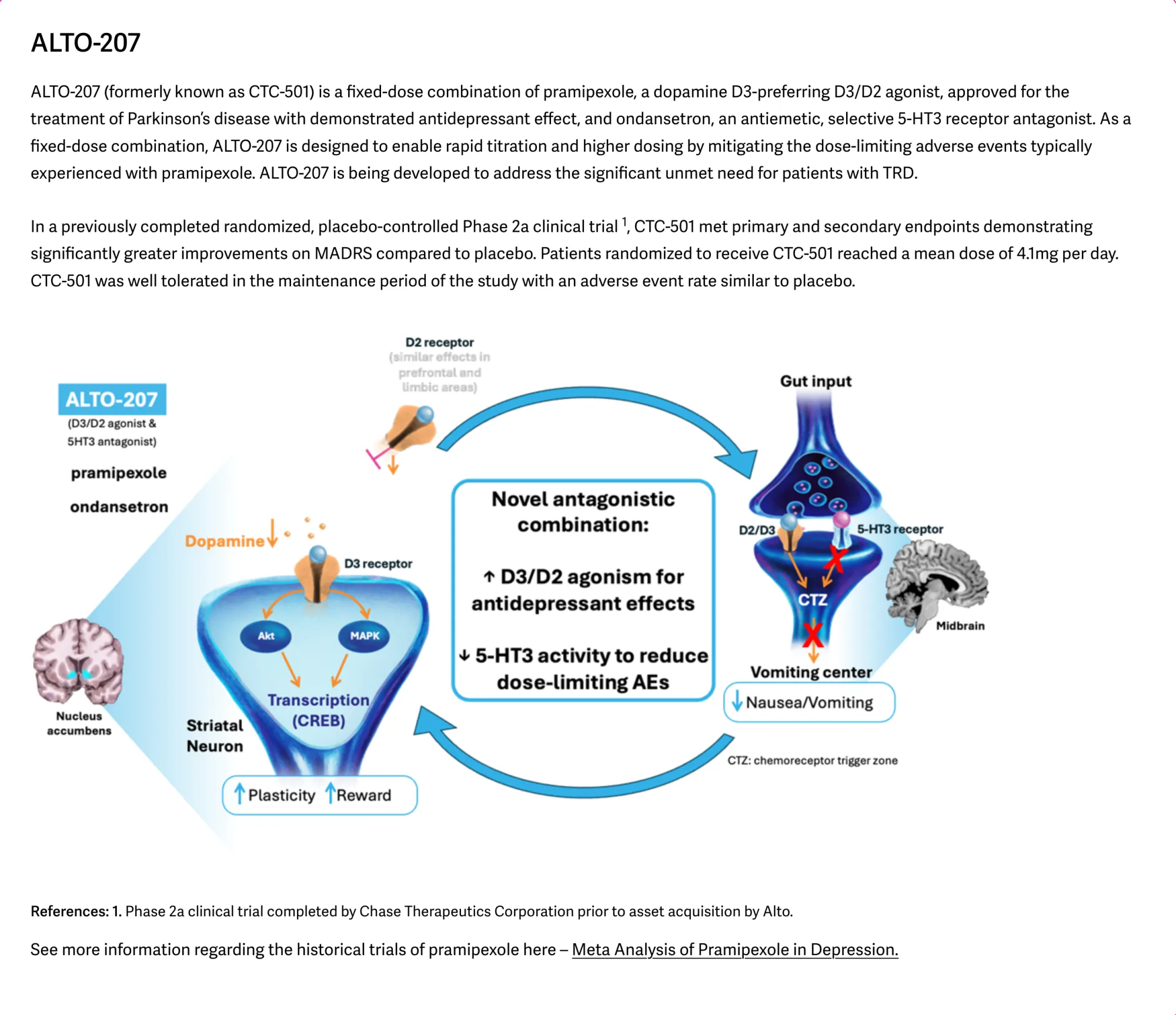
ALTO-101 Misses Primary Endpoint in Schizophrenia Trial, Company Shifts Focus to ALTO-207
ALTO-101 misses Phase 2 endpoints but shows signals. Alto pivots to ALTO-207 in treatment-resistant depression with strong prior data.

ALTO-101 misses Phase 2 endpoints but shows signals. Alto pivots to ALTO-207 in treatment-resistant depression with strong prior data.

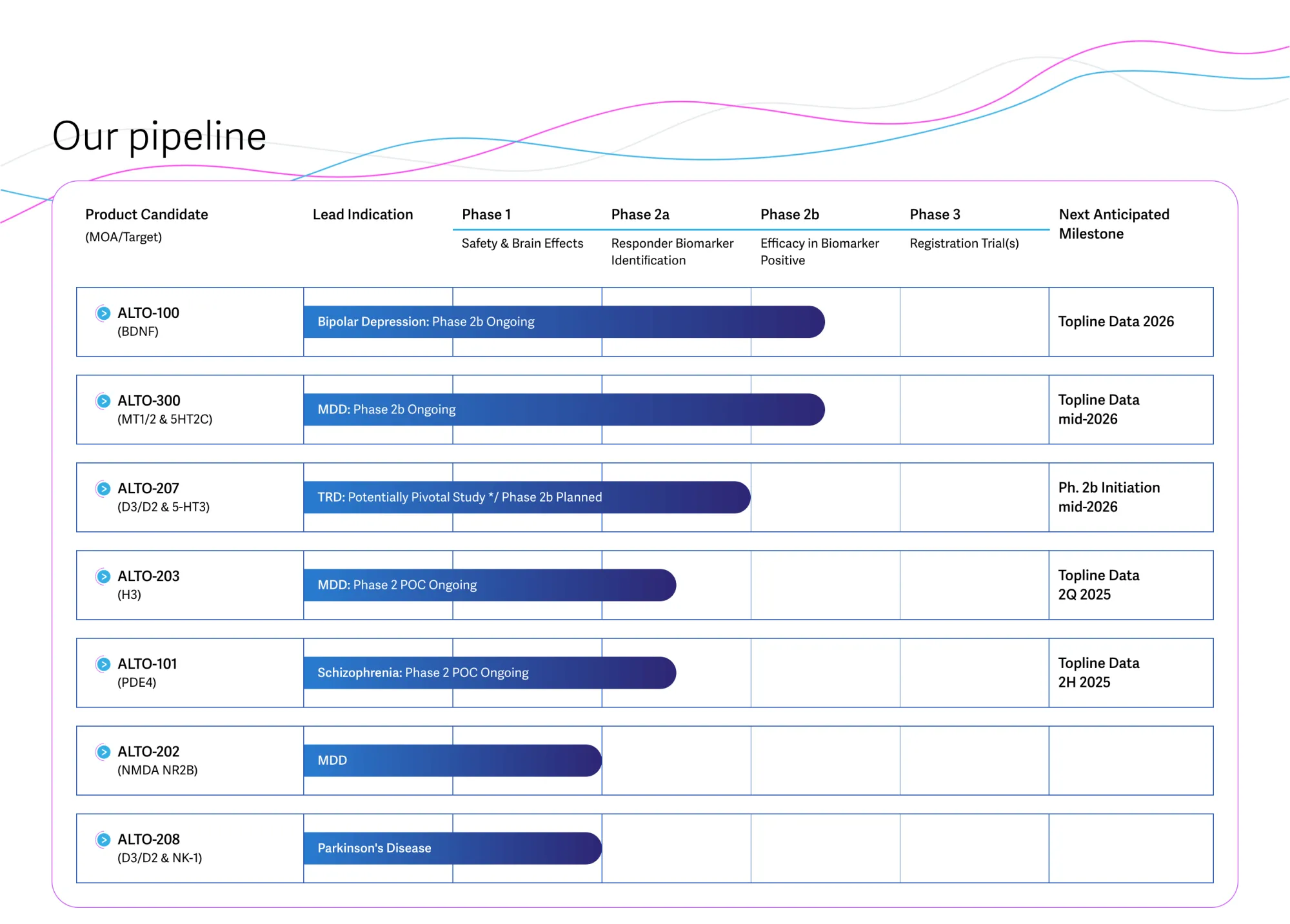
BTIG initiates coverage of Alto Neuroscience with a Buy rating and $27 price target, highlighting innovative biomarkers and upcoming data readouts in psychiatric drug development.
Jefferies boosts Alto Neuroscience price target to $25, citing upcoming Phase II data and investor interest in its precision psychiatry treatments.
Baird raises Alto Neuroscience price target to $16 as company secures $50M funding and FDA approval to fast-track ALTO-207 depression drug development.
Alto Neuroscience has received FDA Fast Track designation for ALTO-101, a potential new treatment for the cognitive difficulties—like poor memory and focus—that millions of schizophrenia patients experience, for which there are currently no approved options.