
Novavax Partners with Pfizer: $30M Deal for Vaccine Technology
Novavax secures $30M upfront payment from Pfizer in licensing deal for Matrix-M adjuvant technology, with potential for $500M more in milestone payments.


Novavax secures $30M upfront payment from Pfizer in licensing deal for Matrix-M adjuvant technology, with potential for $500M more in milestone payments.
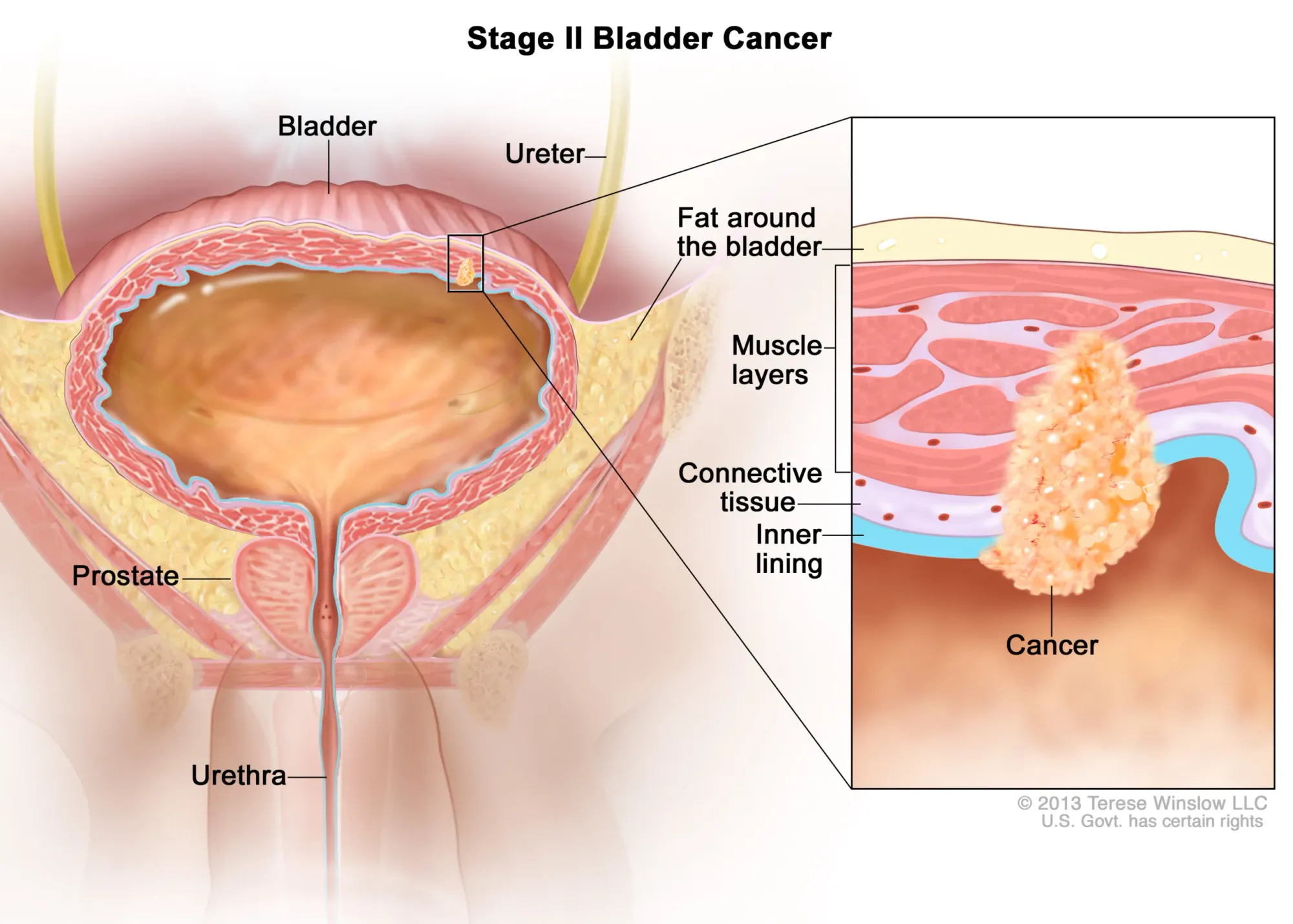
ImmunityBio advances FDA talks for ANKTIVA in papillary bladder cancer. Treatment shows 96% survival & 80% avoid bladder removal at 3 years, offering hope vs. current option: invasive surgery.

Chinese regulators dispatch 100+ investigators to PDD Holdings' Shanghai headquarters following employee clashes with officials. Investigation covers fraud allegations and tax issues, triggering stock decline.

3M surpasses Q4 2025 earnings estimates with $1.83 adjusted EPS as cost cuts and innovation drive growth, though 2026 guidance disappoints investors.
Abivax shares jump 6% on reports of AstraZeneca's €176-per-share offer for the French inflammatory disease drugmaker, adding to ongoing acquisition speculation.

Valneva voluntarily withdraws its IXCHIQ chikungunya vaccine from the U.S. following FDA suspension and a serious adverse event report, while maintaining licenses in Europe, Canada, UK, and Brazil.