
FDA Reaffirms Proteinuria Endpoint for FSGS: Positive Signal for Travere Therapeutics
FDA confirms proteinuria reduction as valid FSGS approval endpoint in Dimerix feedback, boosting confidence in Travere Therapeutics' upcoming Filspari decision.

FDA confirms proteinuria reduction as valid FSGS approval endpoint in Dimerix feedback, boosting confidence in Travere Therapeutics' upcoming Filspari decision.

Omeros' (OMER) Yartemlea becomes the first FDA-approved drug for TA-TMA, a life-threatening complication affecting stem cell transplant patients, with launch planned for January 2026.
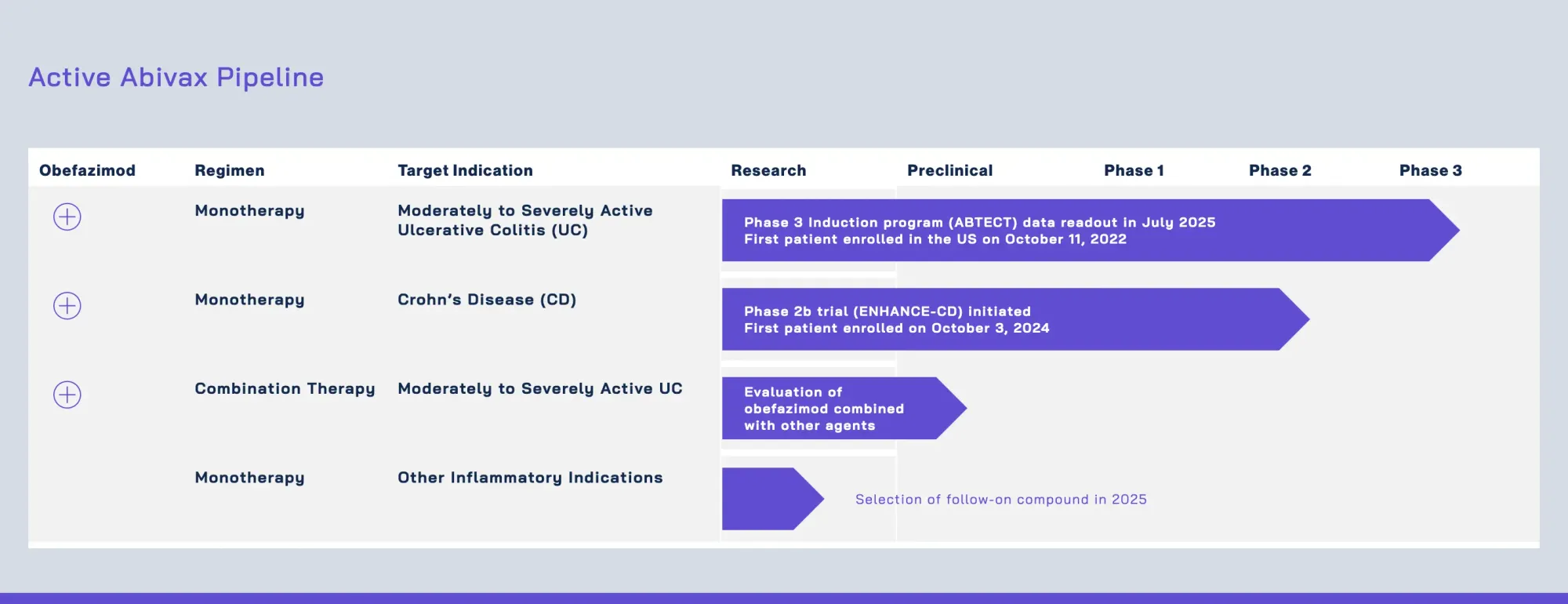
Pharma giant Eli Lilly considers acquiring French biotech Abivax for its promising ulcerative colitis treatment, expected to launch in 2027.
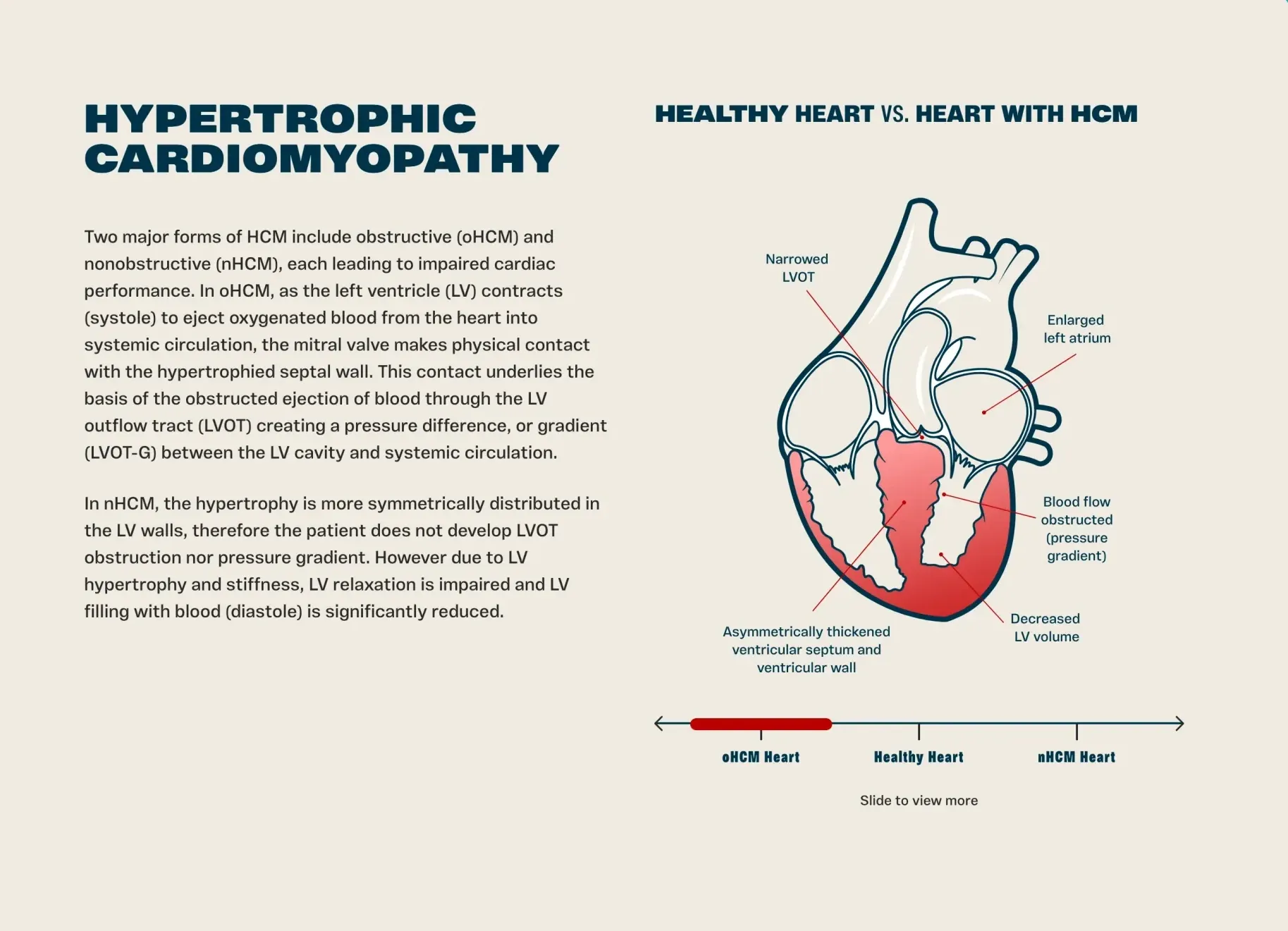
Edgewise Therapeutics (EWTX) shares positive safety data from EDG-7500 trial for hypertrophic cardiomyopathy, showing no heart function decline with comprehensive results expected in Q2 2026.

Gilead Sciences purchases RP-3467, a promising cancer treatment targeting BRCA mutations, from Repare Therapeutics in a deal worth up to $30 million.

French drugmaker Sanofi agrees to purchase U.S. vaccines company Dynavax Technologies for $2.2 billion, gaining access to hepatitis B vaccine and experimental shingles shot.