
Merck and Eisai Phase 3 Miss Shakes Up Kidney Cancer Race – Analysts Split on Arcus
Merck’s new kidney cancer combos failed in Phase 3, missing key goals; analysts see mixed impact for Arcus and its drug.

Merck’s new kidney cancer combos failed in Phase 3, missing key goals; analysts see mixed impact for Arcus and its drug.

Ionis drug zilganersen showed promise for rare brain disease (Alexander disease), improving symptoms; FDA decision due Sept 2026.

BBOT receives U.S. FDA Fast Track Designation for BBO-11818, its pan-KRAS inhibitor for advanced KRAS-mutant pancreatic cancer. Phase 1 data expected H2 2026.
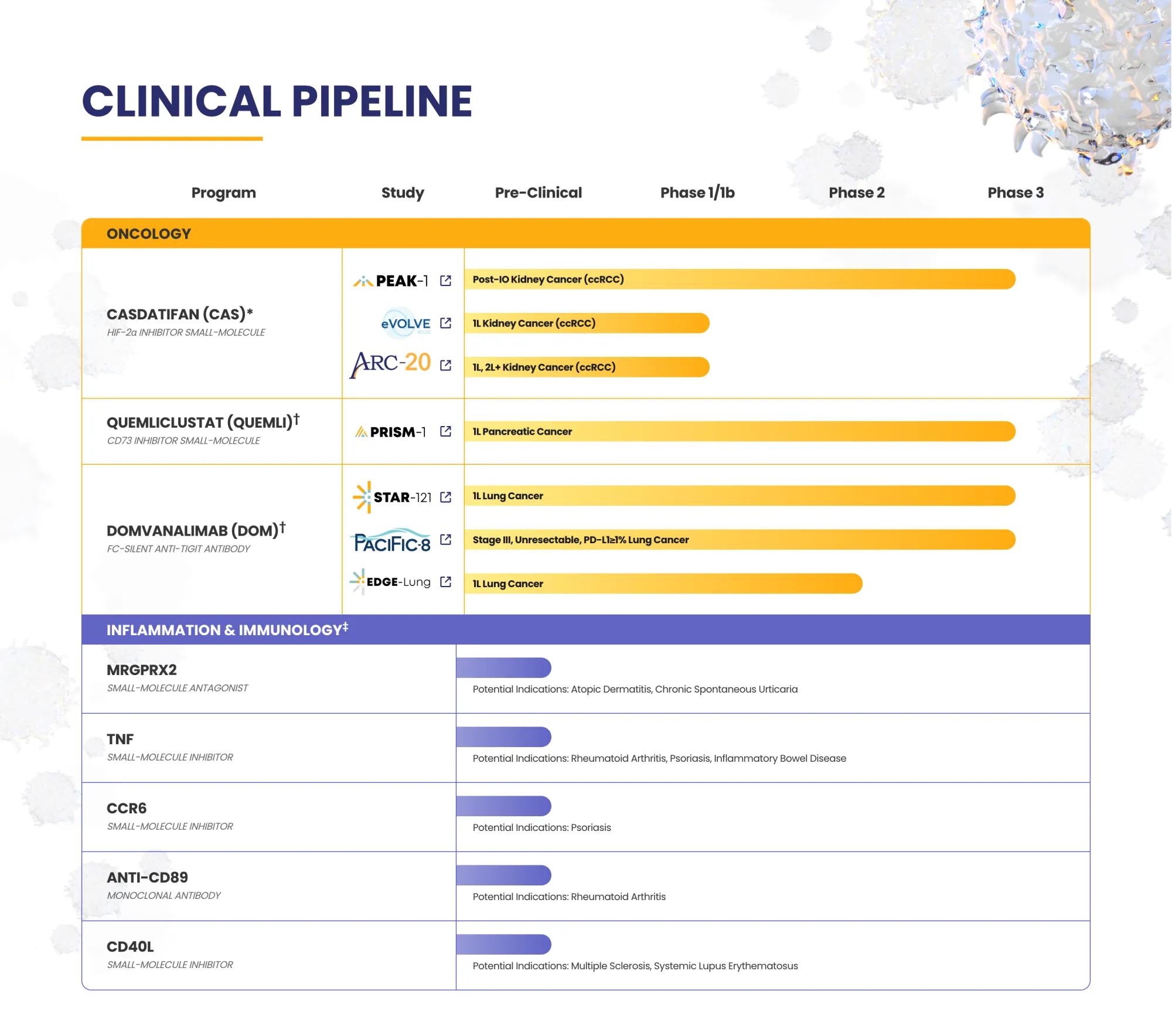
Arcus stops a lung cancer trial early after poor outlook, ends a partner study, and gains more control as Gilead scales back the deal.

Nektar drug showed promising hair regrowth in severe alopecia areata after 52 weeks, with good safety and twice-monthly shots.
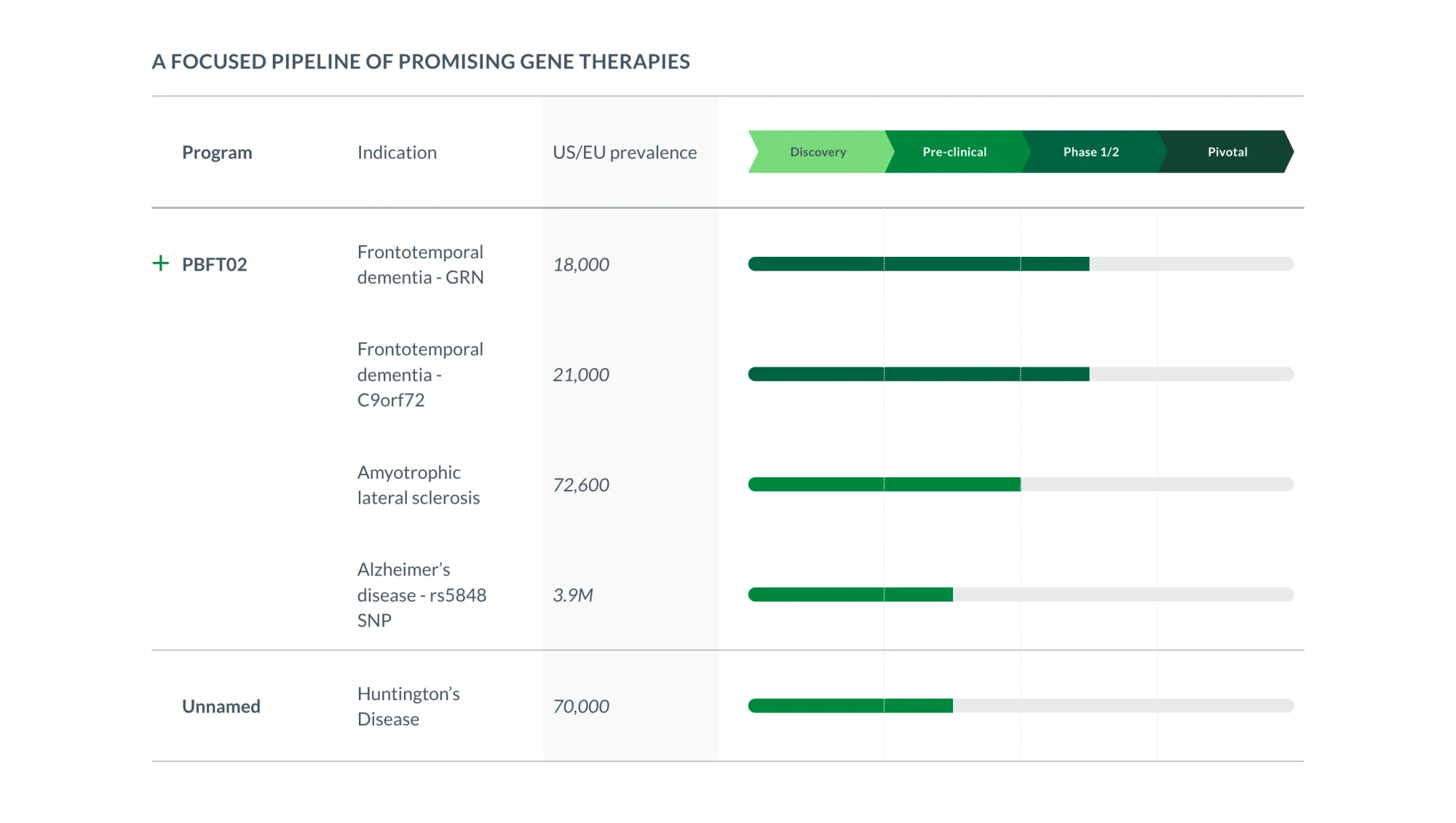
Passage Bio fell after the FDA demanded a larger trial; the drug shows promise for brain disease, but its future is now costlier and slower.