FDA Greenlights AUVELITY: First New Treatment for Alzheimer’s Agitation in Years
FDA approves AUVELITY to treat Alzheimer’s agitation, offering a safer, effective option to reduce distress and improve patient care.

FDA approves AUVELITY to treat Alzheimer’s agitation, offering a safer, effective option to reduce distress and improve patient care.

Chiesi will buy KalVista for $1.9B to get EKTERLY, a new oral rare disease drug, and grow its global rare disease business.

Pfizer extends Vyndamax patent to 2031, delaying generics and giving BridgeBio more time to grow its rival heart drug without pressure.
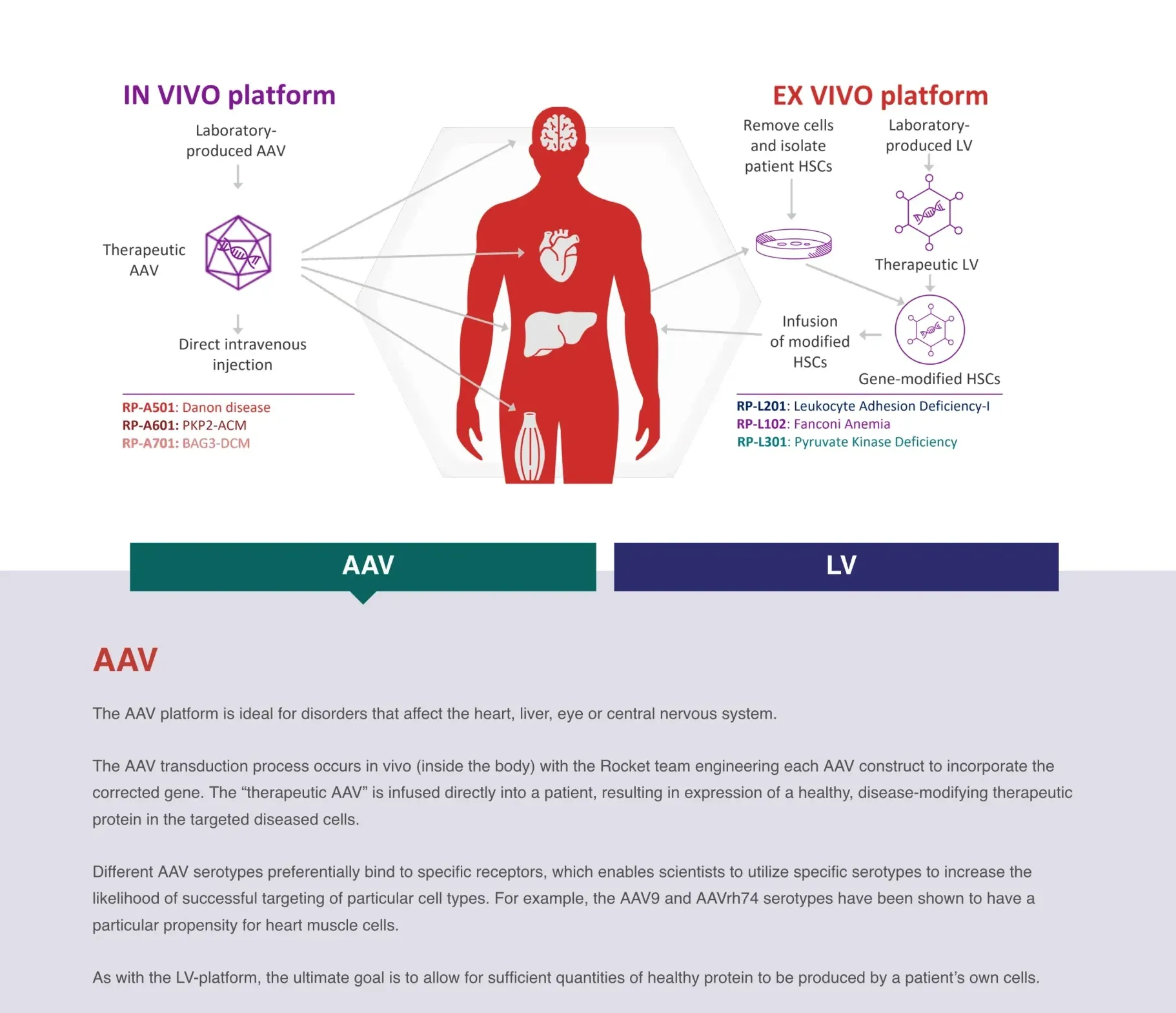
Rocket Pharma sold an FDA voucher for $180M cash, boosting funds without dilution and extending runway to 2028 for heart gene therapies.
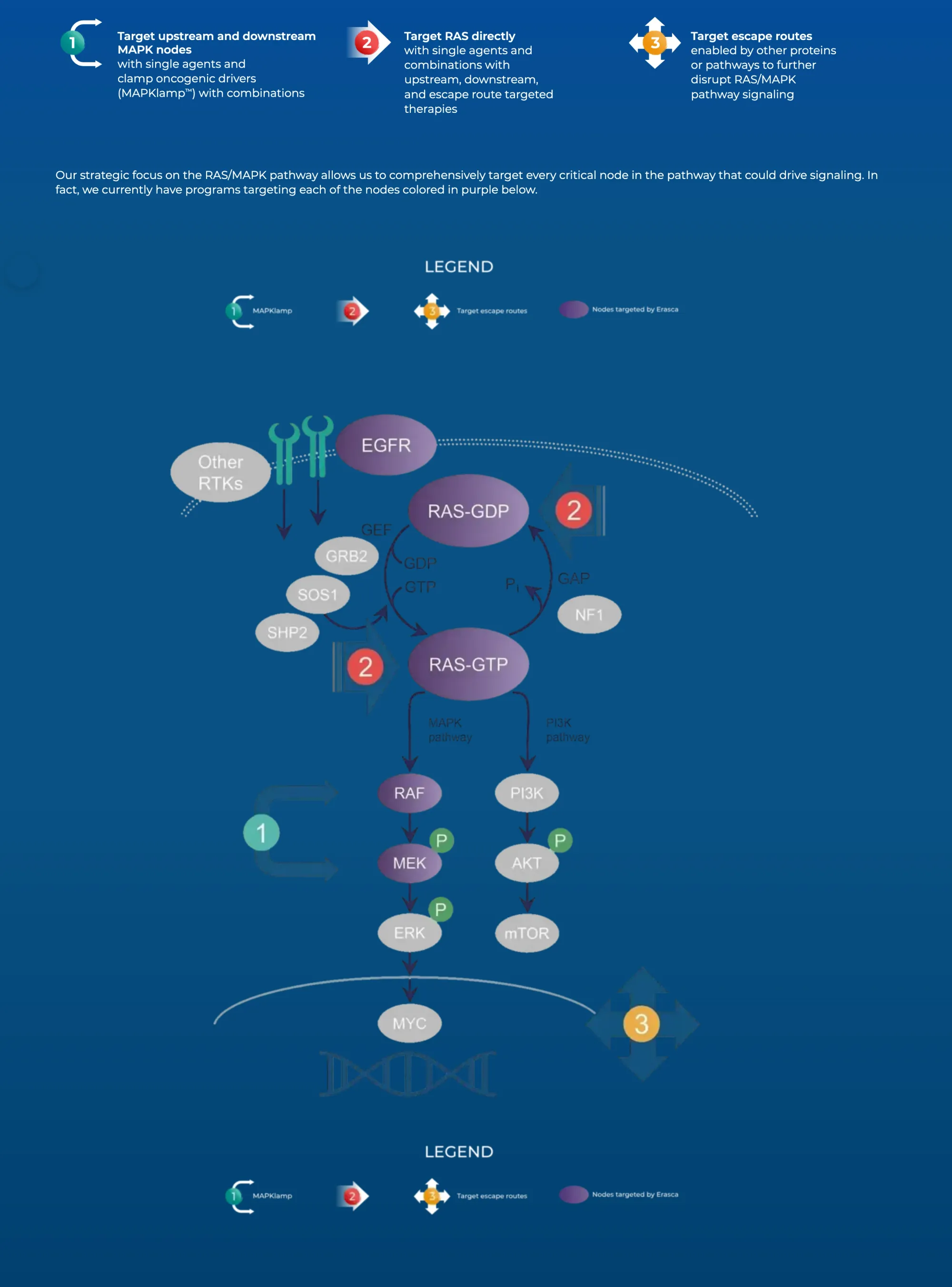
Erasca stock fell after a patient died in a cancer drug trial. Analysts say it’s likely a rare case, drug still shows promise.
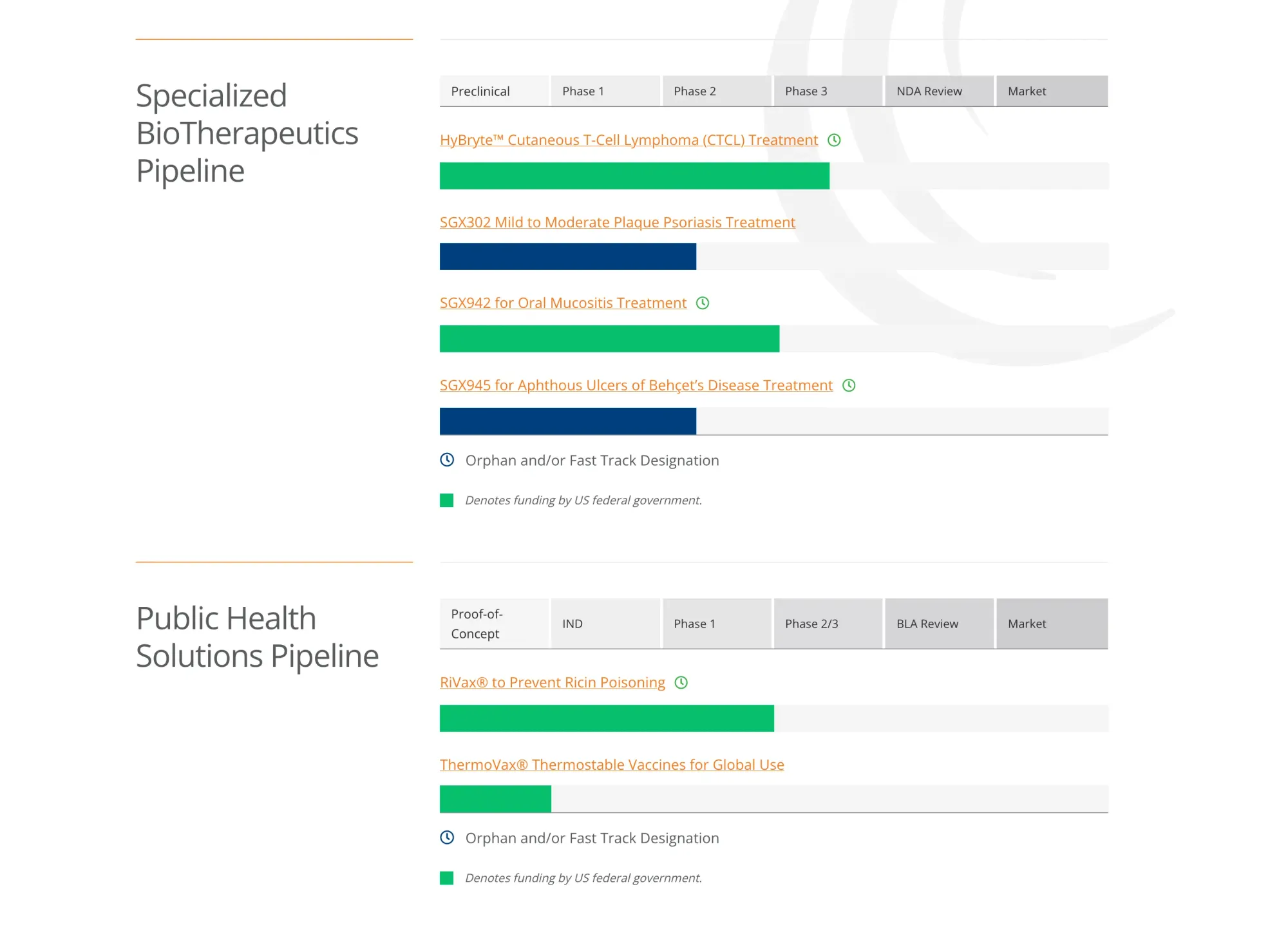
Soligenix stopped HyBryte CTCL trial early as it didn’t work well, though it was safe; company will review data and options.