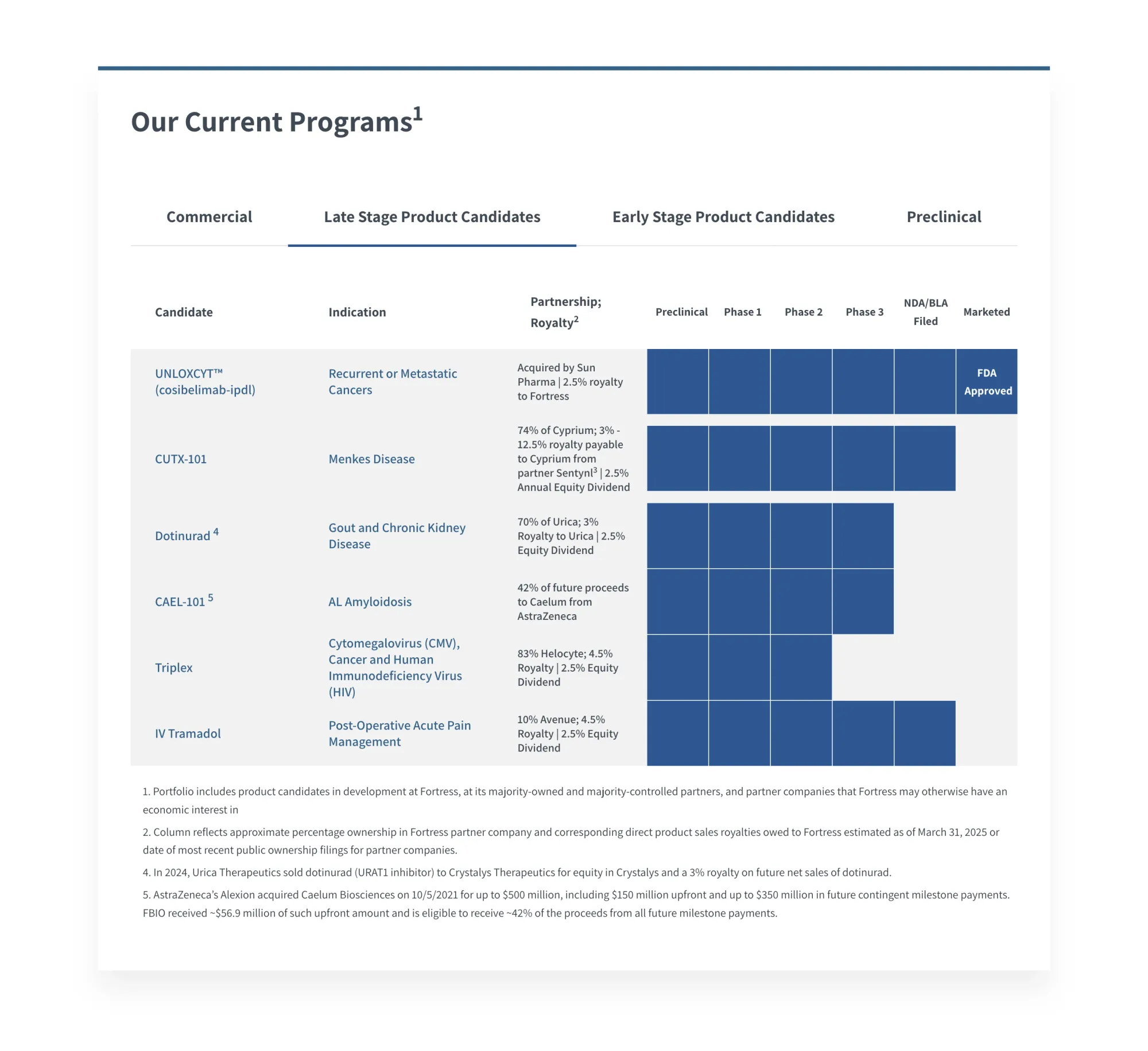
FDA Approves First Treatment for Rare Menkes Disease
ZYCUBO becomes the first FDA-approved treatment for Menkes disease, a rare genetic disorder affecting copper transport in children, offering new hope for patients.

ZYCUBO becomes the first FDA-approved treatment for Menkes disease, a rare genetic disorder affecting copper transport in children, offering new hope for patients.

Goldman Sachs upgraded Arcus Biosciences to Buy, raising its price target to $28 on promising results for its lead cancer drug Casdatifan.
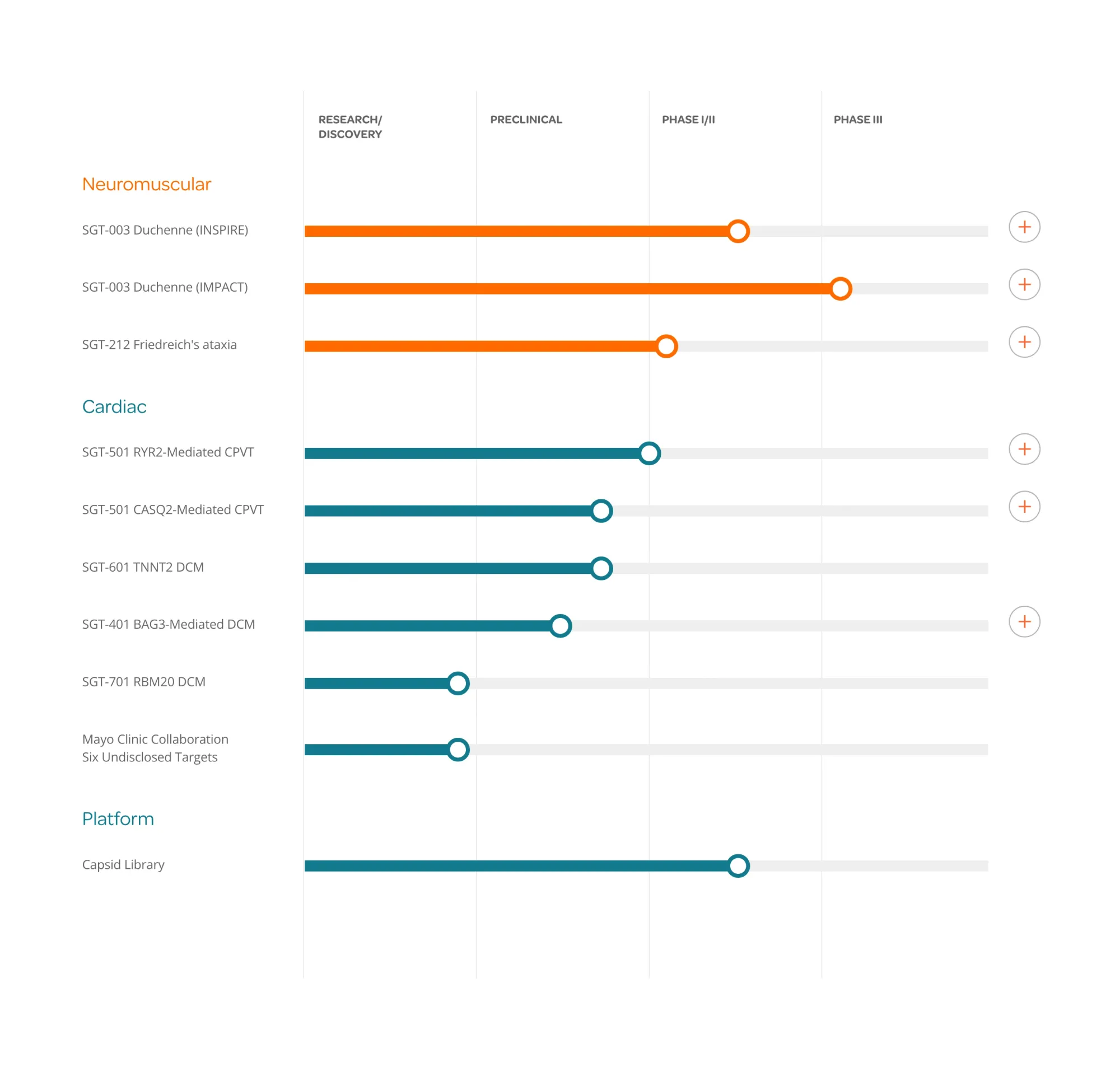
Solid Biosciences receives FDA Orphan Drug designation for SGT-212, a dual-route gene therapy treating Friedreich's ataxia, with first patient dosed in clinical trial.
Nuvation Bio and Sarepta shares tumble on disappointing preliminary results while Adaptive Biotechnologies surges on strong revenue beat.

William Blair maintains Outperform rating on Beam Therapeutics after company announces FDA alignment for BEAM-302 and planned risto-cel application, citing clear regulatory pathway ahead.

Abivax shares surge 4% on reports of Eli Lilly's €15B buyout interest. The French biotech's experimental ulcerative colitis drug obefazimod attracts big pharma attention after trial success.