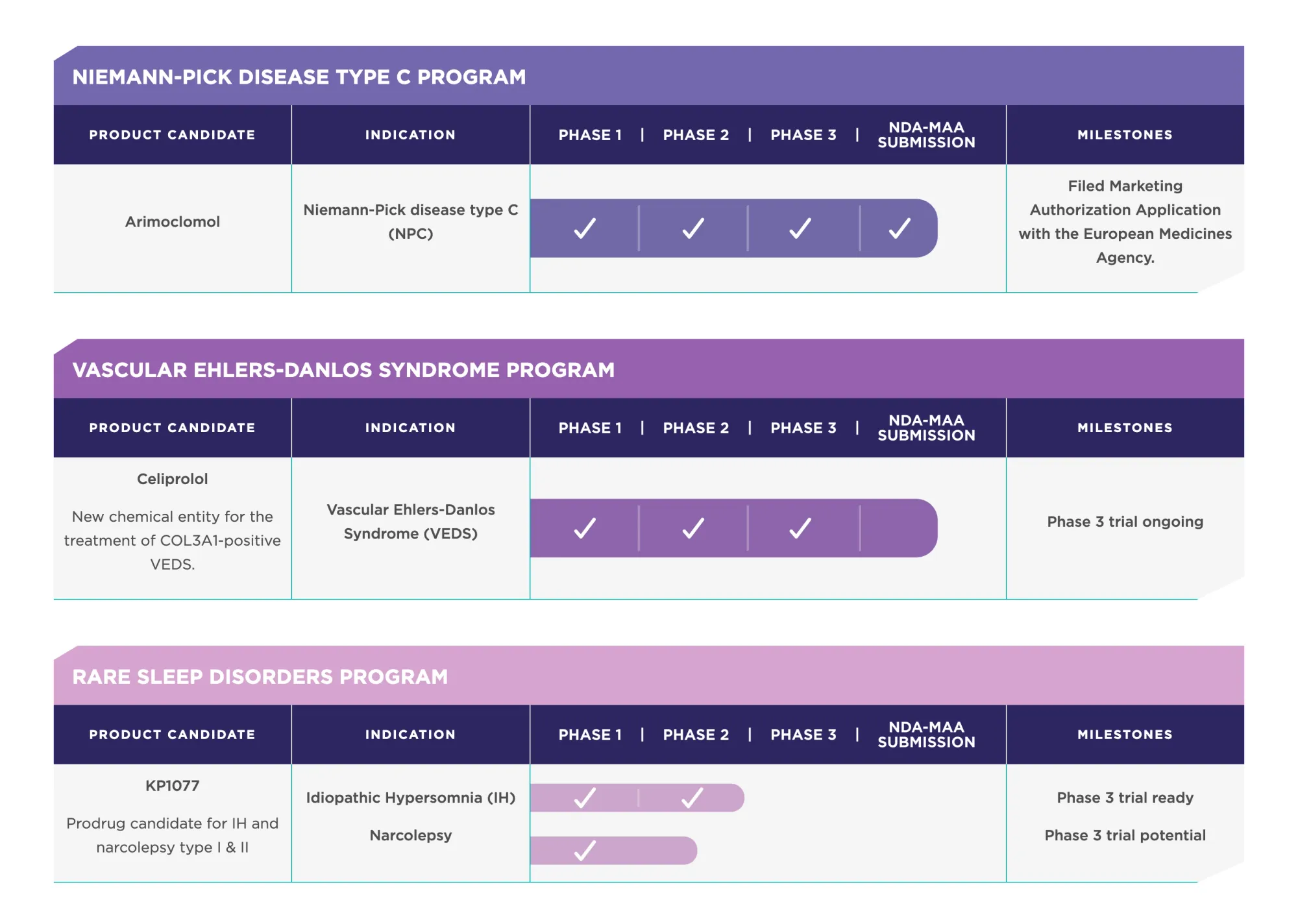
BTIG starts coverage on ZVRA with Buy rating and $23 target after Zevra sells SDX portfolio for $50 million and becomes debt-free.
Jefferies starts Abivax coverage with Buy rating and $160 target. Views obefazimod as differentiated UC therapy; Q2 maintenance data de-risked for approval path.
ImmunityBio just hit a huge milestone – they can now mass-produce and freeze powerful NK cells from healthy and cancer donors. One simple blood draw gives 8–10 ready-to-use doses.
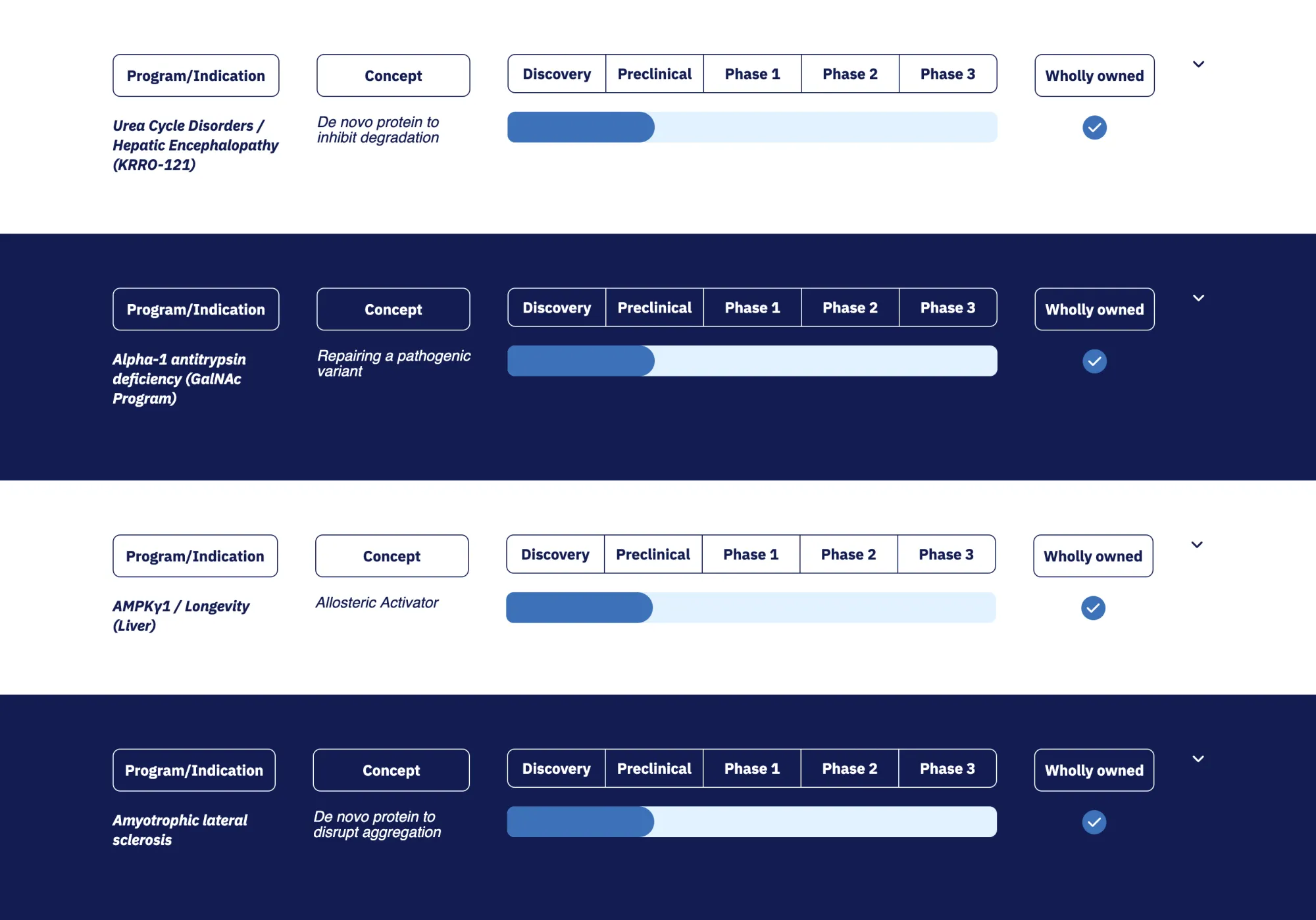
Raymond James upgrades Korro Bio to Outperform from Market Perform with $23 PT, bullish on pivot to hyperammonemia programs in UCDs & HE with strong preclinical data and high unmet need.

Immutep’s key Phase 3 trial for its cancer drug efti was stopped early due to poor results. Analysts from Citizens and Baird downgraded the stock.

Insulet has launched a voluntary medical device correction for specific lots of Omnipod 5 Pods due to possible tubing issues. All other pods remain safe.

Natera launches Zenith Genomics — its next-generation whole-genome sequencing test for faster rare disease diagnosis. GeneDx shares drop 8% as competition intensifies in the rare-disease market, which carries a $1 trillion economic burden.

French biotech company Abivax strongly rejects media claims that it gave AstraZeneca secret access to its data for a possible buyout until March 23, calling the reports "unfounded rumors."

Oppenheimer starts ProQR Therapeutics (PRQR) coverage with Outperform rating and $9 price target, citing RNA editing potential in liver diseases ahead of 2026 data.
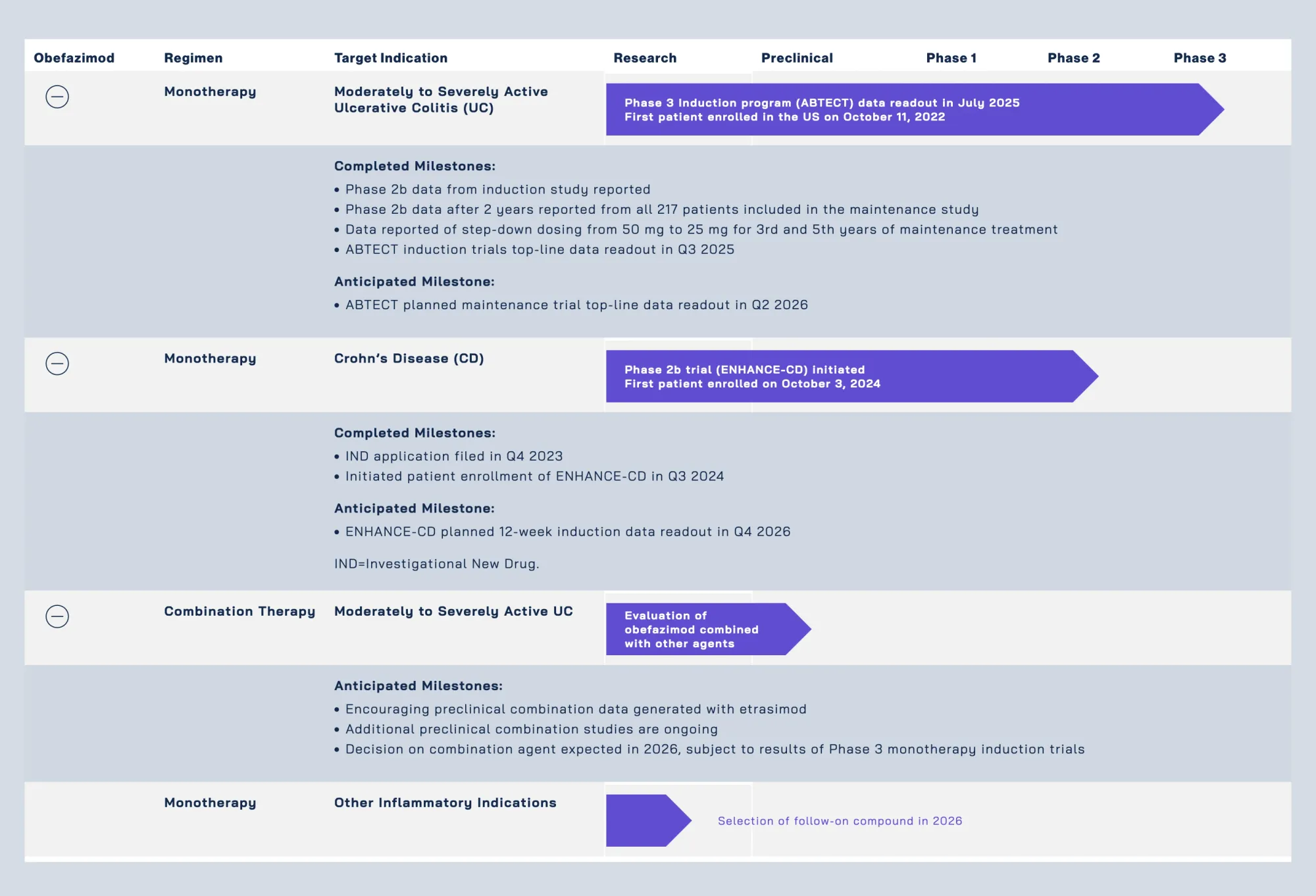
AstraZeneca gains exclusive data access to acquire surging French biotech Abivax (up 1,600% in 2025) before March 23 deadline; Eli Lilly waits in wings amid IBD drug promise.

Solid Biosciences’ gene therapy SGT-003 shows early promise for Duchenne; trial patients improved muscle and heart function, with potential accelerated FDA approval in 2026.
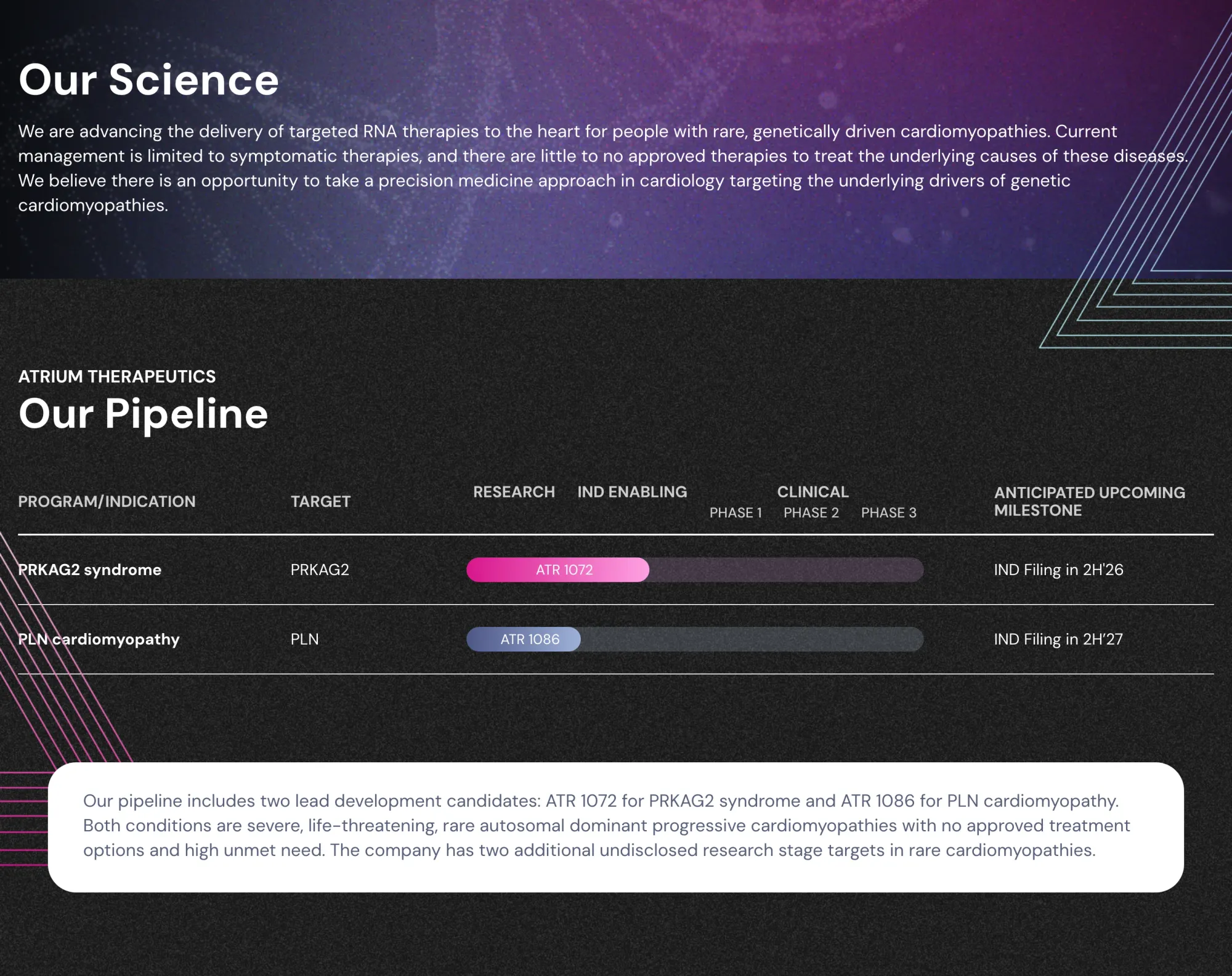
Wells Fargo analyst initiates coverage on Atrium Therapeutics with Overweight rating and $25 price target, highlighting big potential in RNA therapies for rare genetic heart diseases with no current treatments.

Kala Bio shares surged 63% premarket to 48 cents after announcing the launch of its first purpose-built AI agent for biotech in just 14 days through the rebranded Researgency.ai platform.

Oppenheimer starts coverage on Ocugen with an Outperform rating and $10 price target, highlighting its lead gene therapy OCU400 in Phase 3 for retinitis pigmentosa and big market potential by 2027.
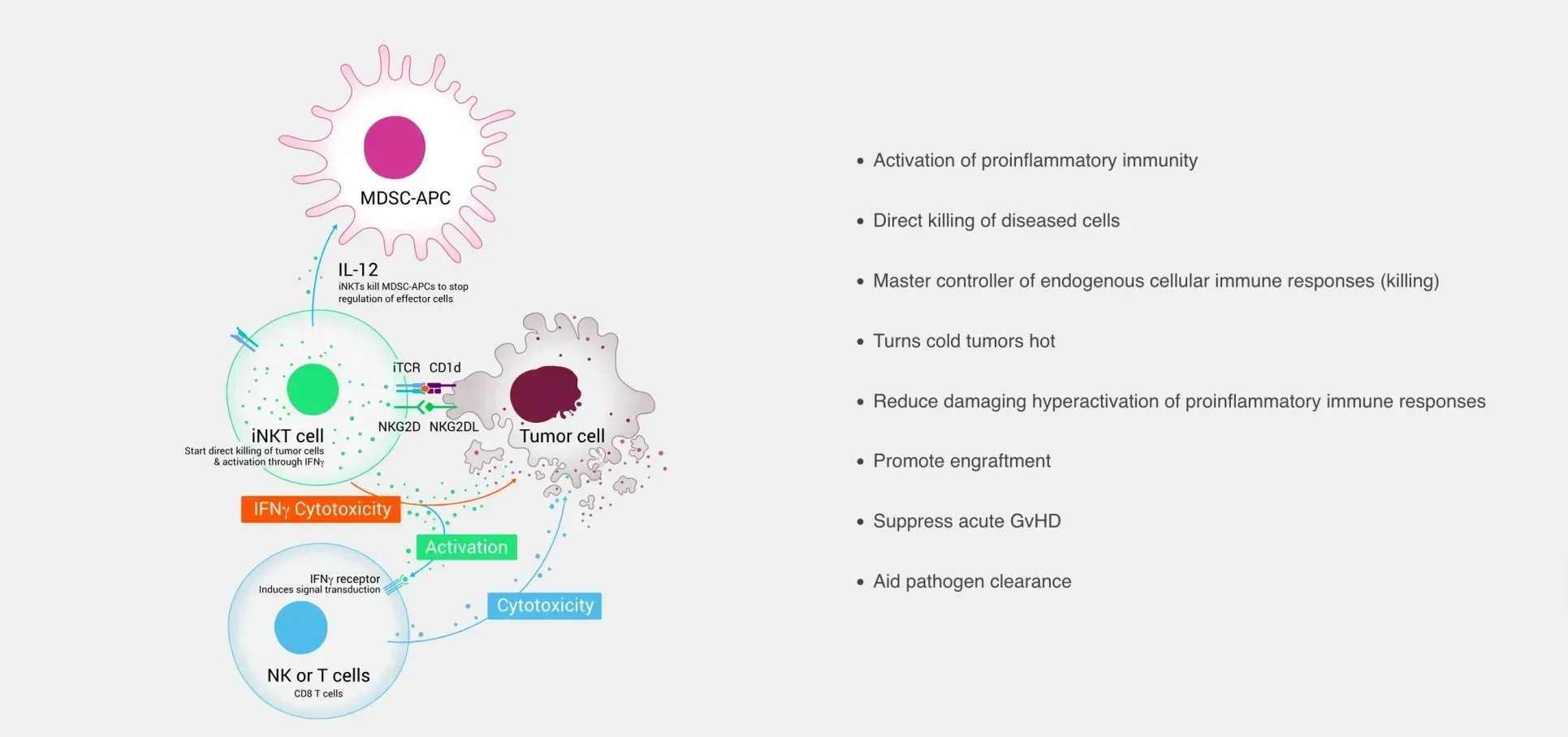
MiNK Therapeutics announces a new collaboration with C-Further to advance PRAME-targeted iNKT cell therapy for pediatric cancers, including up to $1.1 million in funding.

Morgan Stanley downgraded Grifols stock to Equal Weight with a lower €11 price target, citing slowdowns in key products like albumin in China and alpha-1.

BofA reaffirms Buy on Vera Therapeutics; Vertex’s povetacicept Phase 3 interim results in IgAN meet expectations but pose no major competitive threat.

Novo Nordisk receives FDA warning letter after 2025 Plainsboro inspection revealed failures in timely reporting serious side effects for drugs like Ozempic and Victoza. Company says no impact on production.
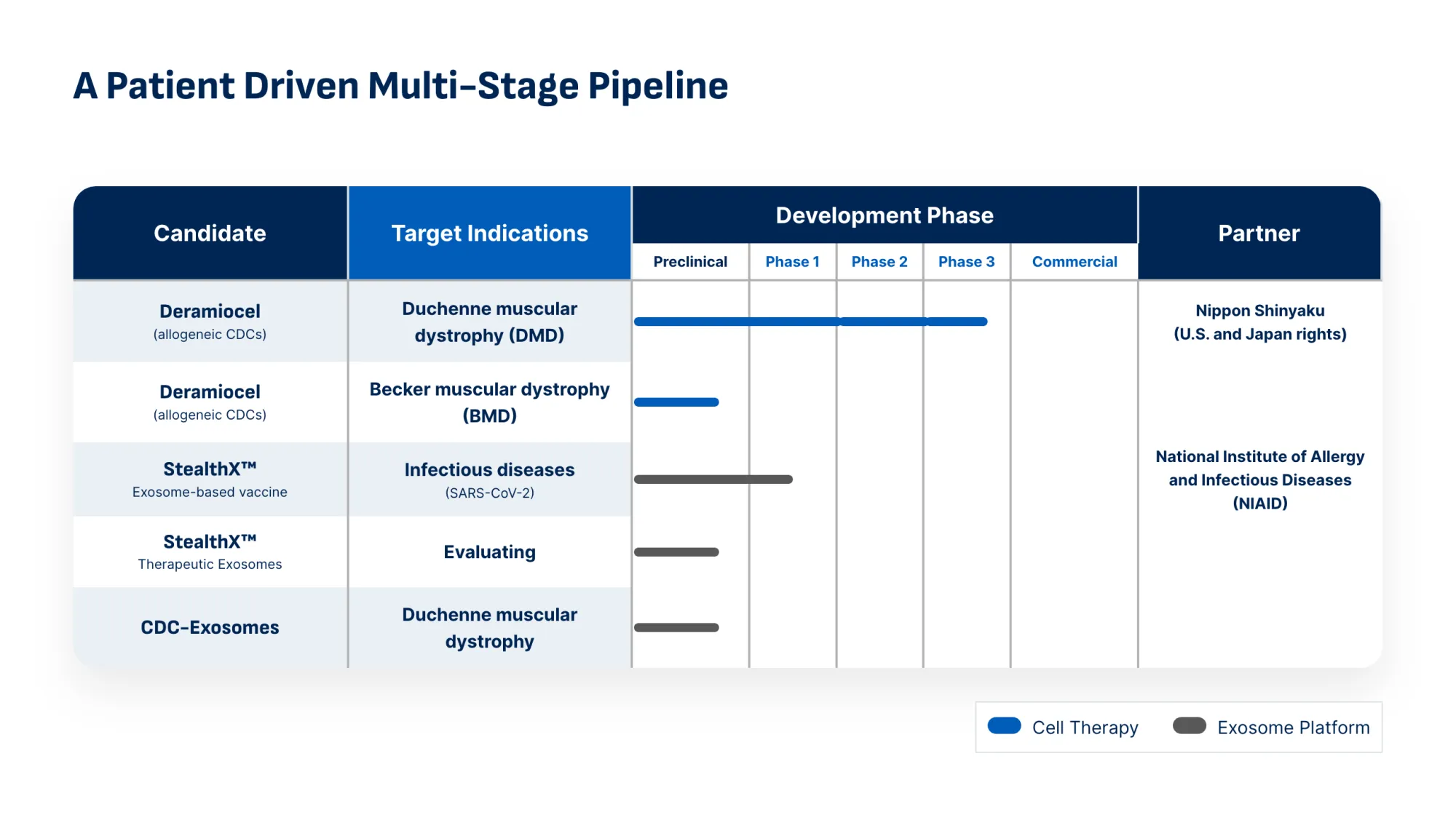
Capricor Therapeutics receives new PDUFA target date of August 22, 2026 for Deramiocel BLA. Potential first therapy to treat both heart and skeletal muscle damage in Duchenne muscular dystrophy (DMD).

BioNTech co-founders Ugur Sahin & Özlem Türeci are starting an independent next-gen mRNA company by end-2026. BioNTech keeps focus on oncology (15 Phase 3 trials).

JPMorgan & William Blair raise BridgeBio price targets to $93–$94 on strong Attruby launch and achondroplasia data.

Deutsche Bank upgrades Teladoc Health to Buy from Hold with $11 price target, calling valuation compelling amid BetterHelp stabilization and potential exit upside.

Mineralys receives FDA acceptance of NDA for lorundrostat for hypertension. PDUFA target date: December 22, 2026. Explore-OSA missed primary endpoint but demonstrated meaningful BP reduction.

Vertex Pharmaceuticals' povetacicept succeeds in Phase 3 RAINIER trial for IgA nephropathy with 52% proteinuria reduction. Positive safety data and FDA submission planned soon.

Trevi Therapeutics shares plunged despite positive FDA alignment for Haduvio in IPF chronic cough. Plans for two Phase 3 trials (instead of one) triggered sell-off; Stifel holds Buy rating on the de-risked program.

Relmada shares jumped sharply after positive 12-month Phase 2 trial data for NDV-01 in bladder cancer and announcement of $160M financing to advance development.

H.C. Wainwright calls Roche’s persevERA trial disappointing but says it’s not a full readthrough for Olema Oncology. Different CDK4/6 inhibitor (ribociclib) may favor palazestrant in OPERA-02.
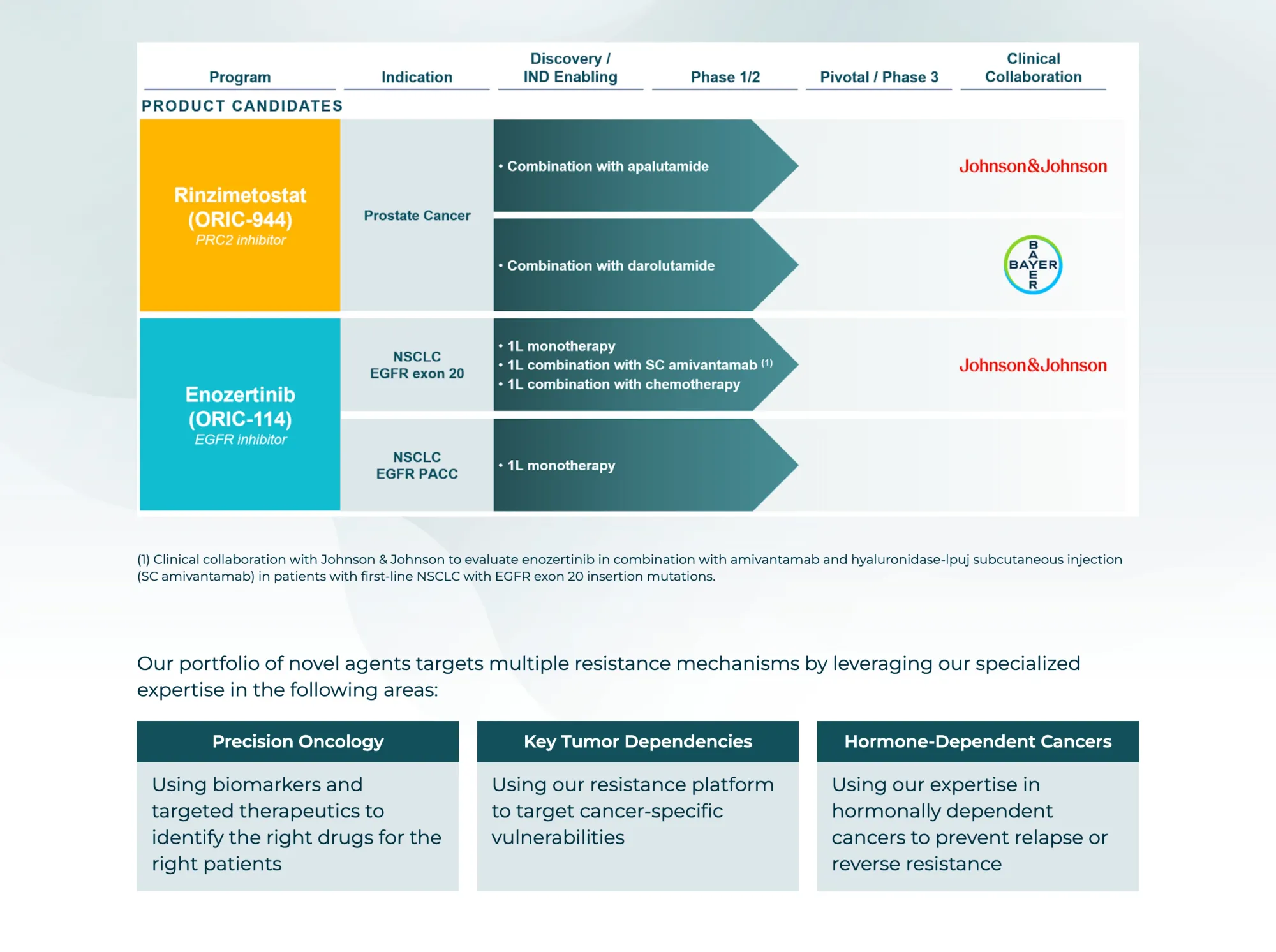
Oric Pharmaceuticals shares fell after Ipsen pulled Tazverik from all markets due to safety concerns in a key trial. Tazverik sales were already weak—a blow to confidence in similar EZH2 drugs like ORIC’s, even if they are not the same.