
Cantor Fitzgerald Starts Coverage on ArriVent Biopharma with Strong Upside
Cantor Fitzgerald initiates coverage on ArriVent Biopharma with an Overweight rating, highlighting strong Phase 3 potential and significant upside.

Cantor Fitzgerald initiates coverage on ArriVent Biopharma with an Overweight rating, highlighting strong Phase 3 potential and significant upside.

Novo Nordisk's Wegovy pill becomes the first FDA-approved oral GLP-1 weight-loss medication, launching January 2026 at $149/month as the Danish drugmaker aims to reclaim market share from Eli Lilly.

Amneal Pharmaceuticals and mAbxience announce FDA approval of Boncresa and Oziltus, biosimilar alternatives to Prolia and XGEVA for bone health treatment.
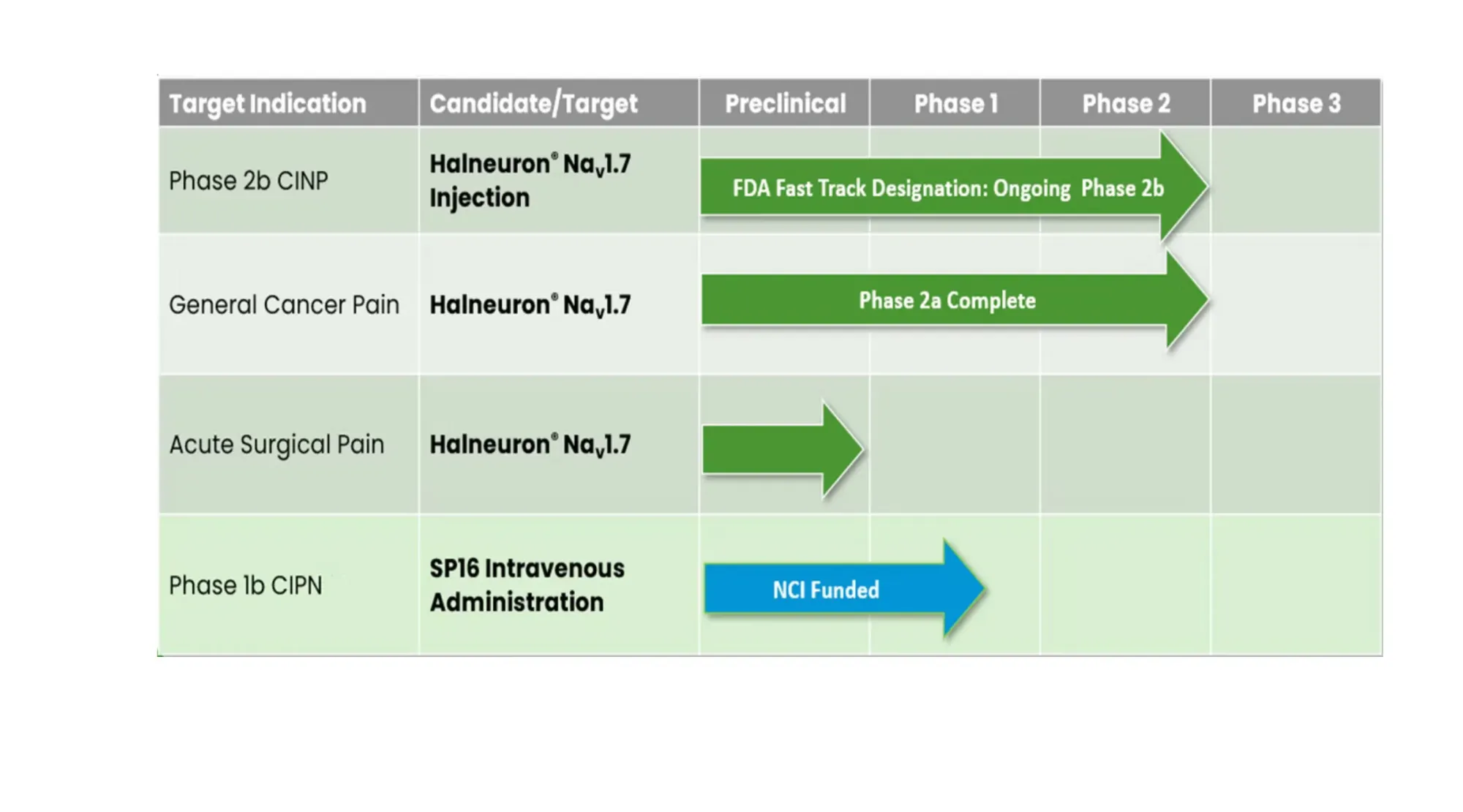
Dogwood Therapeutics' (DWTX) Phase 2b trial results reveal gaps, delayed timelines, and questions about their chemotherapy pain treatment Halneuron.
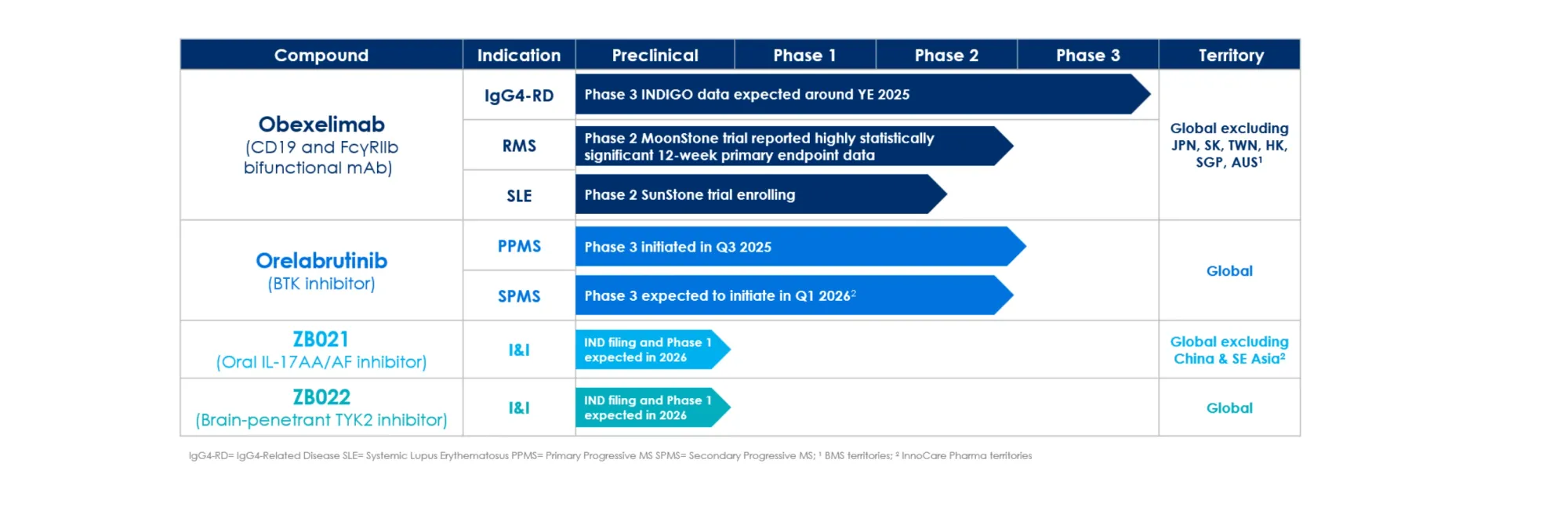
Citi, H.C. Wainwright, JonesResearch, and LifeSci Capital share new ratings and price targets on Zenas BioPharma, Spruce Biosciences, Context Therapeutics, and Ovid Therapeutics.

Wells Fargo initiates coverage of Arcellx with an Overweight rating and a $100 price target, citing strong potential for its CAR-T therapy anito-cel in multiple myeloma.