Rocket Scores $180 Million Cash Boost – Extends Runway to 2028 for Heart Gene Therapies
Rocket Pharma sold an FDA voucher for $180M cash, boosting funds without dilution and extending runway to 2028 for heart gene therapies.

Rocket Pharma sold an FDA voucher for $180M cash, boosting funds without dilution and extending runway to 2028 for heart gene therapies.
FDA may pick Dr. Hemmati to lead CBER, which could speed up vaccine and biotech drug approvals, but decision is not final.

Rocket Pharmaceuticals won FDA approval for Kresladi and a valuable Priority Review Voucher, yet the stock dropped. Analysts explain the "sell the news" reaction, dilution concerns, and why they remain bullish with Outperform ratings.

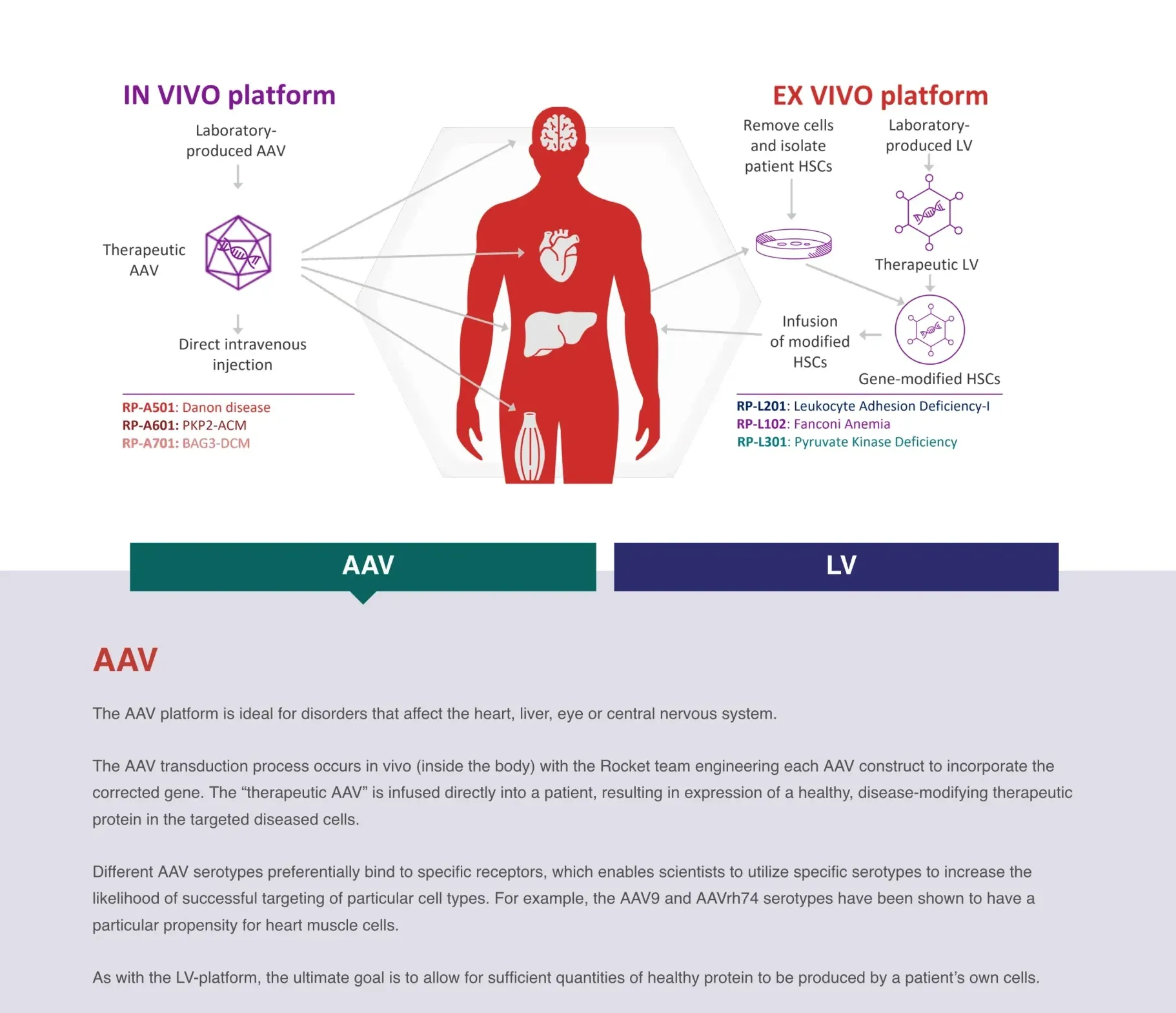
The FDA has approved KRESLADI, Rocket Pharma’s one-time gene therapy for children with severe LAD-I — a rare, life-threatening immune disorder.
Rocket Pharma's RP-A501 Danon disease trial resumed after FDA lifted hold due to patient death. Shares surged. It will use a lower dose & modified regimen. Signals FDA support for gene therapy despite investor wariness & other patient deaths, like Sarepta's and Neurogene's.