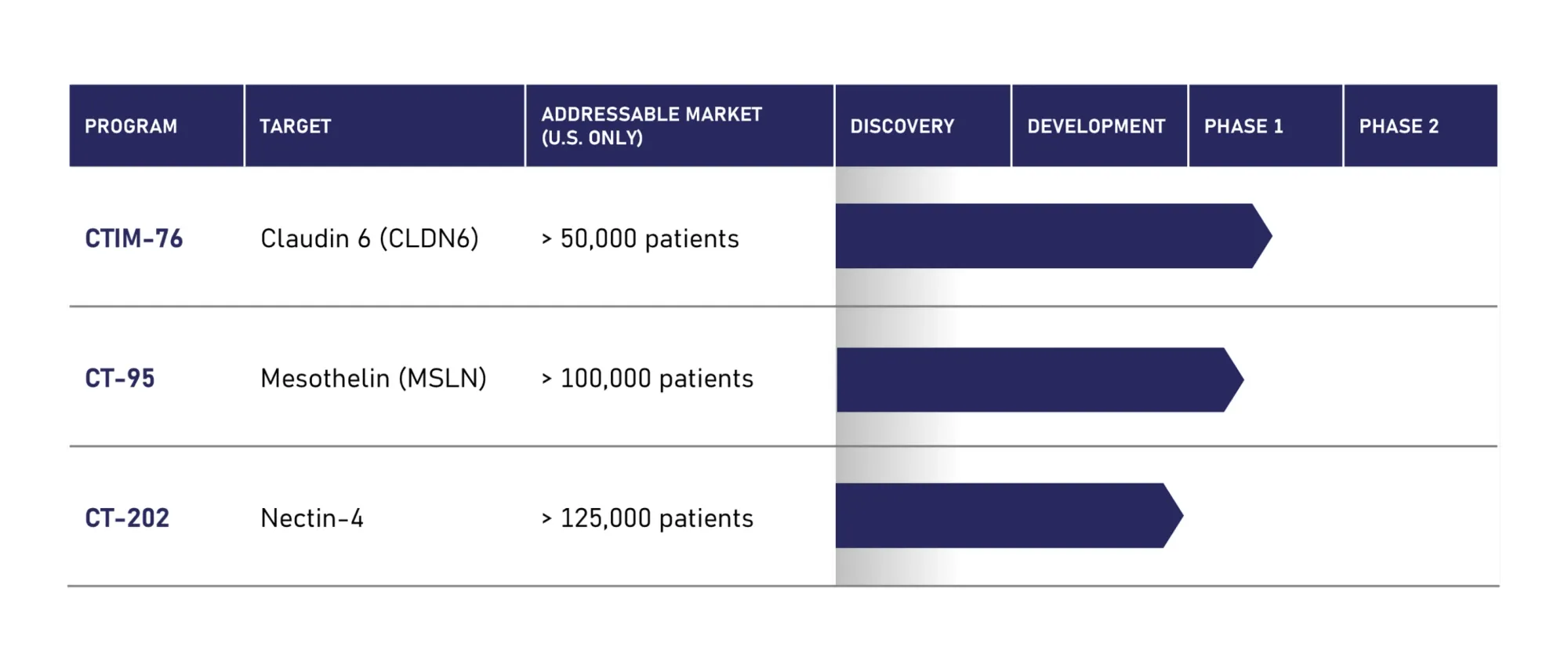
FDA Fast Track Boosts Context Therapeutics’ CTIM-76 for Hard-to-Treat Ovarian Cancer
Context Therapeutics’ CTIM-76 wins FDA Fast Track Designation for platinum-resistant ovarian cancer. Phase 1a trial ongoing with interim data expected in June 2026.

Context Therapeutics’ CTIM-76 wins FDA Fast Track Designation for platinum-resistant ovarian cancer. Phase 1a trial ongoing with interim data expected in June 2026.
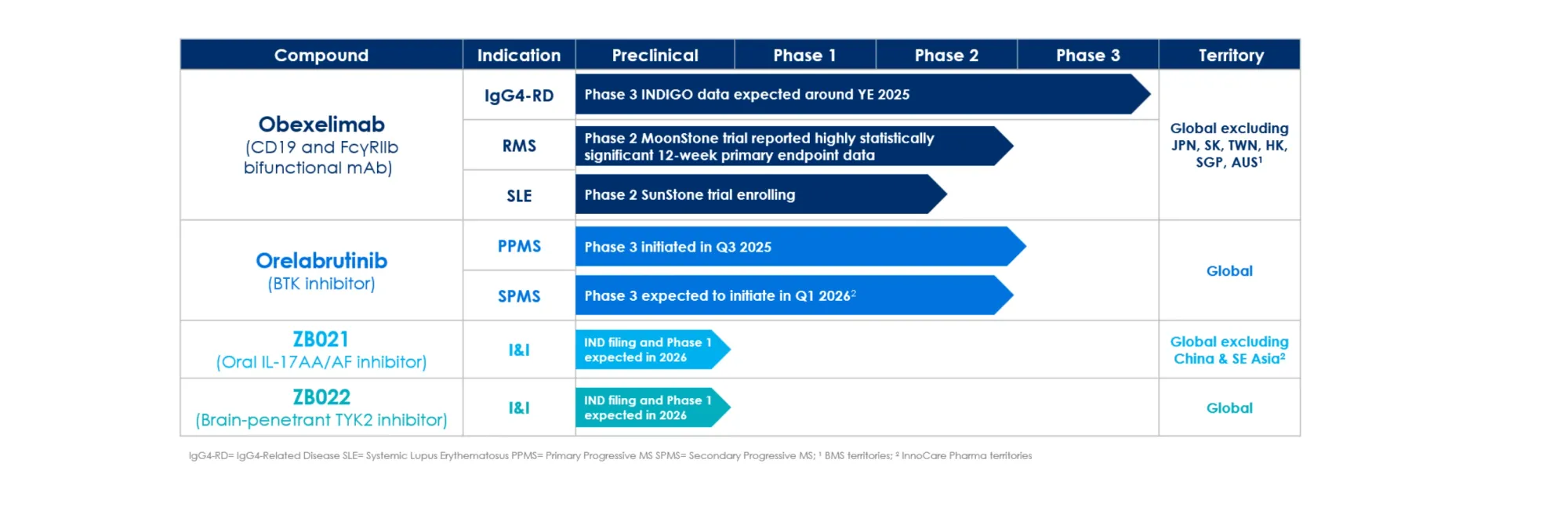
Citi, H.C. Wainwright, JonesResearch, and LifeSci Capital share new ratings and price targets on Zenas BioPharma, Spruce Biosciences, Context Therapeutics, and Ovid Therapeutics.
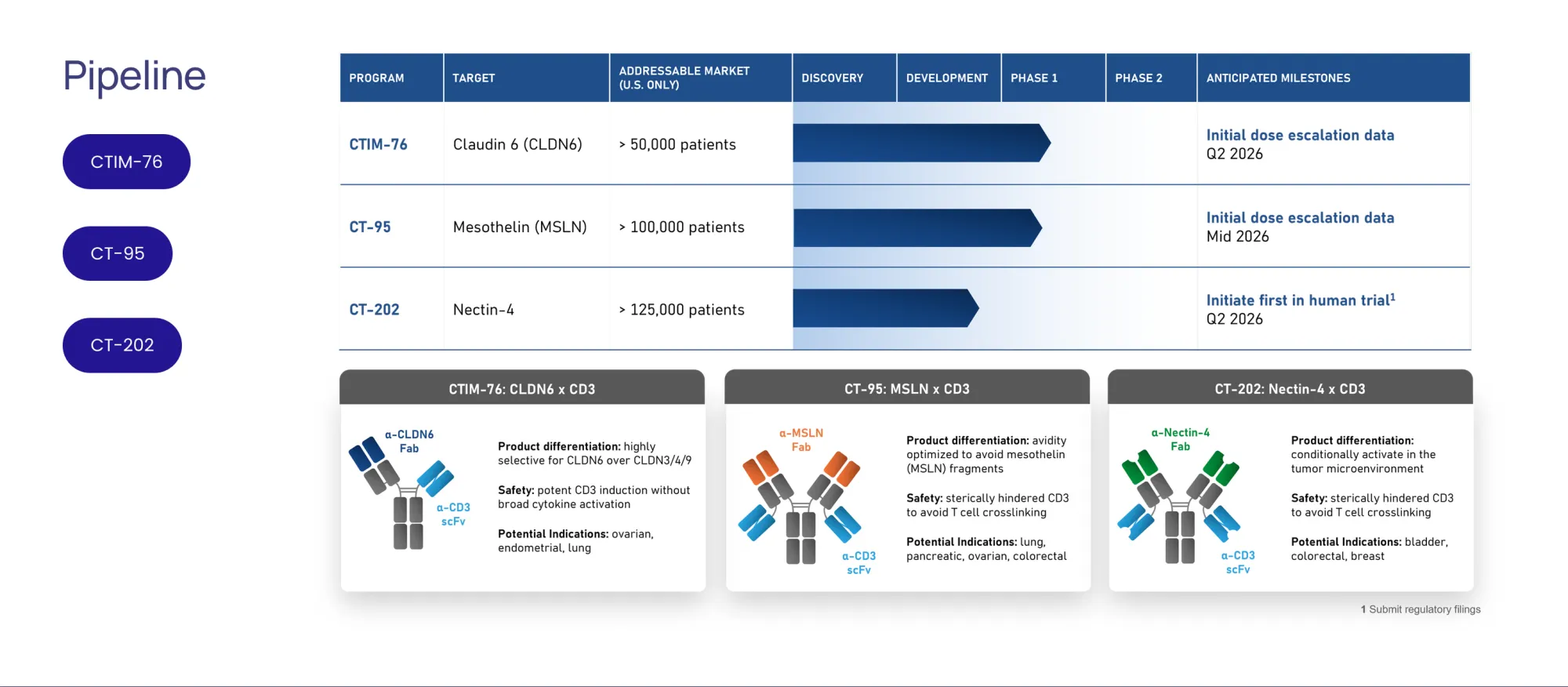
A hidden cancer company, Context, got a top rating. Its ovarian cancer drug, CTIM-76, has key data coming in 2026.