
FDA Tightens Rules on Copycat Ozempic and Wegovy – No More Routine Compounding
FDA limits copycat GLP-1 drugs; pharmacies can only make them during shortages. Original meds like Foundayo now safer & more available.

FDA limits copycat GLP-1 drugs; pharmacies can only make them during shortages. Original meds like Foundayo now safer & more available.
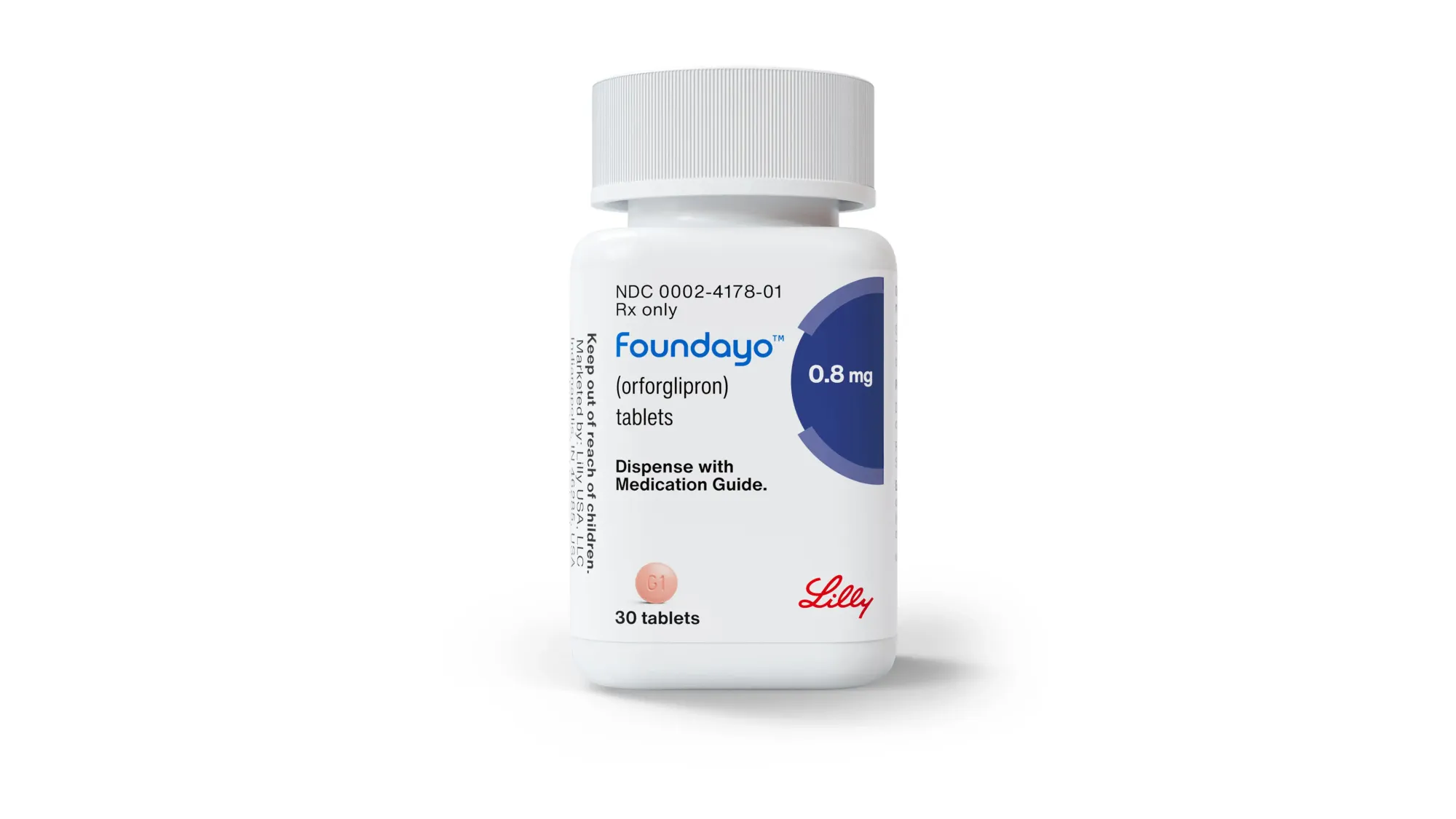
Eli Lilly’s Foundayo (orforglipron) is now FDA-approved — the only GLP-1 pill for weight loss you can take any time of day, with or without food or water. Average 27 lb loss.
Scholar Rock resubmits its BLA for apitegromab to treat SMA. FDA acceptance expected in 30 days, PDUFA target late September 2026.
Eli Lilly has signed a $2.75 billion deal with Hong Kong-listed AI biotech Insilico Medicine for a GLP-1 diabetes drug.

Eli Lilly’s retatrutide delivered up to 2.0% A1C reduction and 16.8% weight loss (36.6 lbs) in its first Phase 3 trial for type 2 diabetes. Analysts call it a key growth driver for Lilly with strong results vs Mounjaro.

Bernstein initiates Underperform on Novo Nordisk, citing endless earnings downgrades, weak catalysts, U.S. price deflation, and 2032 patent cliff risks.