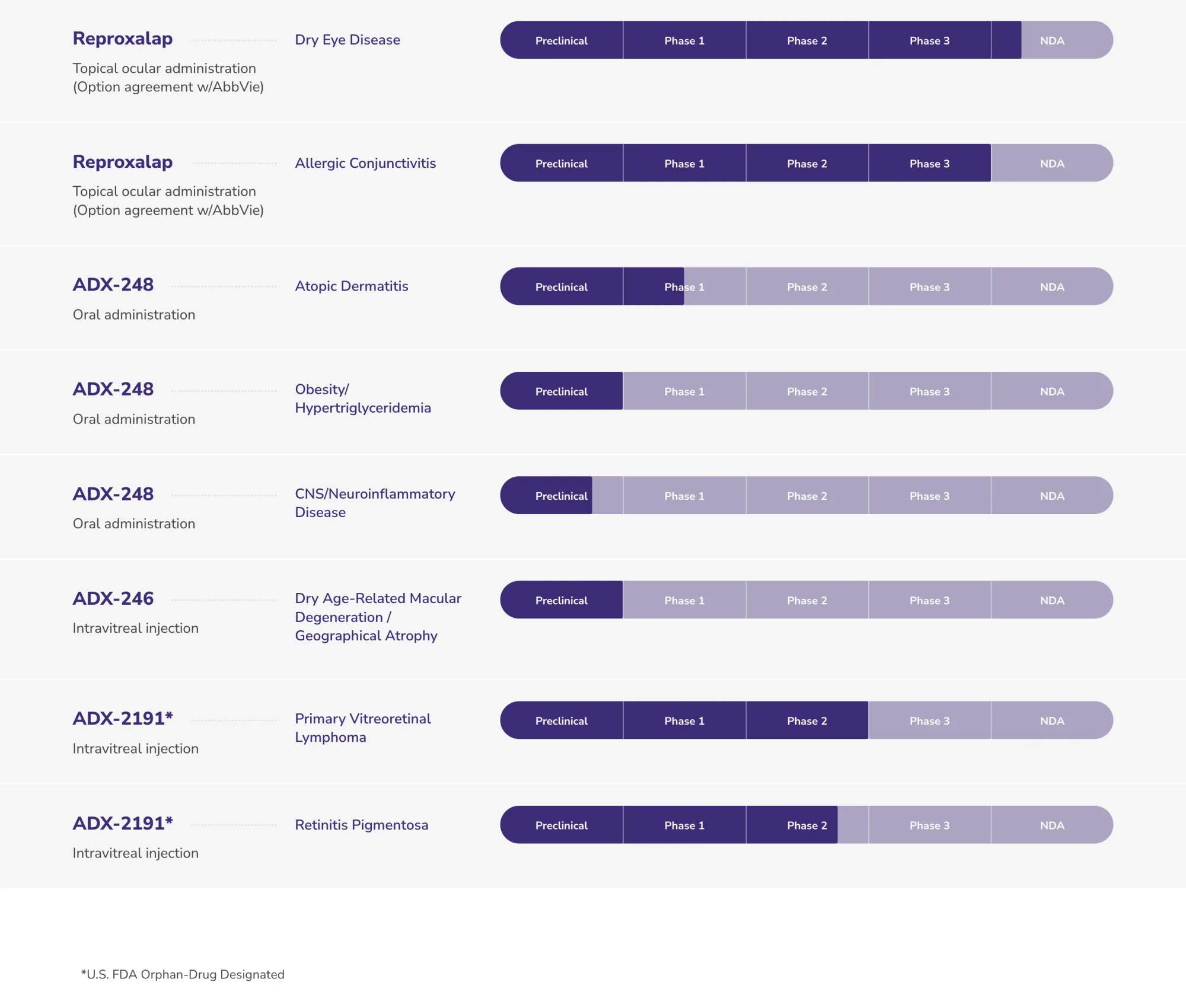
Aldeyra Stock Plunges as FDA Rejects Reproxalap Dry Eye Drug in Major Setback
Aldeyra Therapeutics receives FDA Complete Response Letter rejecting reproxalap for dry eye disease due to insufficient efficacy. No safety issues. ALDX shares crash.

Aldeyra Therapeutics receives FDA Complete Response Letter rejecting reproxalap for dry eye disease due to insufficient efficacy. No safety issues. ALDX shares crash.

Aldeyra Therapeutics (ALDX) receives FDA extension for reproxalap dry eye drug review. New decision date set for March 2026 as agency requests additional trial documentation.