
FDA Rejects Atara's Cancer Treatment Application
Atara Biotherapeutics (ATRA) receives second FDA rejection for EBVALLO cancer therapy despite resolving manufacturing issues, citing concerns about clinical trial design.

Atara Biotherapeutics (ATRA) receives second FDA rejection for EBVALLO cancer therapy despite resolving manufacturing issues, citing concerns about clinical trial design.

Lyra Therapeutics suspends LYR-210 development for chronic rhinosinusitis, lays off 28 employees, and explores strategic alternatives as stock plummets 44% in premarket trading.

Goldman Sachs and JPMorgan highlight new clinical data that strengthen the outlook for Crinetics and Fulcrum Therapeutics, citing clearer paths to success and upside potential.
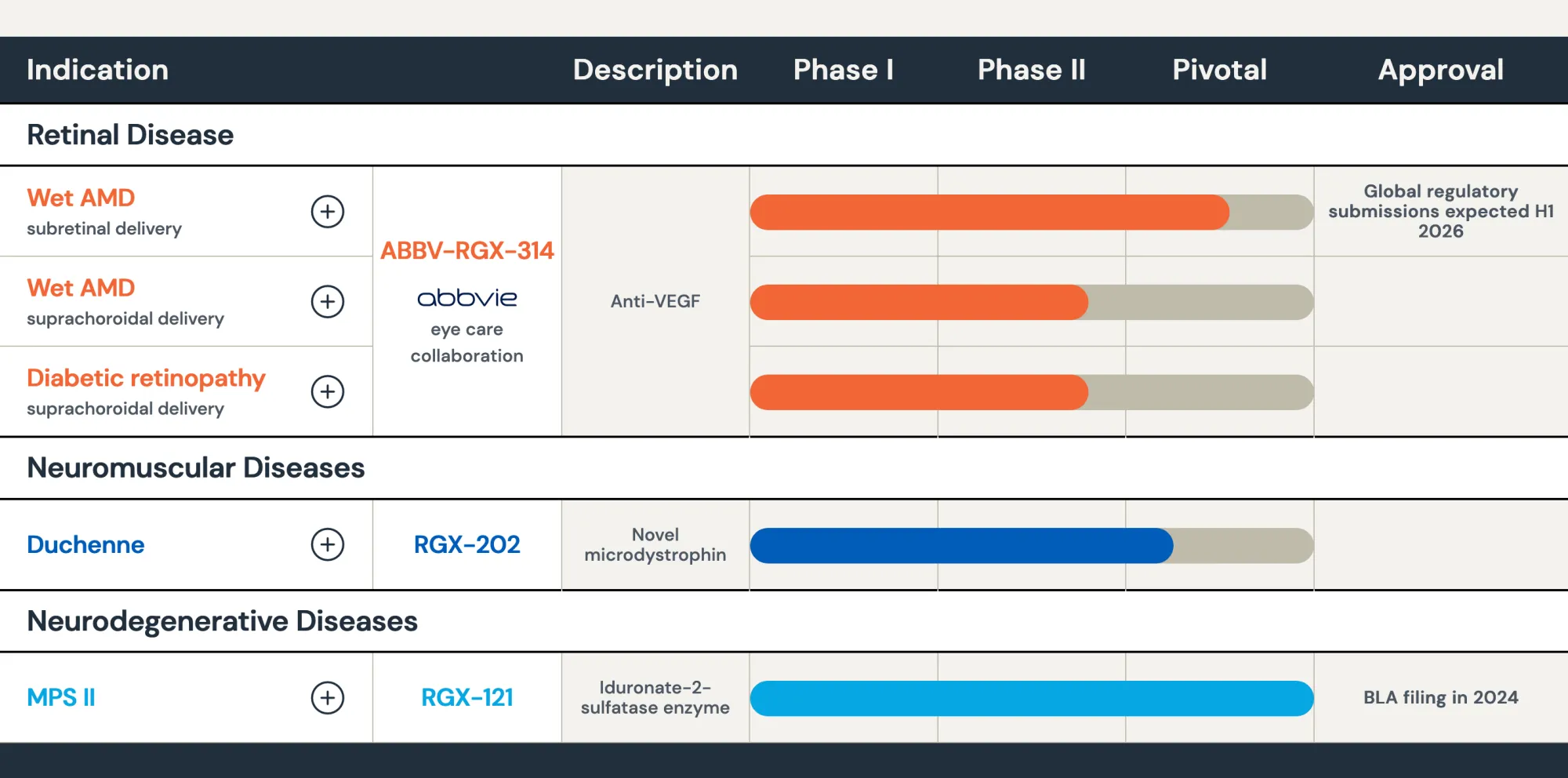
REGENXBIO stock rises 8% after positive 18-month trial data for RGX-202, its one-time gene therapy treating Duchenne muscular dystrophy in boys.
Tempus AI achieves record $1.1 billion in total contract value, partners with 70+ pharmaceutical companies, and reports 31% revenue growth in 2025.

Stoke Therapeutics continues discussions with FDA for expedited approval of zorevunersen for Dravet syndrome after regulators request more information on the epilepsy treatment.