
FDA Lifts Hold on Intellia’s Phase 3 Gene-Editing Trial for Deadly Heart Disease
Intellia Therapeutics gets FDA approval to resume its Phase 3 trial for nexiguran ziclumeran, a gene-editing treatment for ATTR-CM heart disease, boosting shares by 6%.

Intellia Therapeutics gets FDA approval to resume its Phase 3 trial for nexiguran ziclumeran, a gene-editing treatment for ATTR-CM heart disease, boosting shares by 6%.

Intellia Therapeutics’ stock rises as the FDA clears a Phase 3 trial for its gene therapy, nexiguran ziclumeran, for a rare genetic disease.

Intellia Therapeutics (NTLA) shares fell 29% premarket after the FDA paused two late-stage gene-editing trials following a patient death linked to liver complications.
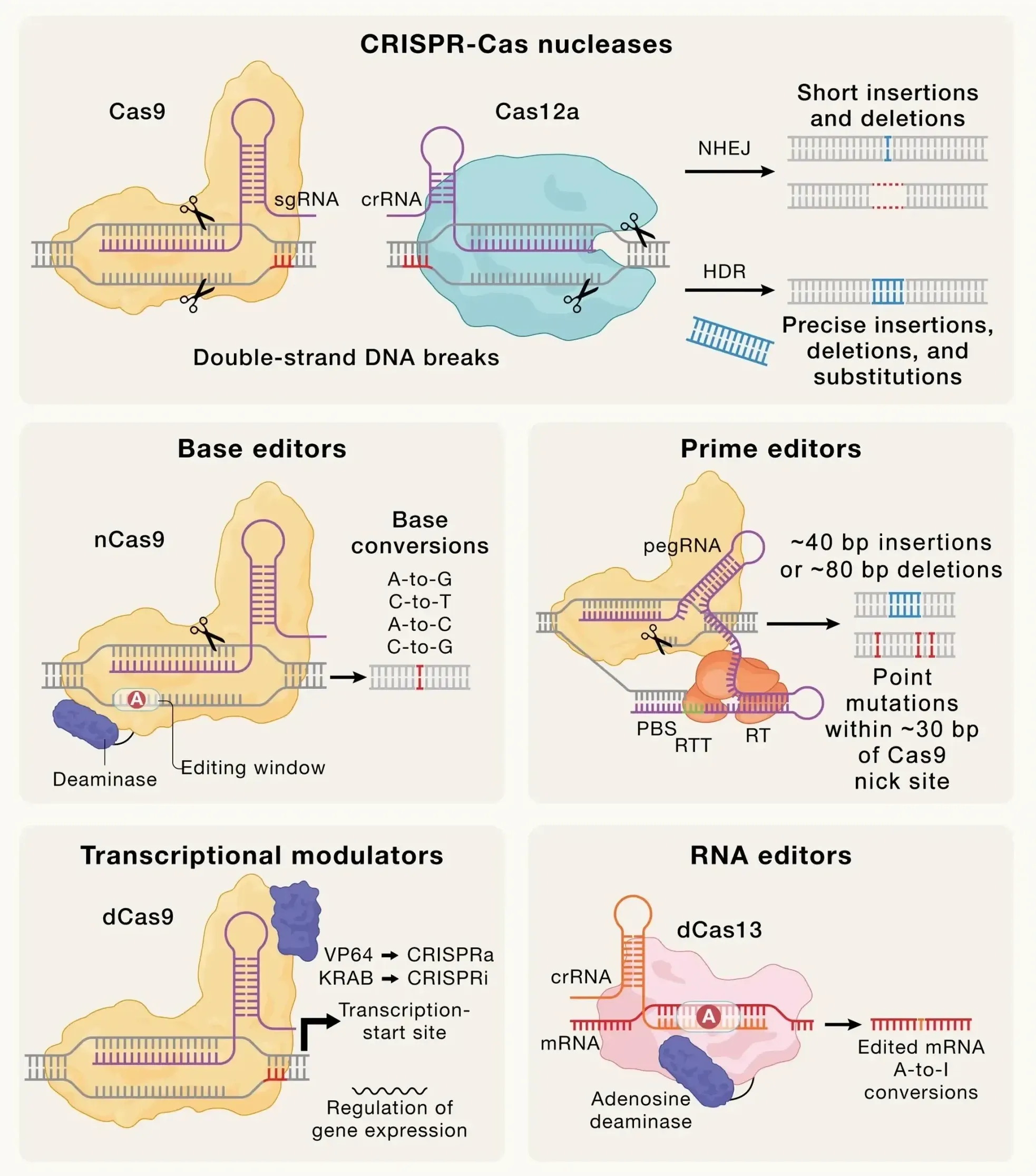
The FDA is unveiling a faster approval pathway for custom gene-editing therapies, aiming to spark major investment and accelerate cures for rare diseases through Crispr-based innovations.

Intellia Therapeutics (NTLA) shares drop 15% after FDA halts late-stage gene therapy trials following serious liver safety concerns in patient.

Intellia Therapeutics halts MAGNITUDE trials of nex-z gene therapy after patient hospitalization with Grade 4 liver toxicity. Over 650 patients enrolled in ATTR amyloidosis studies.