China’s NMPA Fast-Tracks Gyre Therapeutics’ Hydronidone (F351)
China’s NMPA has granted priority review to Gyre Therapeutics’ NDA for Hydronidone (F351) for chronic hepatitis B liver fibrosis.

China’s NMPA has granted priority review to Gyre Therapeutics’ NDA for Hydronidone (F351) for chronic hepatitis B liver fibrosis.
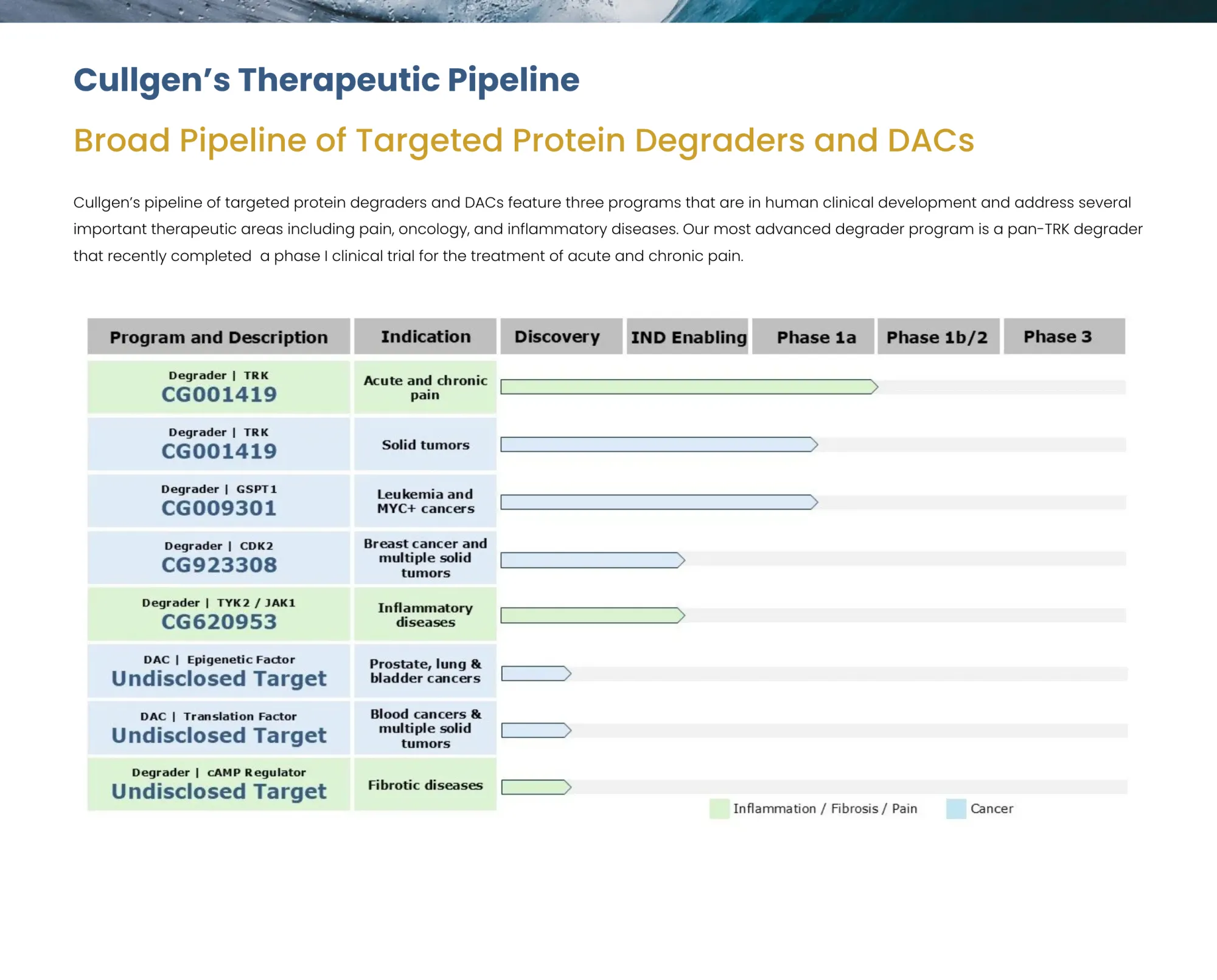
Gyre Therapeutics agrees to acquire Cullgen in $300M all-stock deal, gaining TPD/DAC platform for oncology, pain, and inflammatory diseases. Deal to close Q2 2026 after Cullgen ends Pulmatrix merger.

Gyre Therapeutics (GYRE) stock jumps 14% after Chinese regulators agree to conditional approval pathway for Hydronidone, a first-in-class liver fibrosis treatment.