Senti Bio's Cancer Treatment Gets Major FDA Recognition
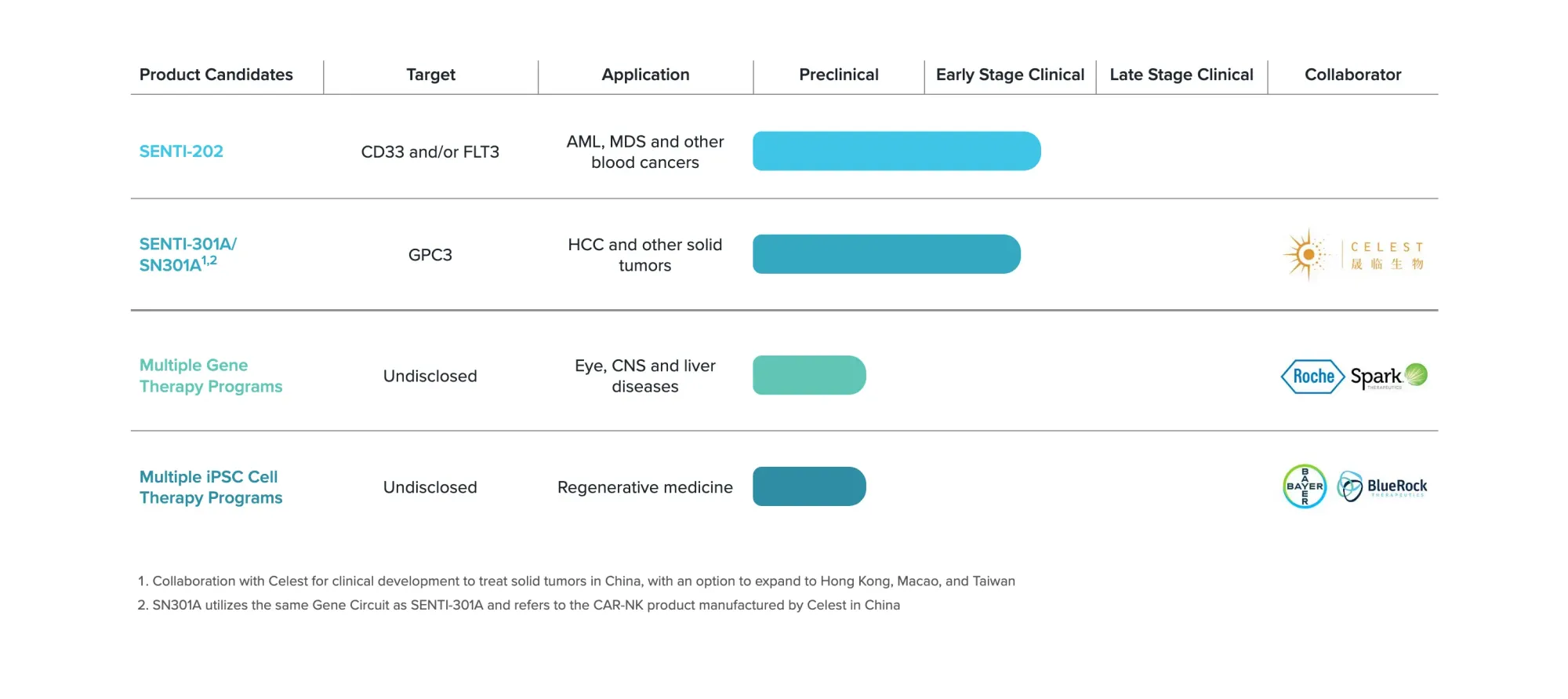
Senti Bio receives FDA's RMAT designation for SENTI-202, a breakthrough cell therapy showing promising results in treating relapsed acute myeloid leukemia patients.

Senti Bio receives FDA's RMAT designation for SENTI-202, a breakthrough cell therapy showing promising results in treating relapsed acute myeloid leukemia patients.

H.C. Wainwright initiates Senti Bio with Buy rating and $12 target. Cancer therapy developer's CAR-NK platform shows promise for blood cancers and solid tumors.