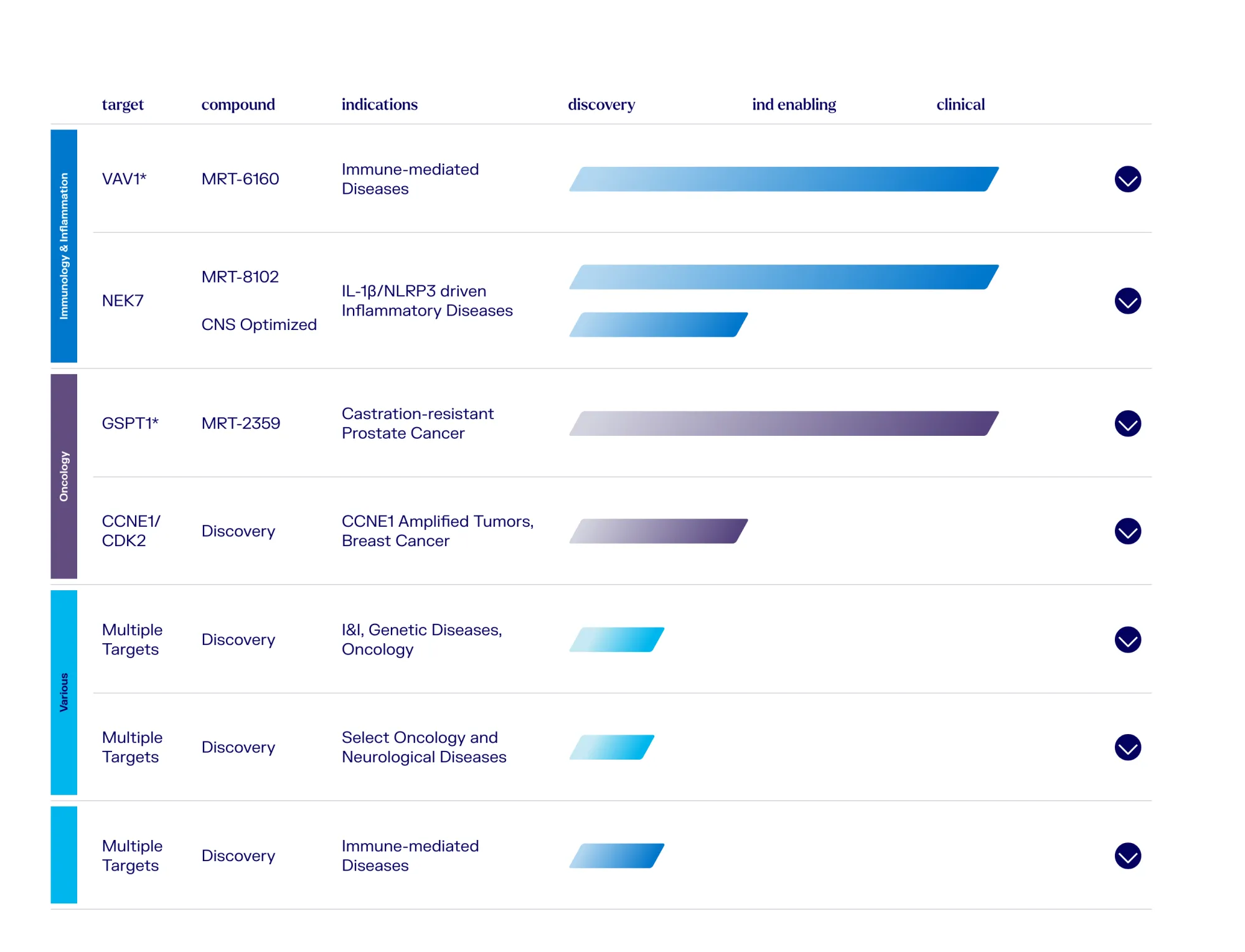
Wells Fargo Upgrades GLUE, Jefferies Downgrades GBIO
Wells Fargo upgrades Monte Rosa Therapeutics on 2026 catalysts, while Jefferies downgrades Generation Bio due to its pending acquisition by Xoma.

Wells Fargo upgrades Monte Rosa Therapeutics on 2026 catalysts, while Jefferies downgrades Generation Bio due to its pending acquisition by Xoma.

Aldeyra Therapeutics (ALDX) receives FDA extension for reproxalap dry eye drug review. New decision date set for March 2026 as agency requests additional trial documentation.
Organogenesis receives FDA clearance to file for approval of ReNu, a regenerative treatment for knee osteoarthritis pain affecting millions of Americans.

Vanda Pharmaceuticals files FDA application for imsidolimab, a promising new treatment for generalized pustular psoriasis, with potential approval by mid-2026.

Amphastar Pharmaceuticals receives FDA approval for generic teriparatide injection, offering an affordable alternative to Eli Lilly's FORTEO for osteoporosis patients.

Jefferies starts coverage of Sera Prognostics with a Buy rating, highlighting its PreTRM test for predicting preterm birth and $1.6B U.S. market potential.