
MoonLake's Sonelokimab Gets FDA Fast Track for Rare Skin Disease
MoonLake Immunotherapeutics receives FDA Fast Track designation for sonelokimab in palmoplantar pustulosis, accelerating development for this untreated skin condition.


MoonLake Immunotherapeutics receives FDA Fast Track designation for sonelokimab in palmoplantar pustulosis, accelerating development for this untreated skin condition.

President Trump unveils Project Vault, a $12 billion initiative to stockpile critical minerals, reducing US dependence on China and protecting manufacturers from supply disruptions.

Aquestive Therapeutics receives FDA Complete Response Letter for Anaphylm with packaging-related deficiencies. Company plans rapid resolution and Q3 2026 resubmission.

Guggenheim starts coverage on Cullinan Therapeutics with a Buy rating, citing promising Phase 1 data for T-cell engagers in autoimmune diseases and AML.
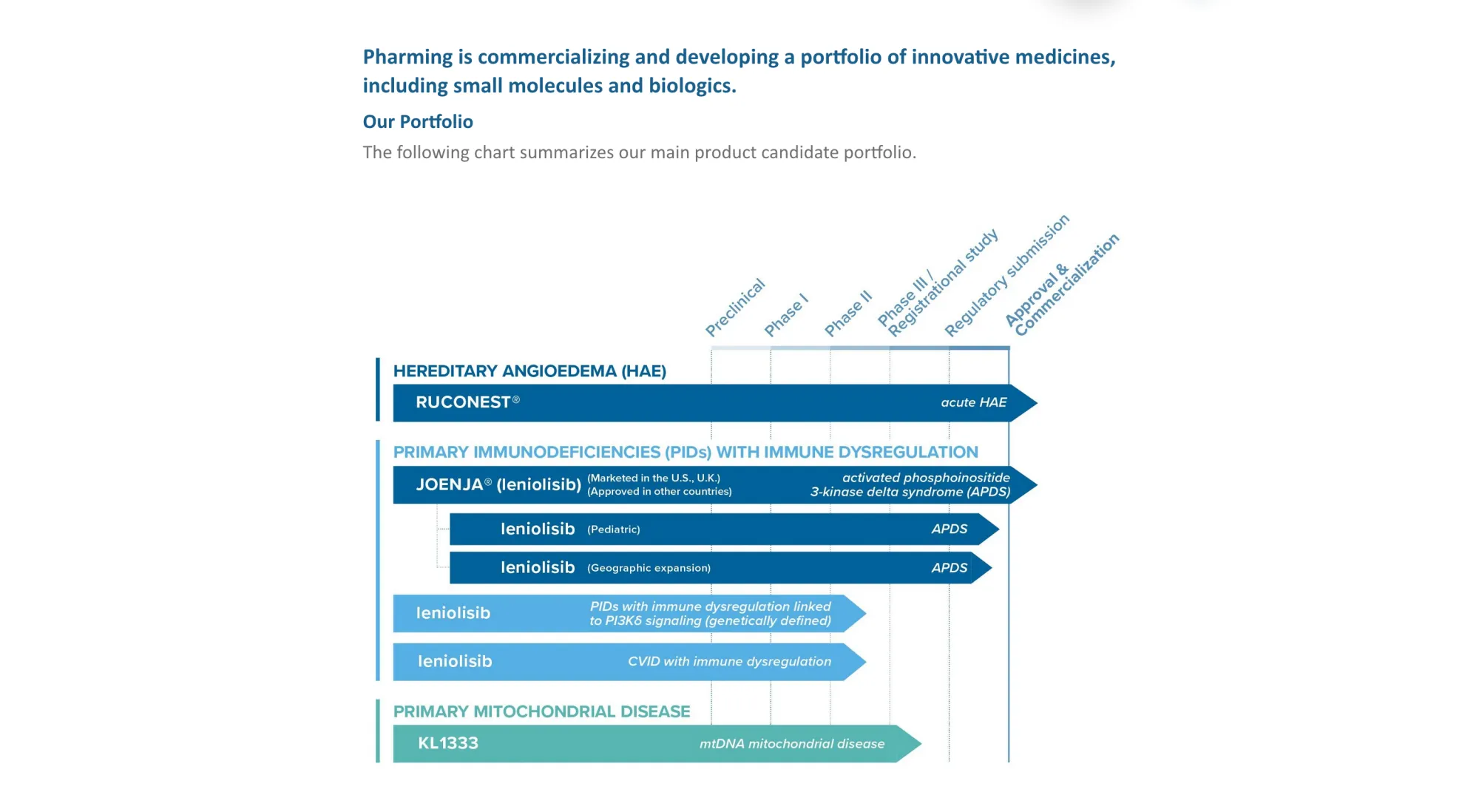
Pharming (PHAR) receives FDA Complete Response Letter for Joenja (leniolisib) in children aged 4-11 with APDS. Additional data needed on dosing and testing methods.

Guggenheim starts coverage of Avalo Therapeutics with a Buy rating, highlighting AVTX-009 as a promising IL-1beta treatment for Hidradenitis suppurativa (HS) and $2.1B peak sales potential.