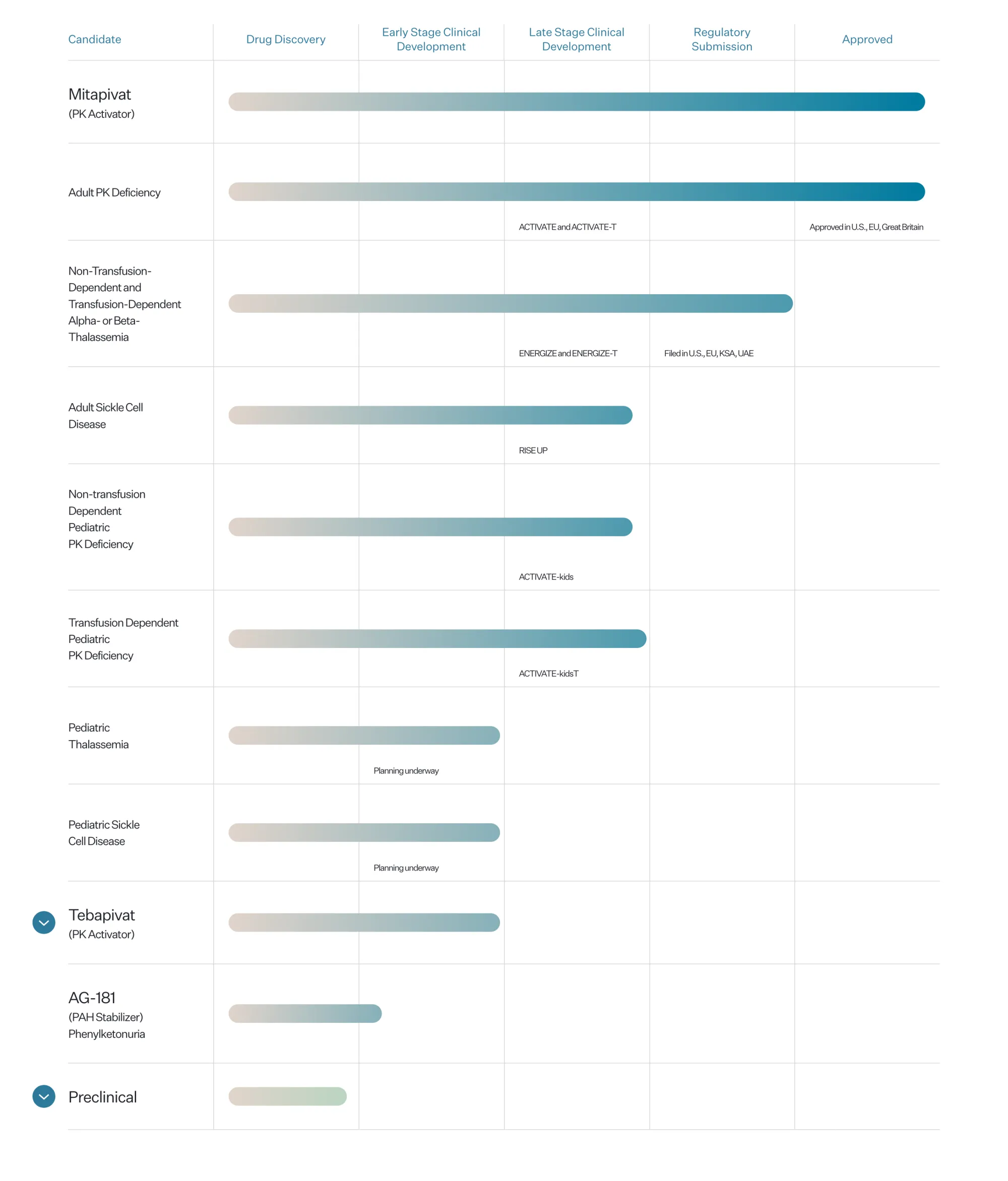
FDA Approves AQVESME: First Treatment for Thalassemia Anemia
The FDA has approved Agios’ AQVESME (mitapivat), the first medicine for treating anemia in adults with alpha or beta-thalassemia, offering hope for thousands of patients.

The FDA has approved Agios’ AQVESME (mitapivat), the first medicine for treating anemia in adults with alpha or beta-thalassemia, offering hope for thousands of patients.

Pfizer investigates death of hemophilia patient in Hympavzi trial after thrombotic stroke. Company says no expected safety impact for approved use.

FDA rejects Reviva's NDA plans for brilaroxazine, requiring additional Phase 3 trial. Launch delayed until at least 2026+, contingent on securing funding for the study.
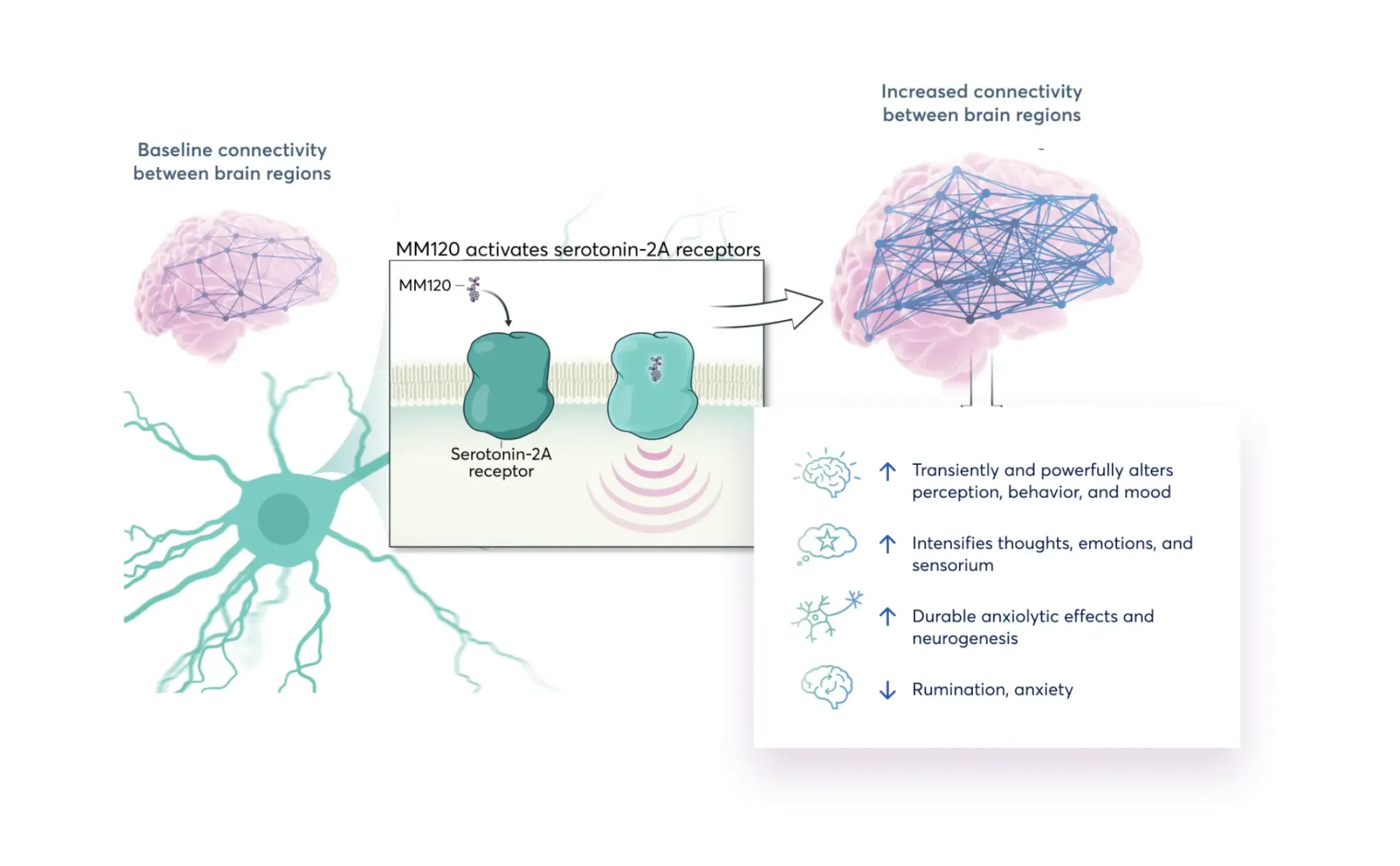
JonesResearch gives Buy ratings to Atai Beckley and Mind Medicine, highlighting potential in psychedelic drug development for depression and anxiety.
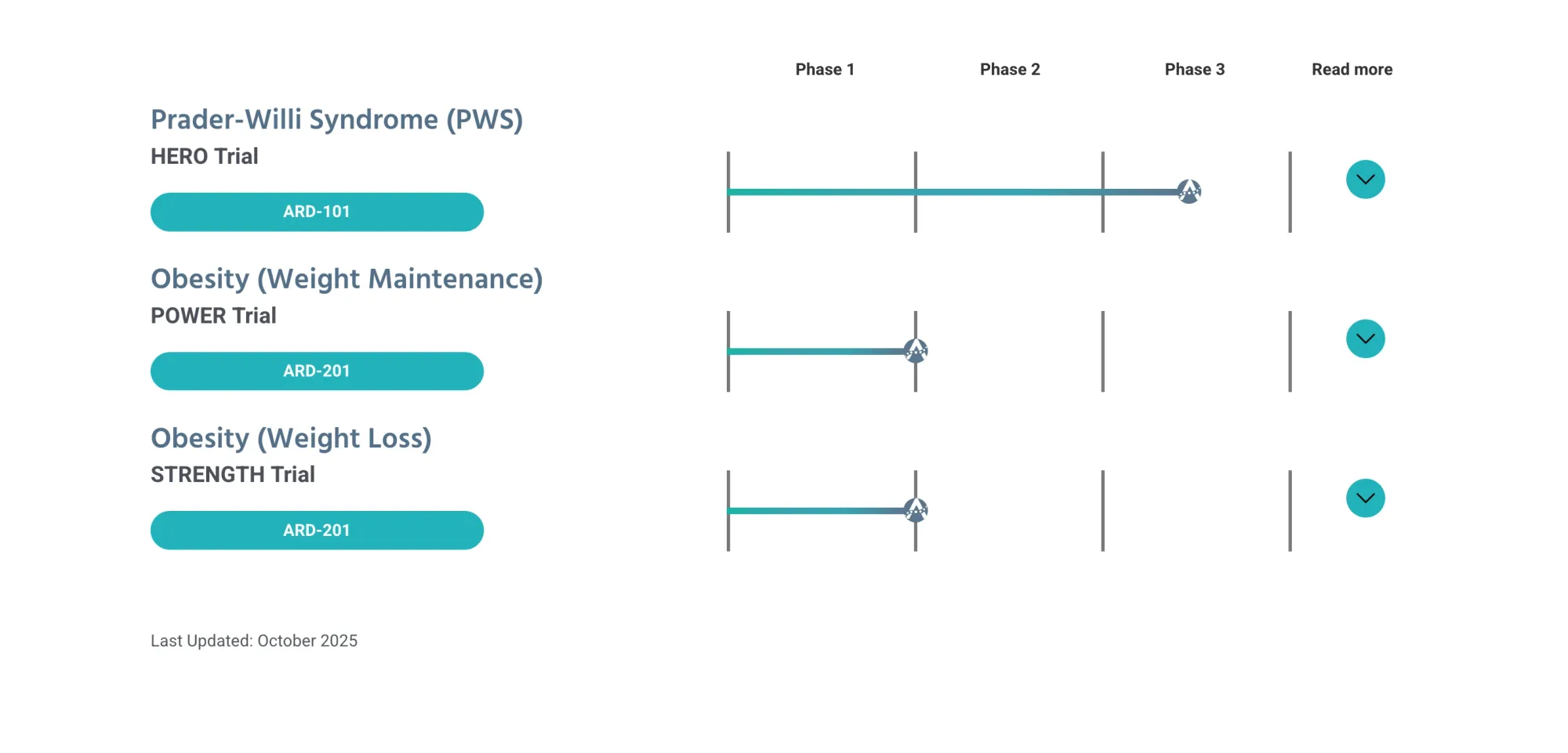
Oppenheimer analyst gives Aardvark Therapeutics an Outperform rating with $35 price target, highlighting rare and common obesity drug programs and a key Phase 3 trial.

Oppenheimer initiates coverage of Spruce Biosciences with an Outperform rating, citing strong FDA signals, accelerated approval potential, and possible acquisition interest from BioMarin.