Senti Bio's Cancer Treatment Gets Major FDA Recognition
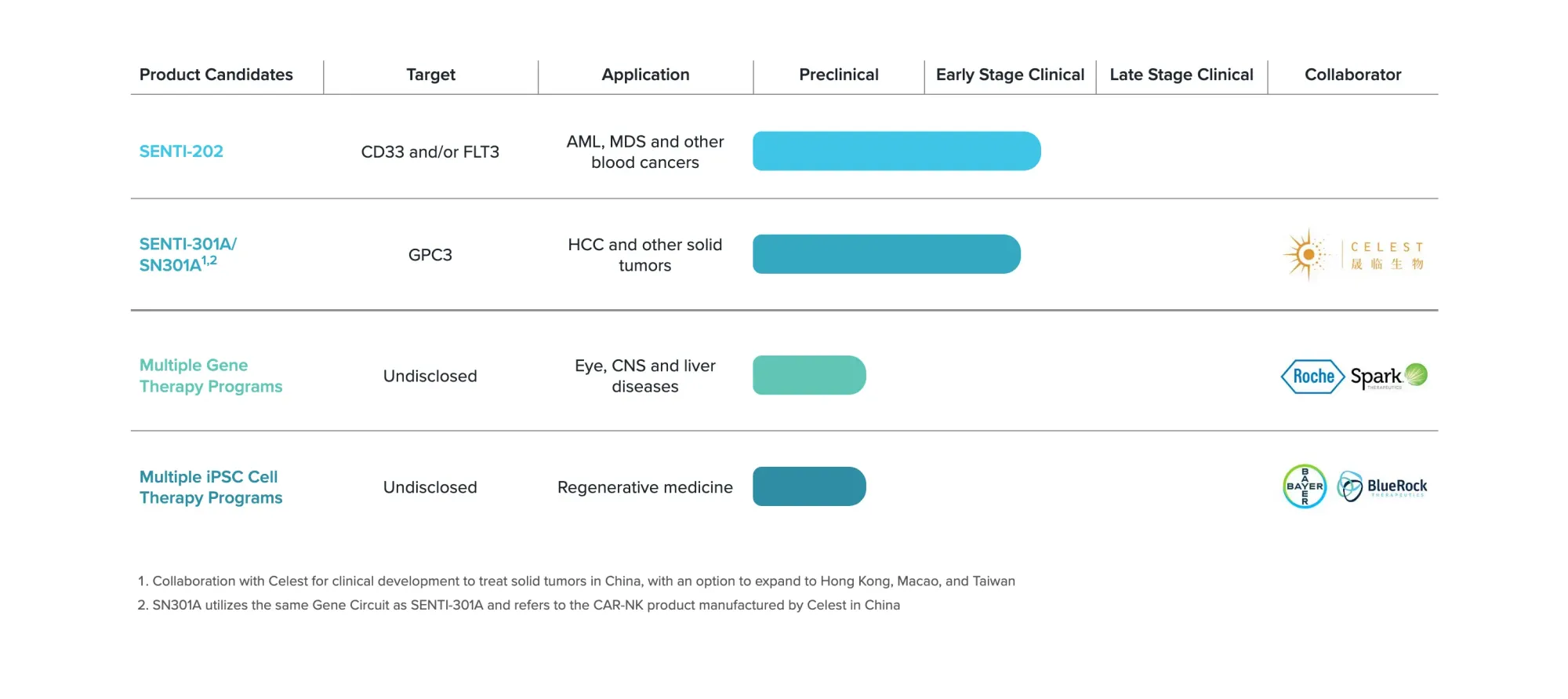
Senti Bio receives FDA's RMAT designation for SENTI-202, a breakthrough cell therapy showing promising results in treating relapsed acute myeloid leukemia patients.

Senti Bio receives FDA's RMAT designation for SENTI-202, a breakthrough cell therapy showing promising results in treating relapsed acute myeloid leukemia patients.

CVS Health raises 2025 revenue guidance to $400B and projects adjusted earnings of $7.00-$7.20 per share for 2026, signaling turnaround momentum under CEO David Joyner.

JPMorgan initiates coverage of Vor Bio with an Overweight rating and a $43 price target, highlighting strong confidence in telitacicept and major valuation upside.
Exicure shares positive Phase 2 data on burixafor (GPC-100) showing reliable and rapid stem cell mobilization for autologous transplant in multiple myeloma patients, even those with prior challenging therapies.

Terns Pharmaceuticals (TERN) announces encouraging Phase 1 results for TERN-701, showing 64% response rates in relapsed/refractory chronic myeloid leukemia patients with favorable safety profile.

Nurix Therapeutics presented positive Phase 1 data for its BTK degrader, bexobrutideg (NX-5948), showing high response rates and favorable safety in heavily pre-treated Waldenström Macroglobulinemia patients.