
PepGen Faces FDA Partial Hold on FREEDOM2 DM1 Trial, Advances in Multiple Countries
PepGen announces FDA partial clinical hold on FREEDOM2-DM1 trial due to preclinical questions, while gaining clearance in South Korea, Australia, New Zealand.

PepGen announces FDA partial clinical hold on FREEDOM2-DM1 trial due to preclinical questions, while gaining clearance in South Korea, Australia, New Zealand.
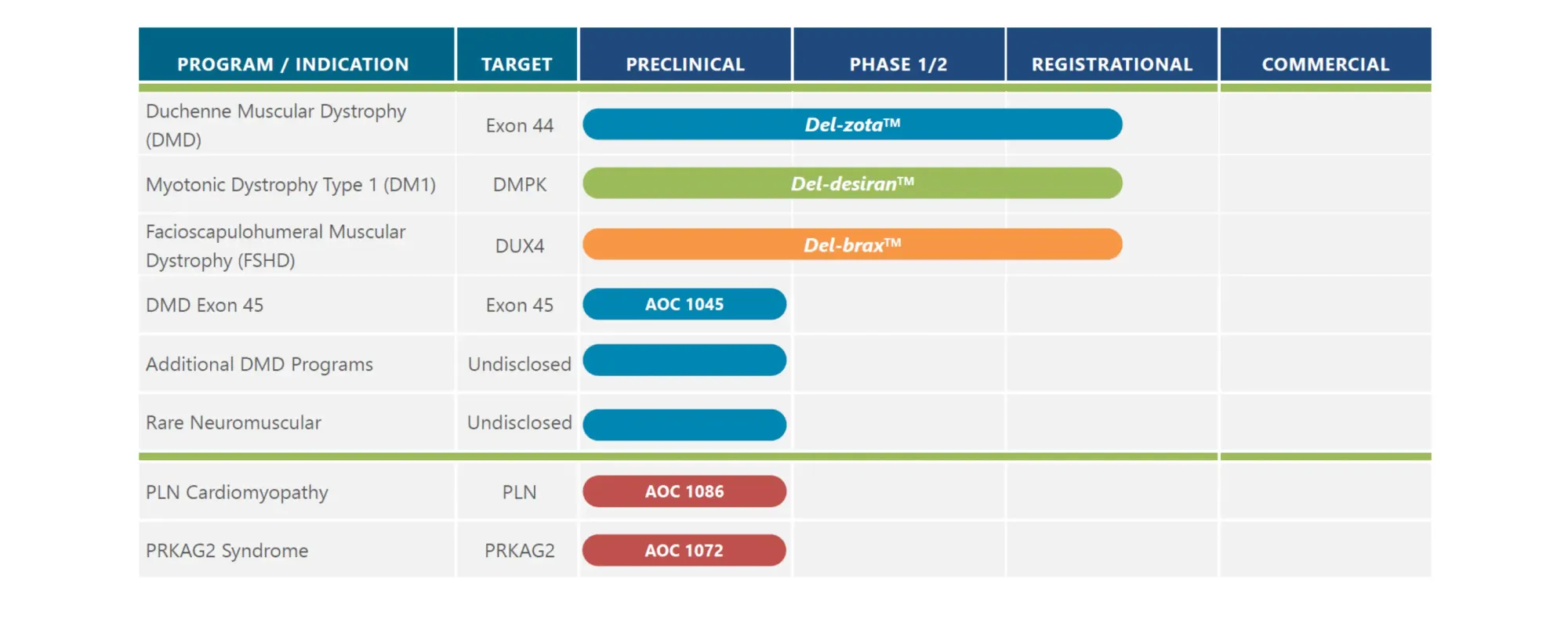
Novartis acquires Avidity Biosciences for $12 billion, gaining access to breakthrough RNA therapeutics for neuromuscular diseases as the Swiss drugmaker prepares for upcoming patent expirations on key drugs.

PepGen's DM1 drug (PGN-EDODM1) showed a record 53.7% splicing correction following a single dose. Experts expect this fix to lead to functional benefits for patients. A minor, reversible kidney issue was noted in one patient.