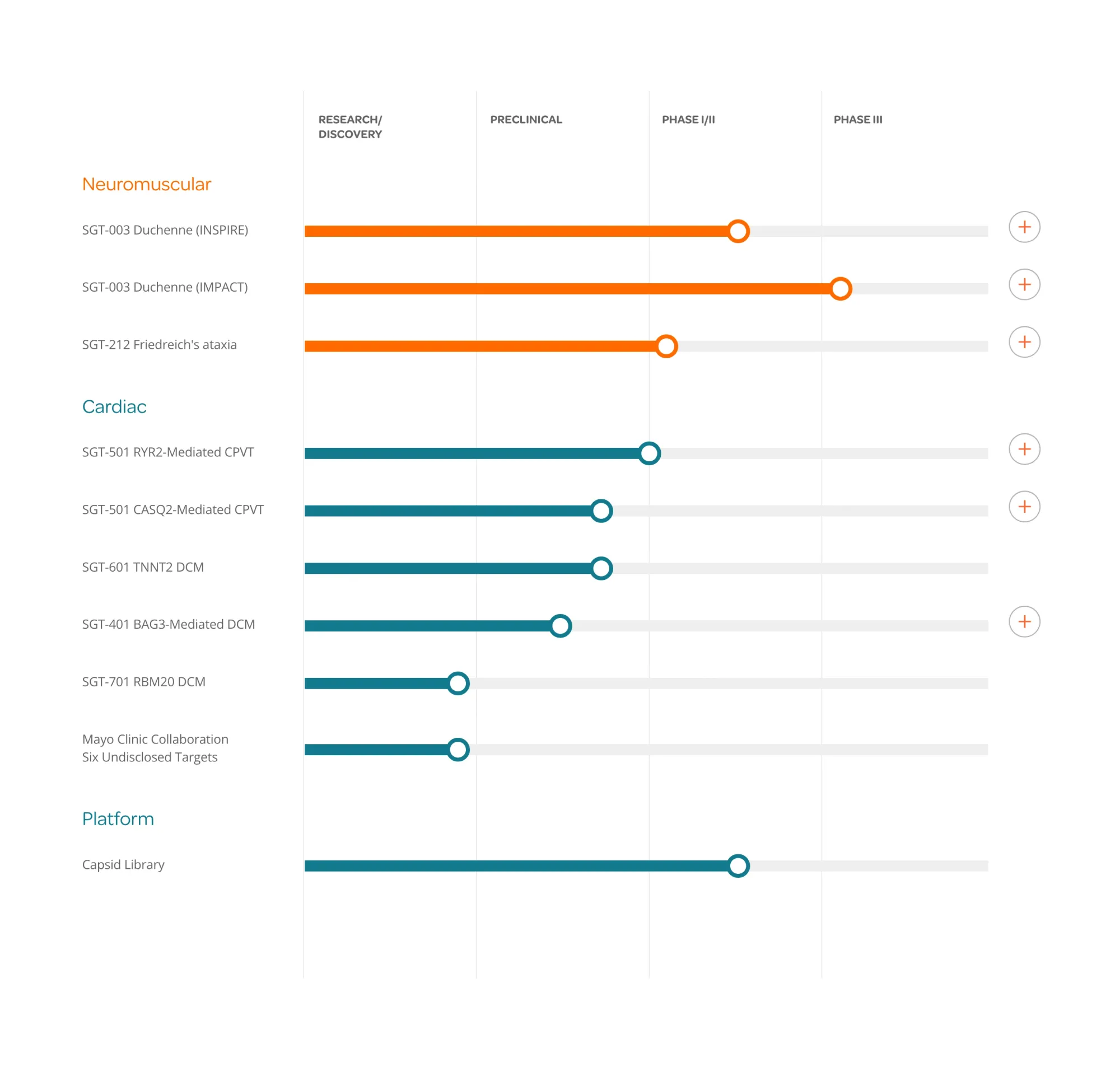
Solid Biosciences Wins FDA Orphan Drug Status for Friedreich's Ataxia Treatment
Solid Biosciences receives FDA Orphan Drug designation for SGT-212, a dual-route gene therapy treating Friedreich's ataxia, with first patient dosed in clinical trial.

Solid Biosciences receives FDA Orphan Drug designation for SGT-212, a dual-route gene therapy treating Friedreich's ataxia, with first patient dosed in clinical trial.

Sarepta rises as HHS recommends newborn screening for Duchenne muscular dystrophy and metachromatic leukodystrophy, aiming for earlier diagnosis and treatment.
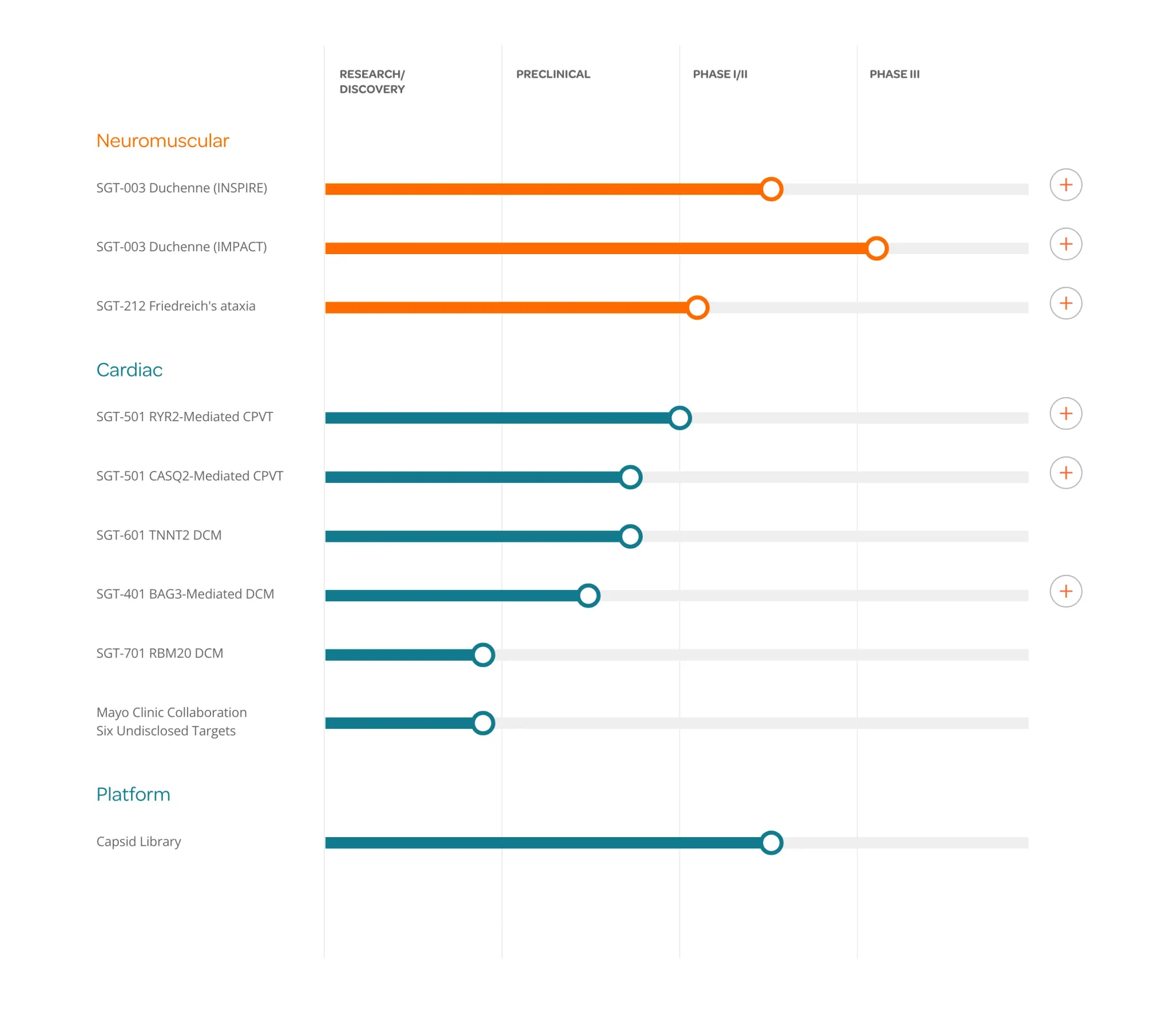
Needham initiates Buy on Solid Biosciences with $16 target, highlighting potential of SGT-003 gene therapy amid concerns over Sarepta’s Elevidys safety.