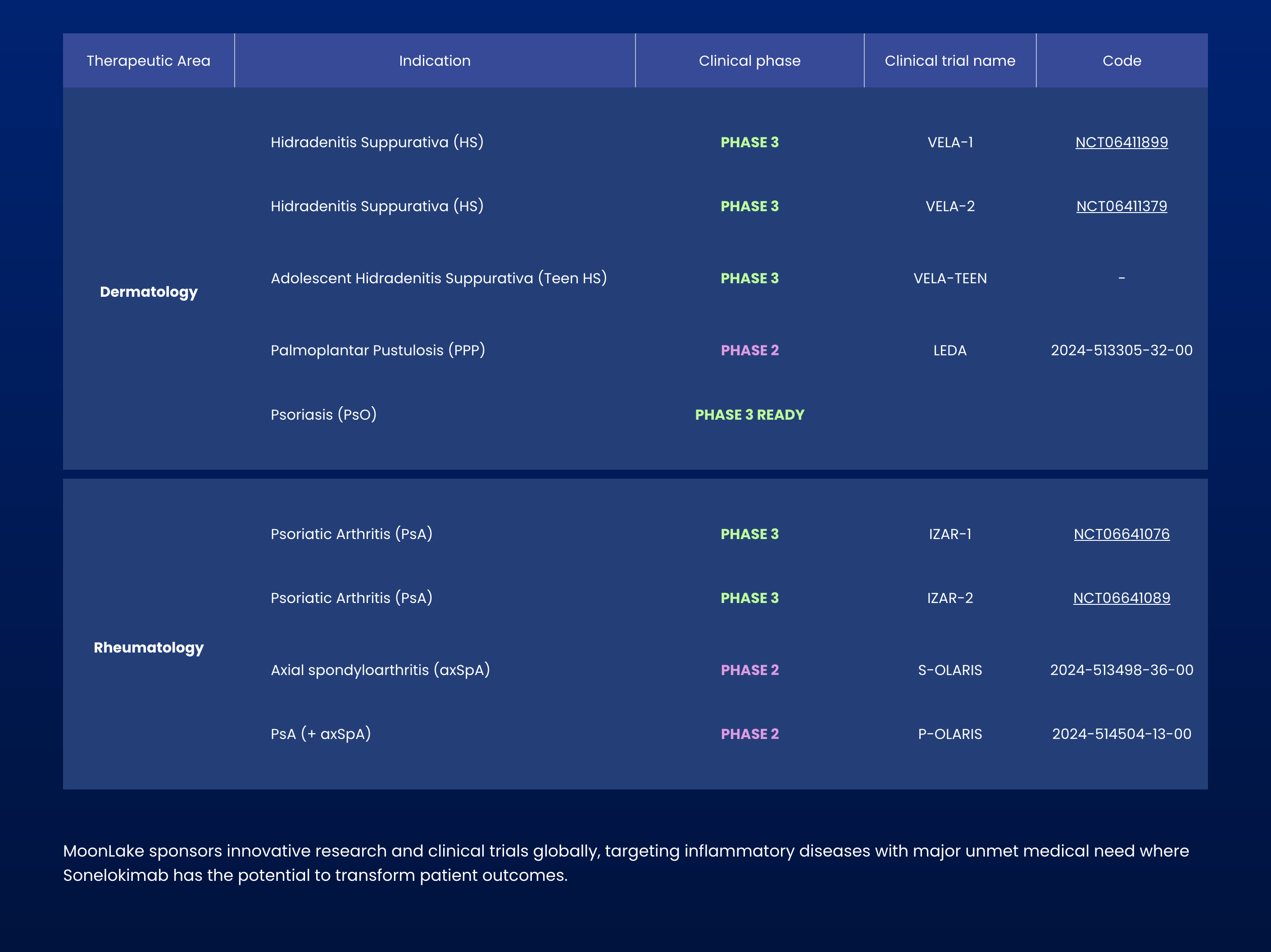
MoonLake Gets FDA Green Light for HS Treatment Approval Path
MoonLake Immunotherapeutics receives positive FDA feedback, clearing pathway to submit drug application for hidradenitis suppurativa treatment without additional trials needed.

MoonLake Immunotherapeutics receives positive FDA feedback, clearing pathway to submit drug application for hidradenitis suppurativa treatment without additional trials needed.

Vanda Pharmaceuticals stock falls in premarket trading after FDA denies Hetlioz approval for jet lag disorder, citing clinical trial design concerns despite positive efficacy data.
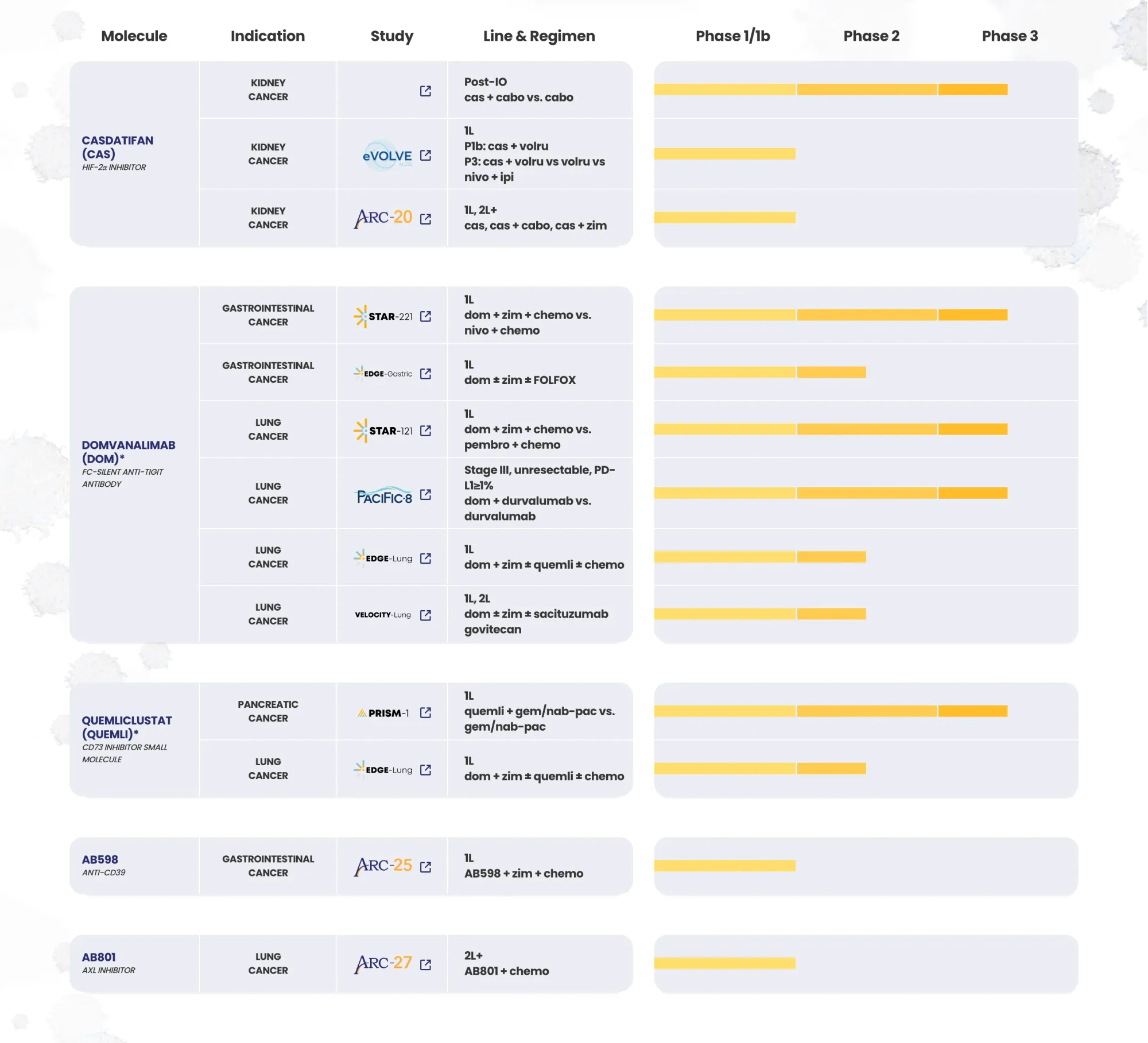
Morgan Stanley updates biotech views, downgrading Arcus and IO Biotech while upgrading Nurix on stronger pipeline prospects.
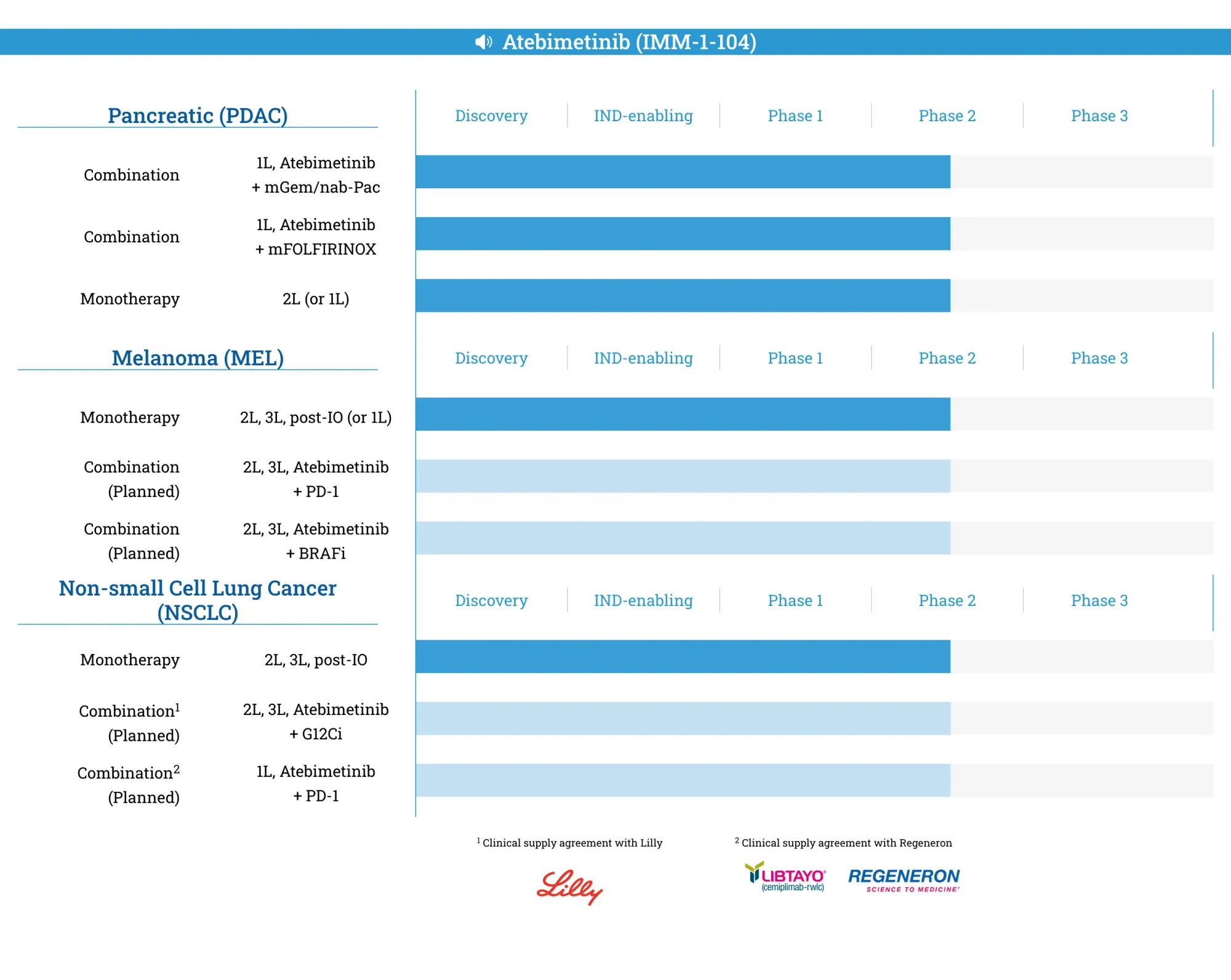
Immuneering (IMRX) reports 64% survival rate for Atebimetinib in pancreatic cancer trial, but shares plunge 23% as investors remain unconvinced about cross-trial comparisons and competitive threats.

Globus Medical (GMED) shares jump 9.4% in premarket trading after reporting preliminary Q4 sales of $823.2M, beating analyst estimates, with strong 2026 guidance signaling continued growth momentum.

AbbVie denies reports of buying Revolution Medicines after Wall Street Journal claimed advanced talks.