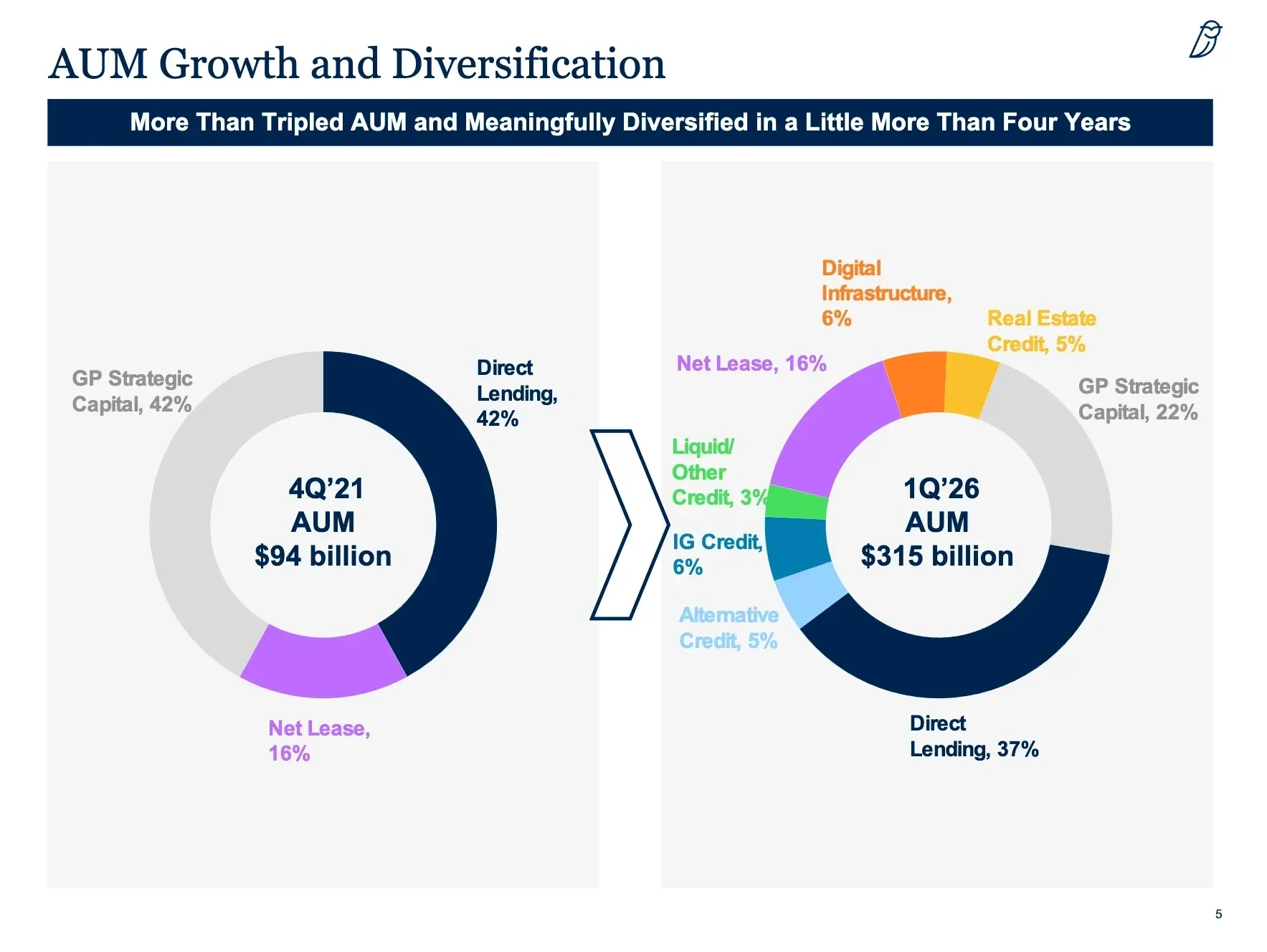
FDA Fast Track Boosts Context Therapeutics’ CTIM-76 for Hard-to-Treat Ovarian Cancer
Context Therapeutics’ CTIM-76 wins FDA Fast Track Designation for platinum-resistant ovarian cancer. Phase 1a trial ongoing with interim data expected in June 2026.
Already have an account? Sign in.