Immunome's Desmoid Tumor Drug Shows 84% Success in Trial
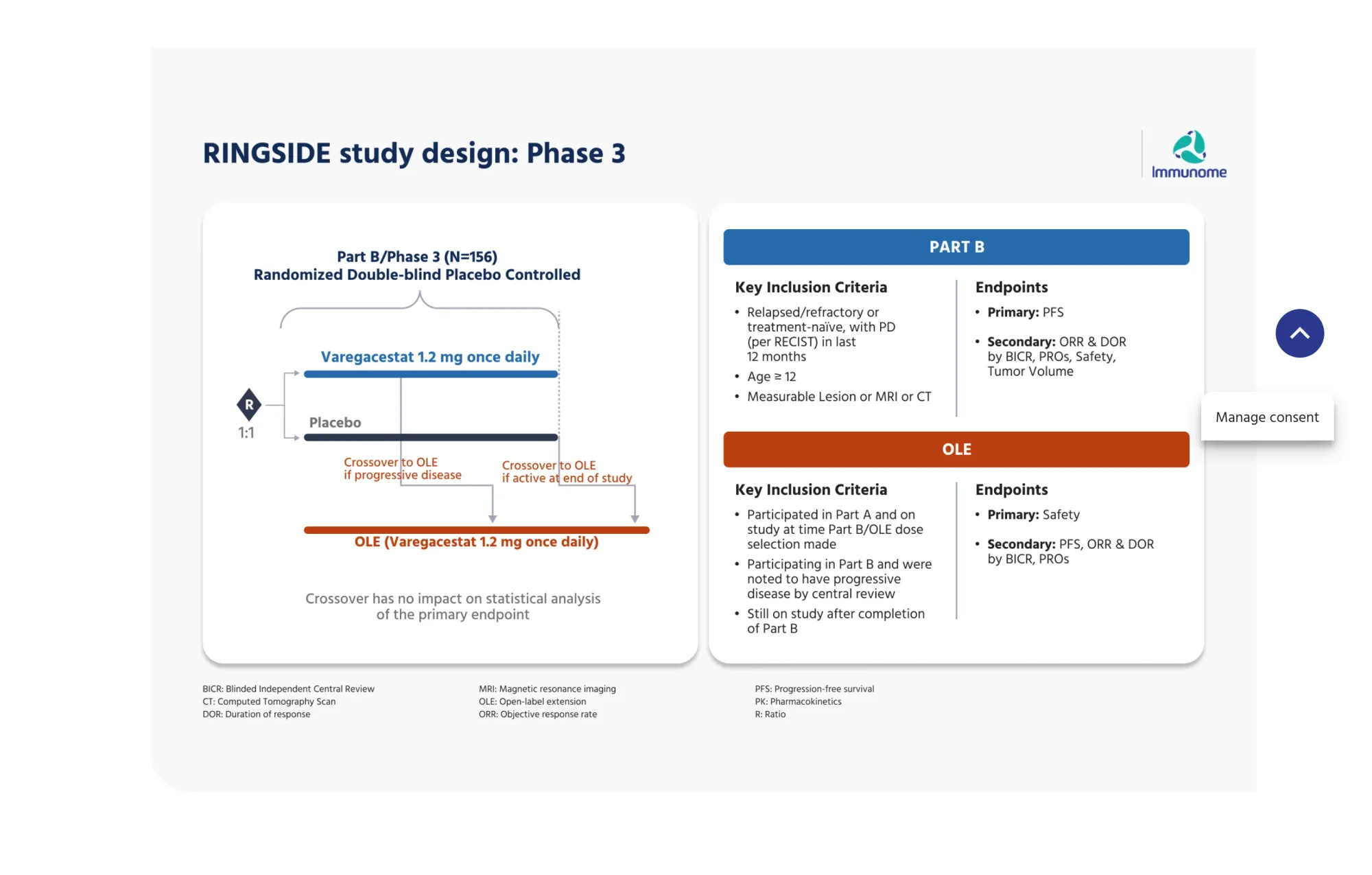
Immunome's (IMNM) Varegacestat reduces disease progression by 84% in Phase 3 desmoid tumor trial. FDA submission planned for Q2 2026. Once-daily oral pill.

Immunome's (IMNM) Varegacestat reduces disease progression by 84% in Phase 3 desmoid tumor trial. FDA submission planned for Q2 2026. Once-daily oral pill.
Goldman Sachs gave Immunome a "Buy" rating ($26 target). The company creates potentially best-in-class cancer drugs, including Varegacestat, which is seen as "differentiated." Key trial results are expected at the end of 2025.