ImmunityBio Quickly Fixes FDA Ad Concerns: Podcast Pulled, TV Spot Never Aired
ImmunityBio addressed FDA ad concerns, removed a podcast, and clarified ANKTIVA info while tightening rules on future promotions.

ImmunityBio addressed FDA ad concerns, removed a podcast, and clarified ANKTIVA info while tightening rules on future promotions.

ImmunityBio strengthens its balance sheet with $100 million financing, including $75M non-dilutive funding from Oberland Capital and $25M debt conversion.
ImmunityBio shares dropped 20% after the FDA issued a warning letter accusing the company of false claims that ANKTIVA cures all cancers.
ImmunityBio just hit a huge milestone – they can now mass-produce and freeze powerful NK cells from healthy and cancer donors. One simple blood draw gives 8–10 ready-to-use doses.
ImmunityBio’s ANKTIVA + BCG just received European Commission approval – the first new immunotherapy in Europe for hard-to-treat bladder cancer. Now available in 33 countries, offering hope without surgery.
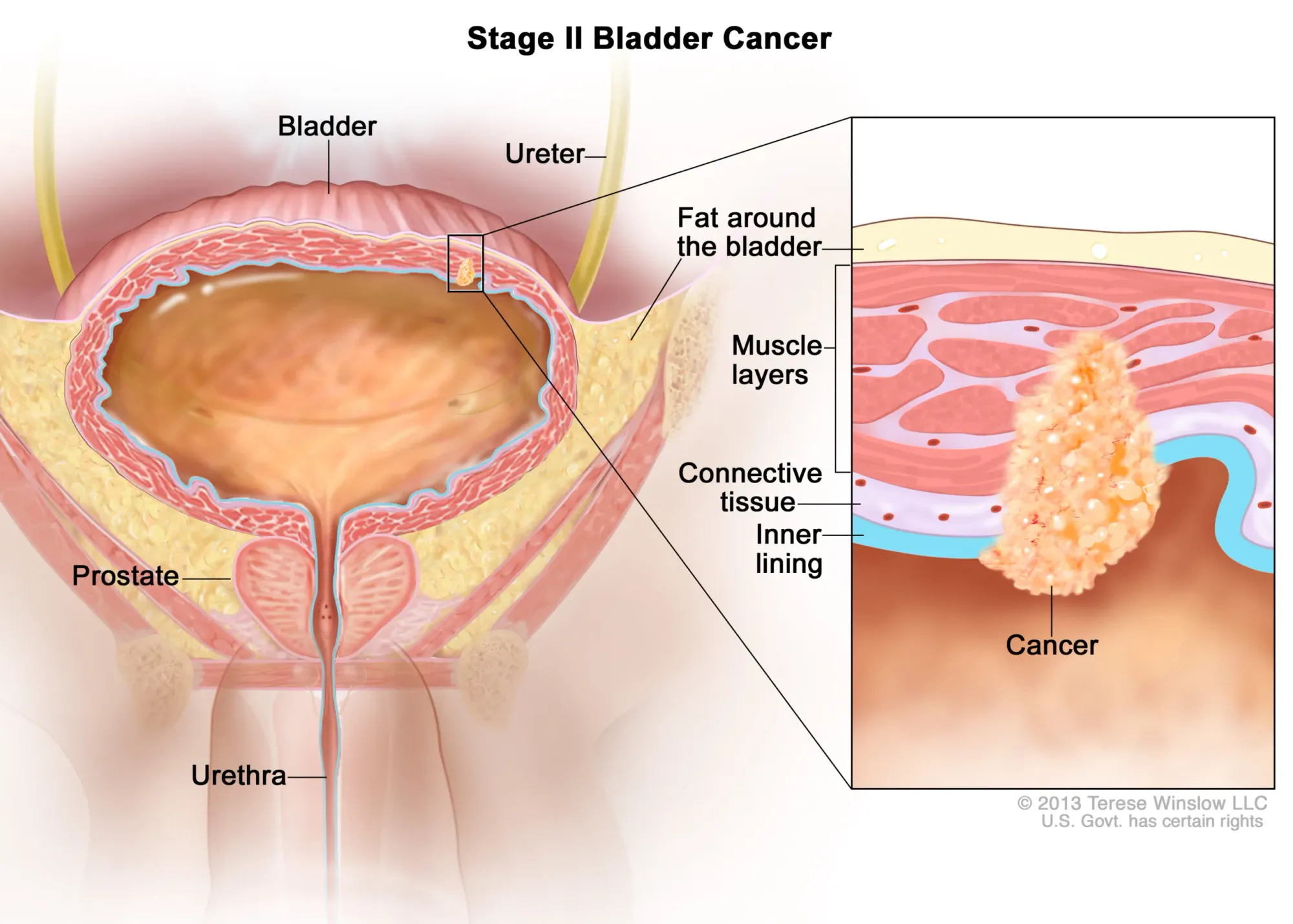
ImmunityBio advances FDA talks for ANKTIVA in papillary bladder cancer. Treatment shows 96% survival & 80% avoid bladder removal at 3 years, offering hope vs. current option: invasive surgery.