
PDS Biotech Submits Protocol Amendment for Faster Cancer Drug Approval
PDS Biotechnology updates Phase 3 trial protocol with FDA to potentially speed up approval of PDS0101 for HPV-related head and neck cancer treatment.

PDS Biotechnology updates Phase 3 trial protocol with FDA to potentially speed up approval of PDS0101 for HPV-related head and neck cancer treatment.

Amazon Pharmacy now sells Wegovy weight-loss pill. Insured pay $25/month, uninsured start at $149. Needle-free option available nationwide.

CG Oncology announces earlier-than-expected results for its Phase 3 bladder cancer trial, moving timeline up by nearly one year due to rapid patient enrollment.

uniQure secures FDA meeting to discuss accelerated approval for AMT-130, bringing hope to the Huntington's disease community with potentially life-changing gene therapy.

Aquestive Therapeutics stock plunges after FDA identifies deficiencies in Anaphylm application, a sublingual film treatment for severe allergic reactions.
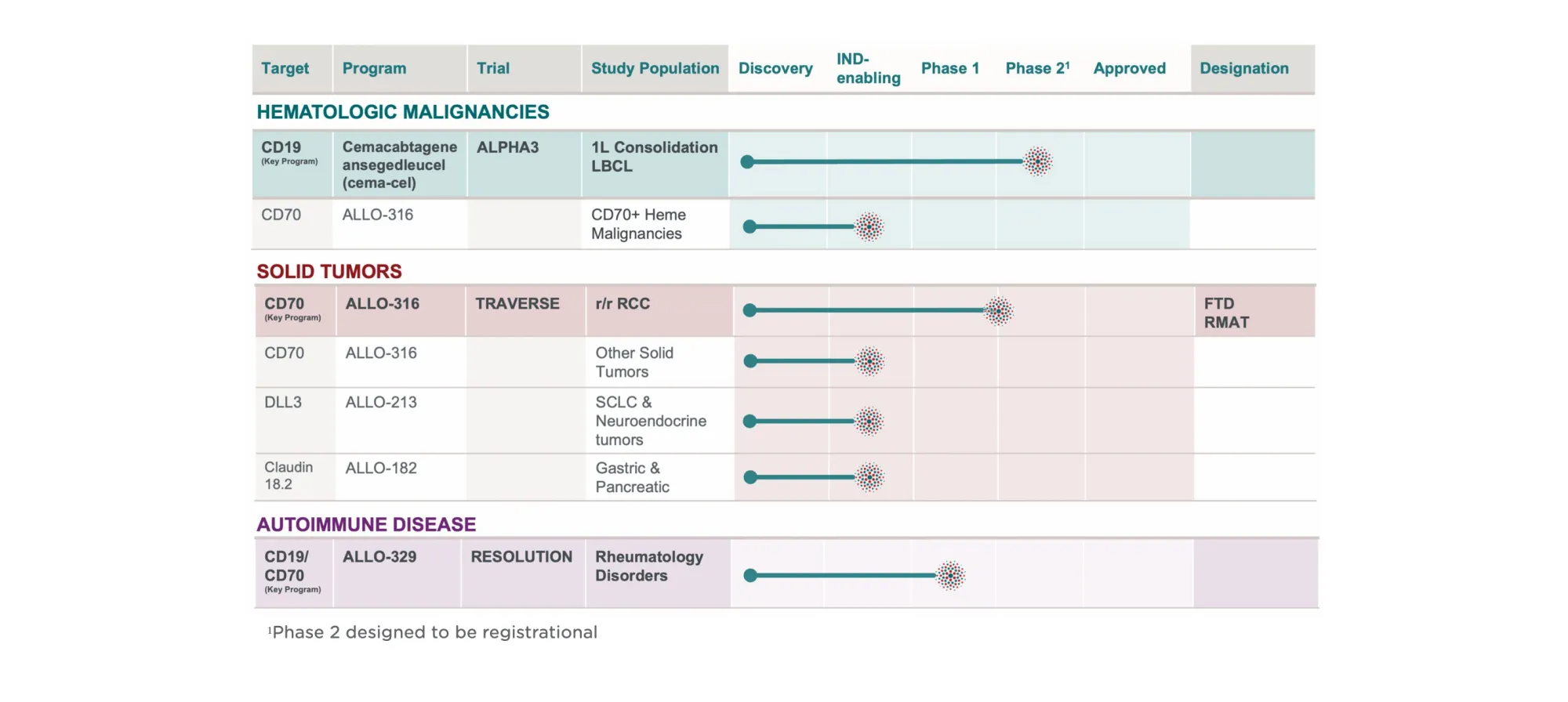
Citizens upgraded Allogene Therapeutics to Outperform with a $5 target, citing improved trial visibility and strong long-term sales potential for cema-cel.