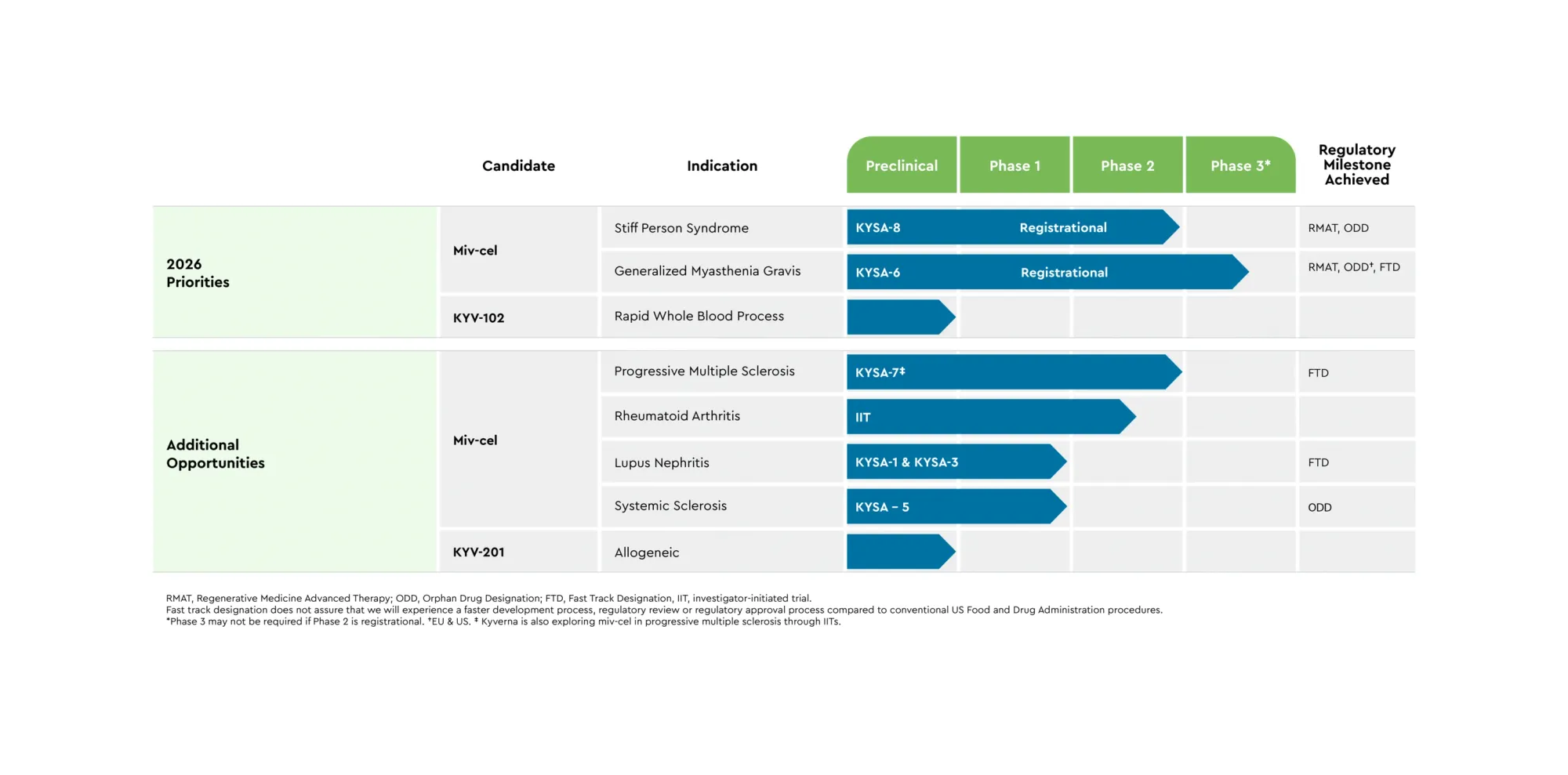
Kyverna’s miv-cel Scores Historic Win in Stiff Person Syndrome – First CAR-T Poised for FDA Approval
Kyverna’s CAR-T drug greatly improved rare SPS: patients walked 46% faster, many stopped meds; FDA filing planned 2026.

Kyverna’s CAR-T drug greatly improved rare SPS: patients walked 46% faster, many stopped meds; FDA filing planned 2026.
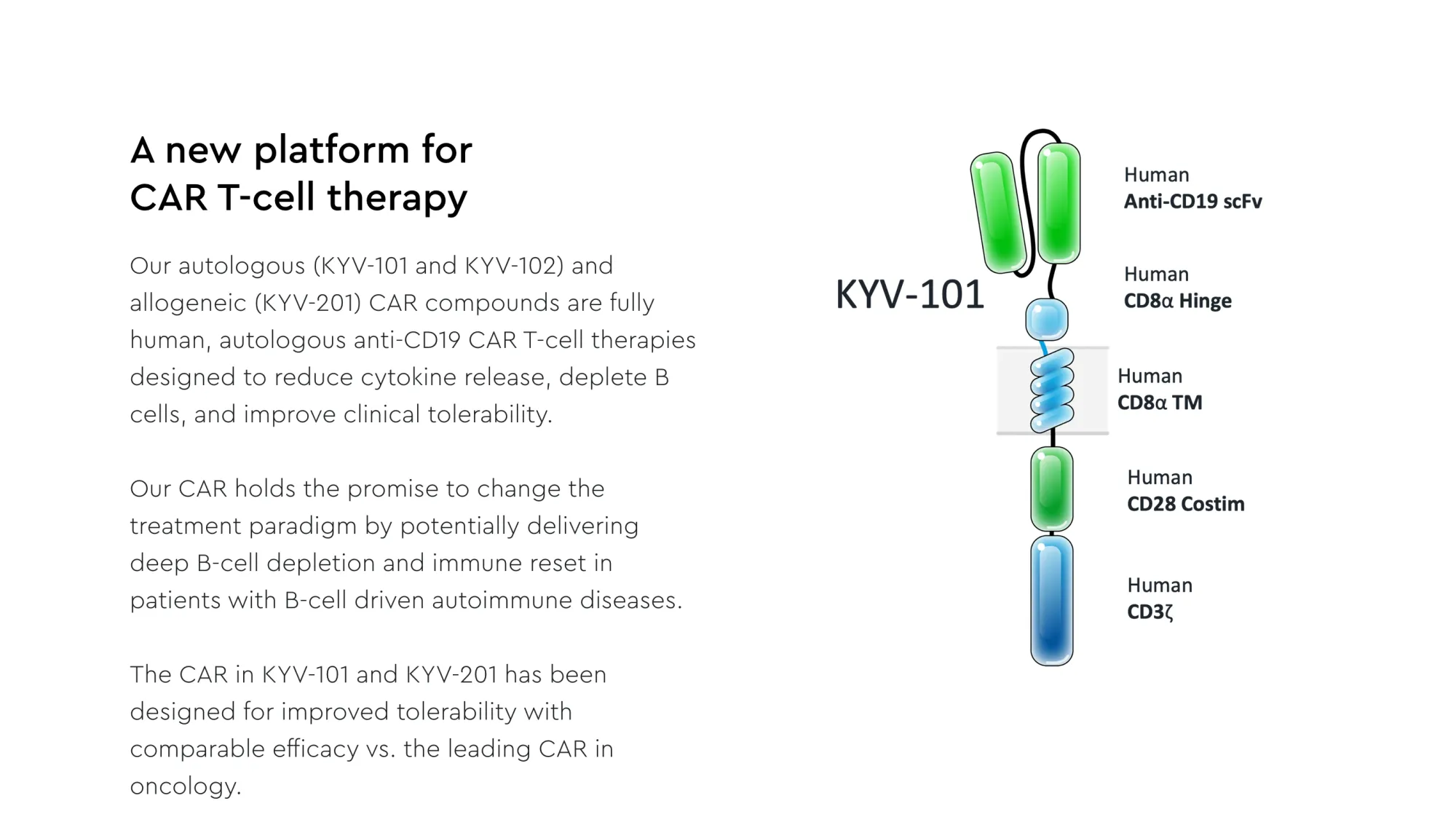
Kyverna Therapeutics (KYTX) announces positive results from clinical trial of miv-cel CAR T-cell therapy for stiff person syndrome, potentially the first FDA-approved treatment for this rare autoimmune disease.