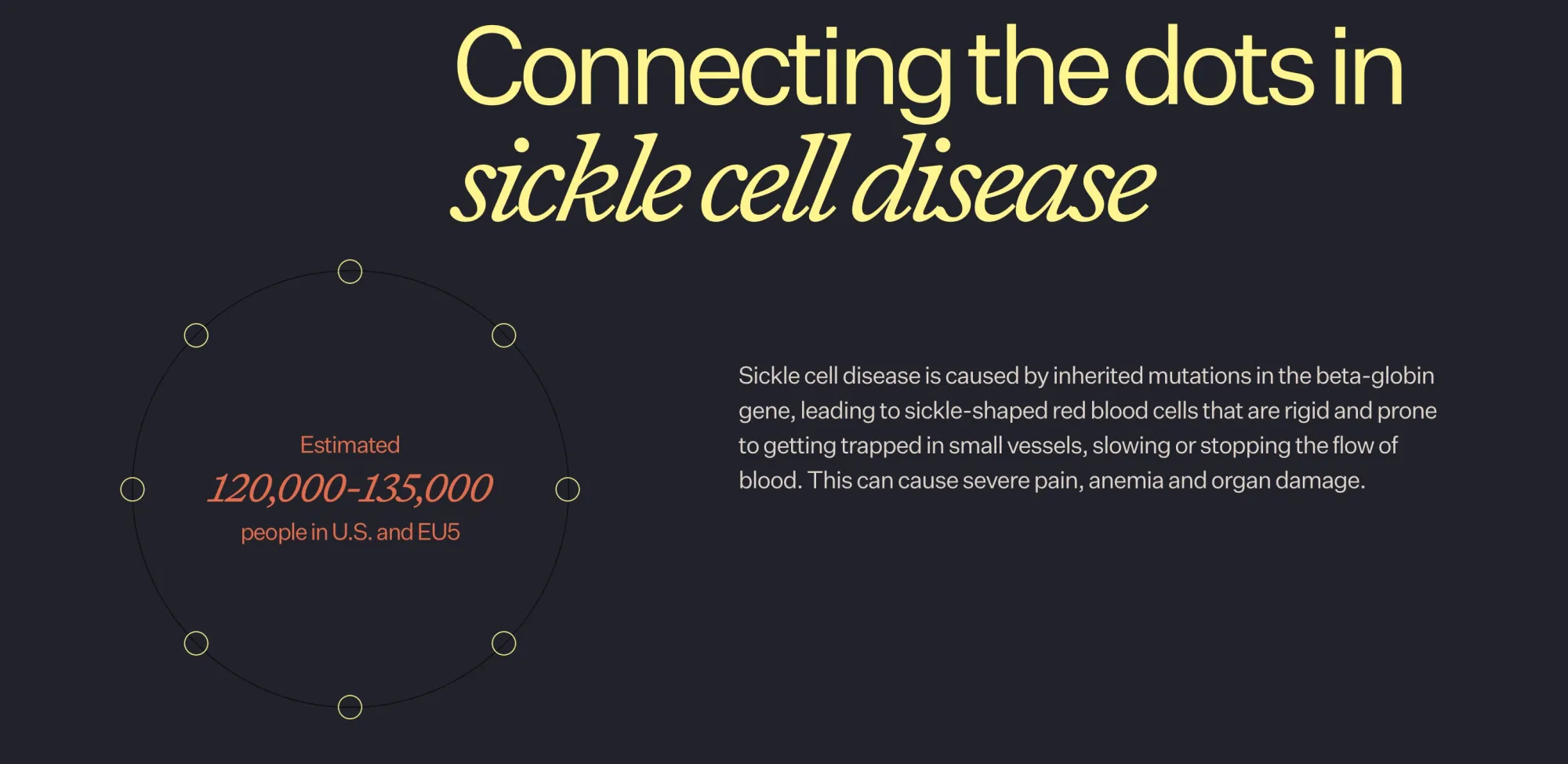
Agios Pharma Shares Crash as Novo Nordisk Posts Strong Phase 3 Sickle Cell Results
Novo Nordisk’s new pill cuts sickle cell pain crises, boosting outcomes and sending Agios Pharma shares down after strong trial results.

Novo Nordisk’s new pill cuts sickle cell pain crises, boosting outcomes and sending Agios Pharma shares down after strong trial results.
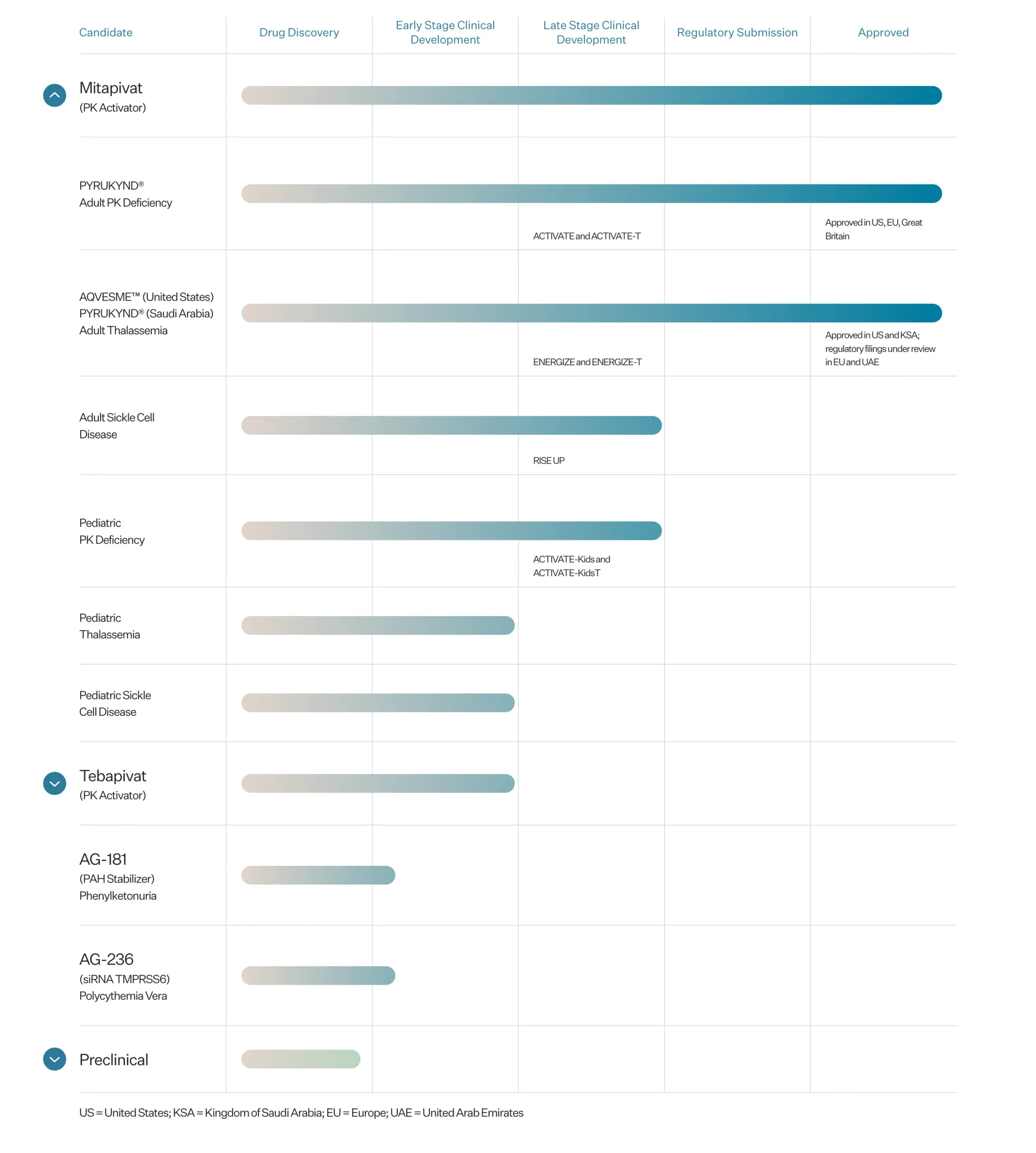
Agios is pursuing accelerated FDA approval for mitapivat, a new oral pill for sickle cell disease. The RISE UP trial showed better hemoglobin levels, fewer pain crises, and good safety.
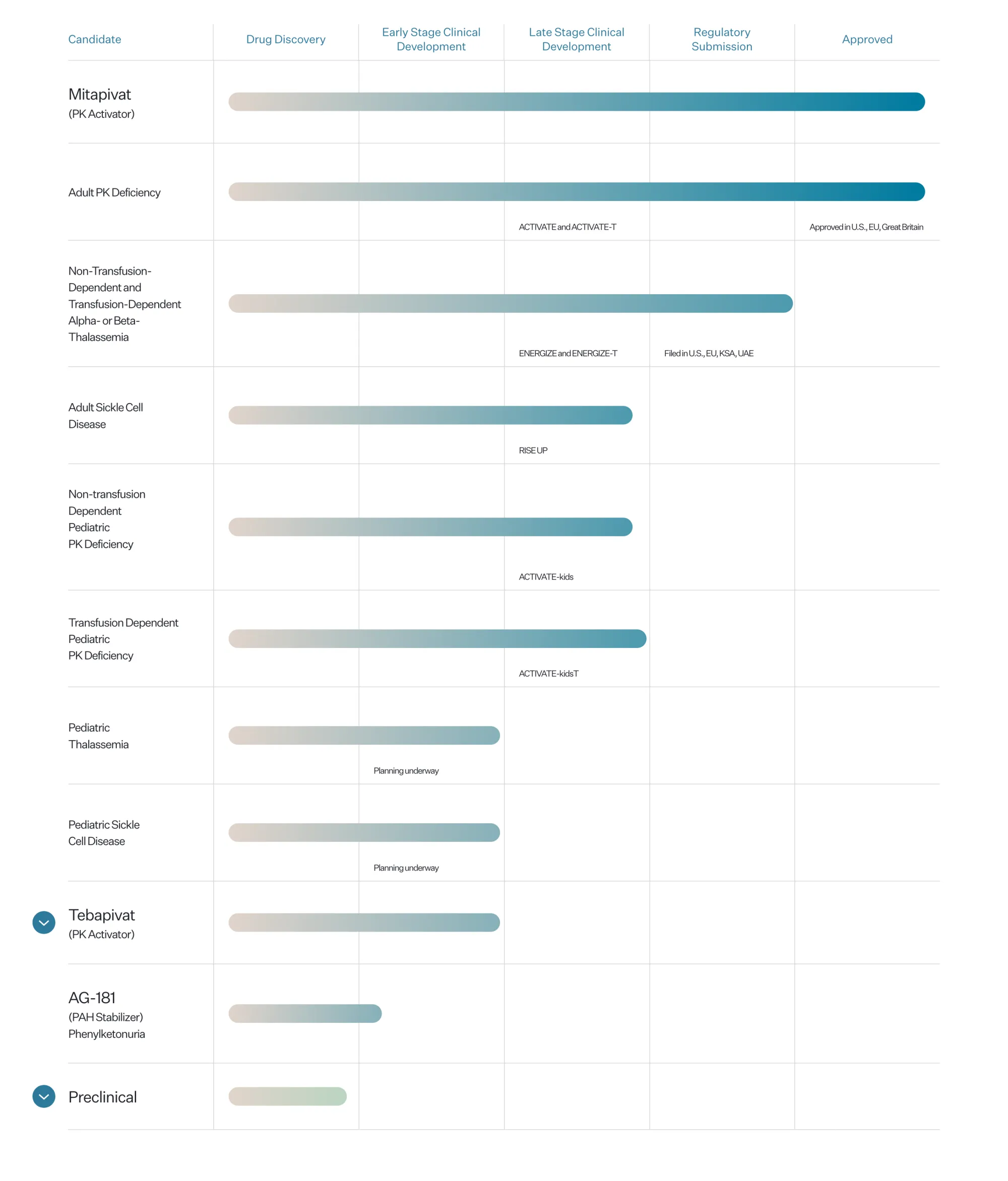
The FDA has approved Agios’ AQVESME (mitapivat), the first medicine for treating anemia in adults with alpha or beta-thalassemia, offering hope for thousands of patients.

Fulcrum Therapeutics rises as Agios’ RISE UP trial falls short. Analysts say results reduce competition in sickle cell disease and strengthen Fulcrum’s position ahead of new PIONEER data.

Agios Pharmaceuticals reports mixed Phase 3 results for mitapivat in sickle cell disease, meeting some endpoints while missing key pain crisis reduction goals.
The FDA extended PYRUKYND's review for thalassemia by 3 months to Dec 7, 2025, after Agios submitted a plan (REMS) to manage liver injury risk. This oral medicine aims to treat adult alpha- or beta-thalassemia.