
Wall Street Backs Achieve Life Sciences: Cytisinicline Labeled 'Best-in-Class' Smoking Cessation Drug
Achieve Life Sciences gets Buy rating as its quit-smoking drug cytisinicline shows strong results; FDA decision due June 2026.

Achieve Life Sciences gets Buy rating as its quit-smoking drug cytisinicline shows strong results; FDA decision due June 2026.
Achieve Life Sciences jumps after CEO change, funding news, and progress on smoking/vaping quit drug cytisinicline.

Analysts say Pfizer listing Chantix on TrumpRx may support Achieve Life Sciences and boost confidence in cytisinicline’s approval prospects.

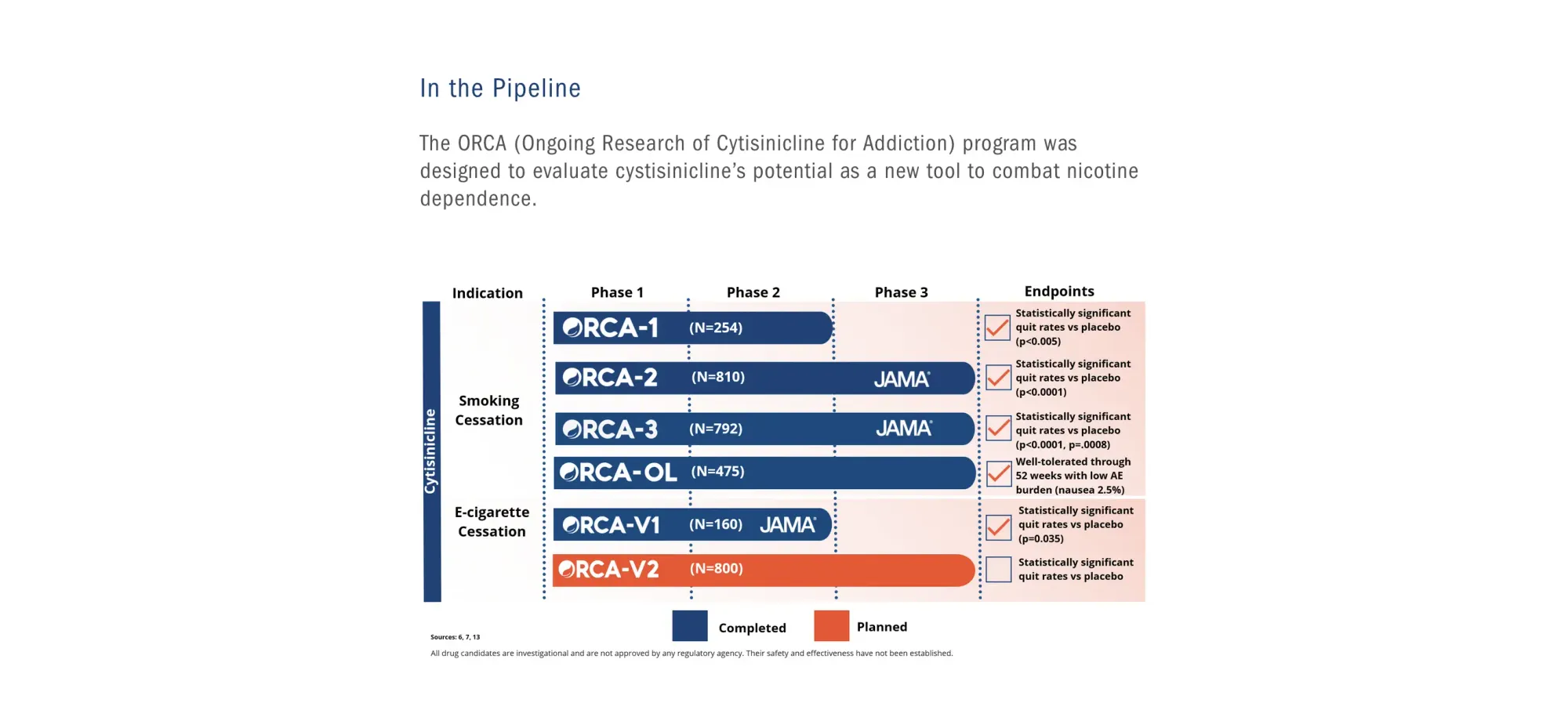
Achieve Life Sciences receives FDA's National Priority Voucher for cytisinicline, potentially the first approved treatment to help people quit vaping and e-cigarettes.

FDA accepted cytisinicline NDA for smoking cessation, with a PDUFA date of June 20, 2026. This marks a significant step, potentially offering the first new FDA-approved therapy in two decades to combat nicotine dependence, supported by strong clinical data and safety.