Prelude Therapeutics Gets FDA Green Light for Groundbreaking Blood Cancer Treatment
Prelude Therapeutics receives FDA clearance to begin testing PRT12396, a targeted therapy for myeloproliferative neoplasms affecting thousands of patients.

Prelude Therapeutics receives FDA clearance to begin testing PRT12396, a targeted therapy for myeloproliferative neoplasms affecting thousands of patients.
Guggenheim initiates coverage on Galecto with a Buy rating and $32 target, citing strong potential for its newly acquired drug DMR-001 and solid funding through 2029.
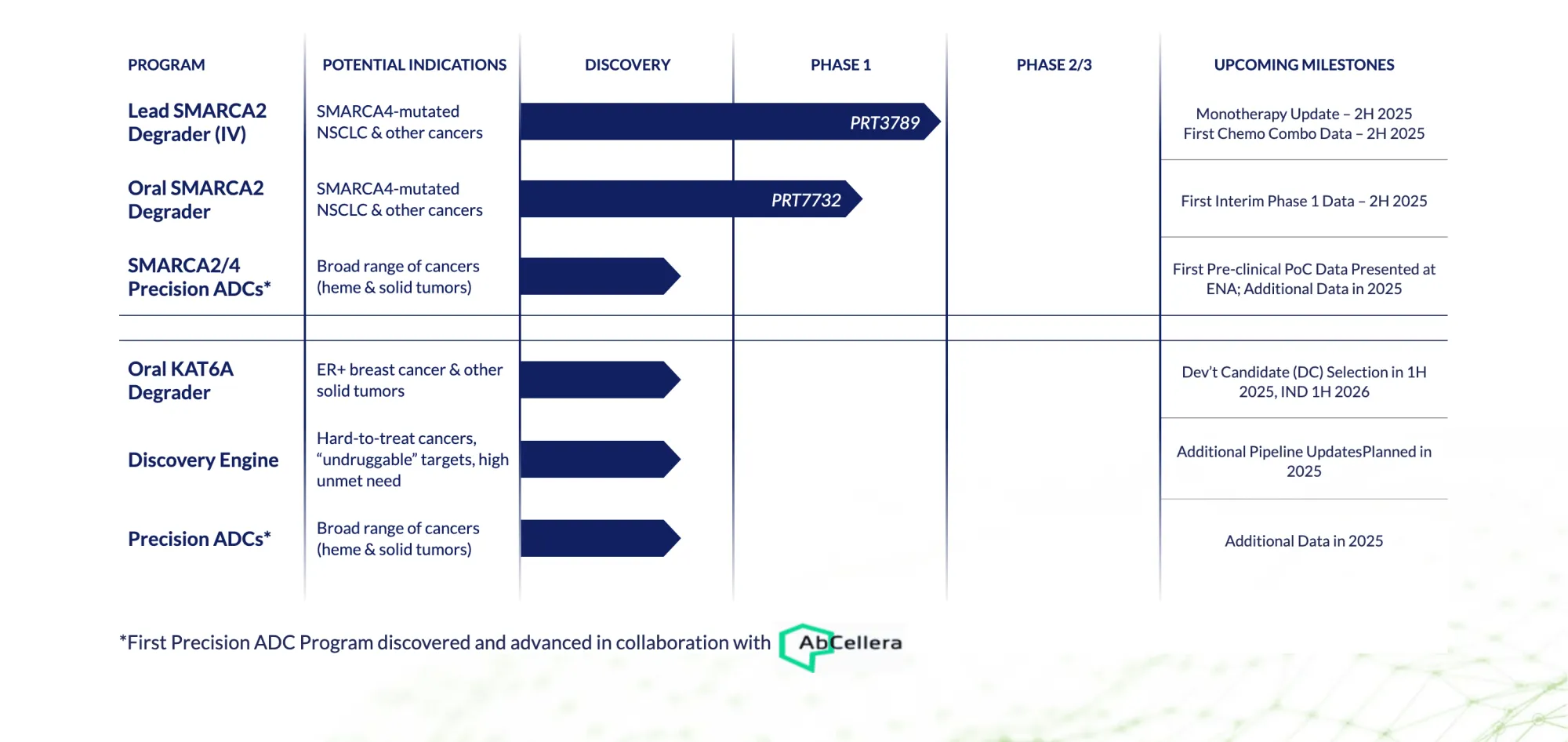
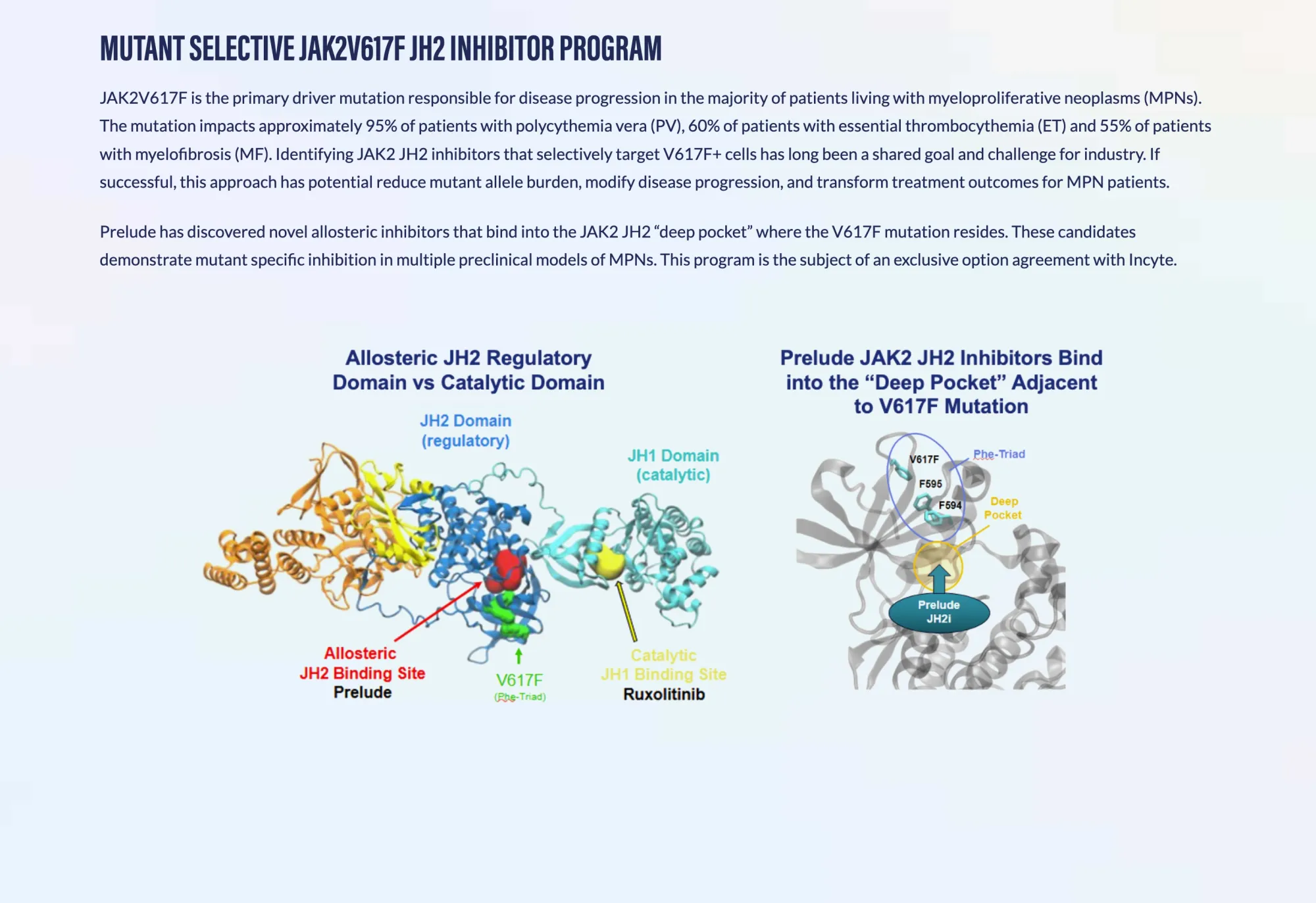
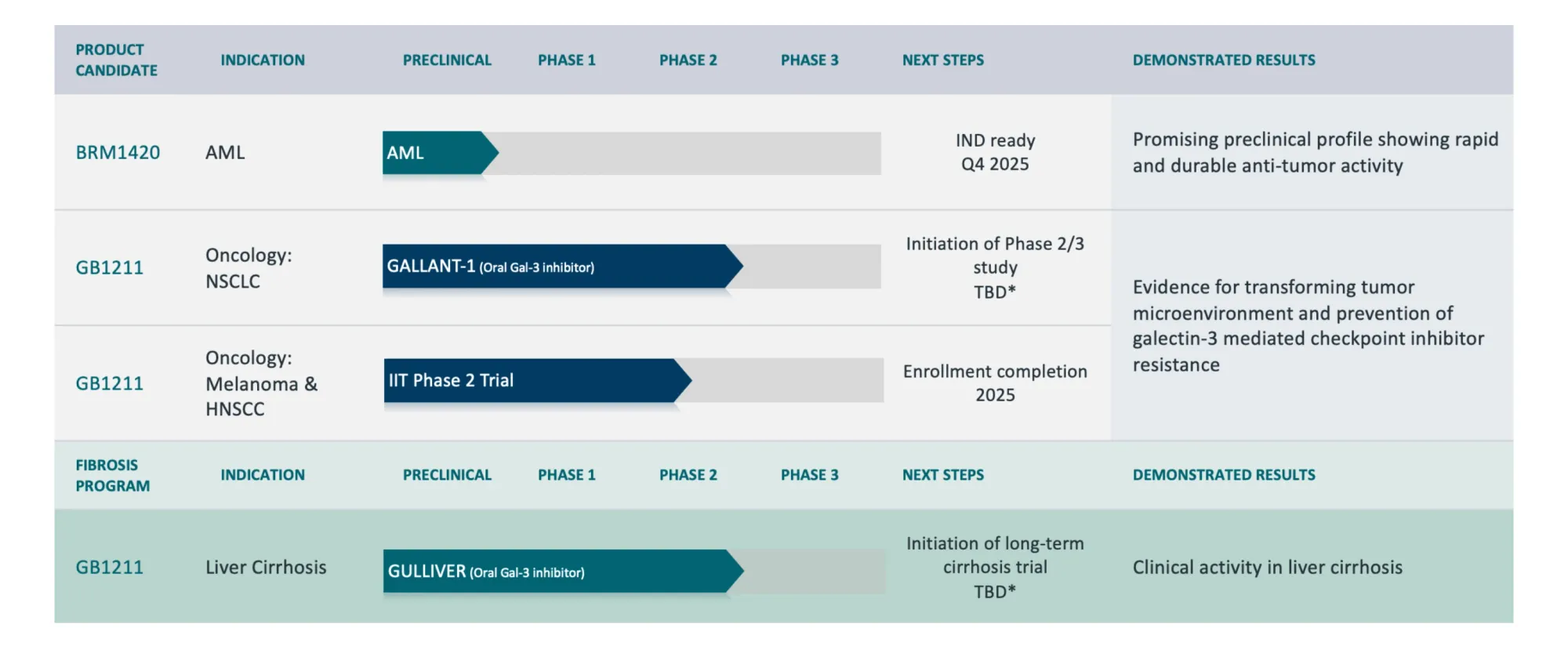
Prelude Therapeutics streamlines its oncology pipeline — prioritizing JAK2V617F and KAT6A programs, pausing SMARCA2, and securing major funding through an Incyte partnership.

Incyte’s early INCB0989 myelofibrosis data at ASH 2025 looks encouraging, prompting analyst upgrades and higher price targets from Mizuho, Wells Fargo, and Guggenheim.