
HHS Adds Rare Diseases to Newborn Tests, Sarepta Rises
Sarepta rises as HHS recommends newborn screening for Duchenne muscular dystrophy and metachromatic leukodystrophy, aiming for earlier diagnosis and treatment.

Sarepta rises as HHS recommends newborn screening for Duchenne muscular dystrophy and metachromatic leukodystrophy, aiming for earlier diagnosis and treatment.

Palvella Therapeutics (PVLA) receives FDA Fast Track designation for QTORIN rapamycin gel to treat angiokeratomas, a rare skin condition with no approved therapies affecting 50,000+ US patients.
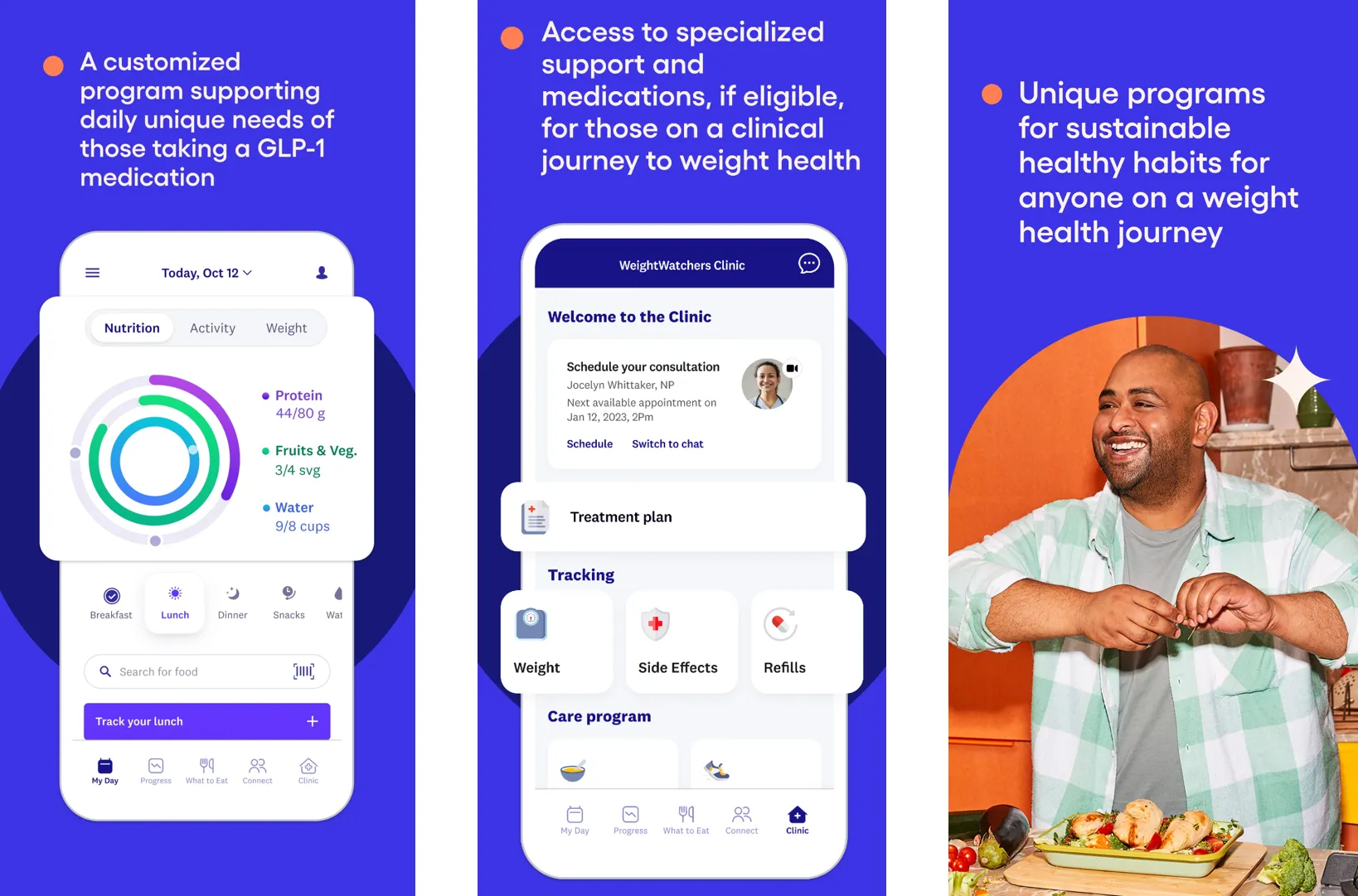
Weight Watchers unveils integrated platform combining GLP-1 medications, personalized nutrition, AI tracking, and community support for sustainable weight loss results.

Pfizer projects $59.5-62.5B revenue for 2026 with minimal growth as it invests in obesity drugs and cuts costs while COVID sales decline and key patents expire.

Nektar's (NKTR) rezpegaldesleukin demonstrates hair regrowth in alopecia areata patients but narrowly misses primary endpoint, presenting mixed results as company advances to Phase 3 trials.

Adagene receives FDA Fast Track designation for muzastotug, a novel cancer therapy targeting hard-to-treat colorectal cancer patients.