
Oncolytics Biotech Wins FDA Fast Track for Colorectal Cancer Treatment
Oncolytics Biotech receives FDA Fast Track designation for pelareorep in treating KRAS-mutant colorectal cancer, showing promising survival improvements.

Oncolytics Biotech receives FDA Fast Track designation for pelareorep in treating KRAS-mutant colorectal cancer, showing promising survival improvements.
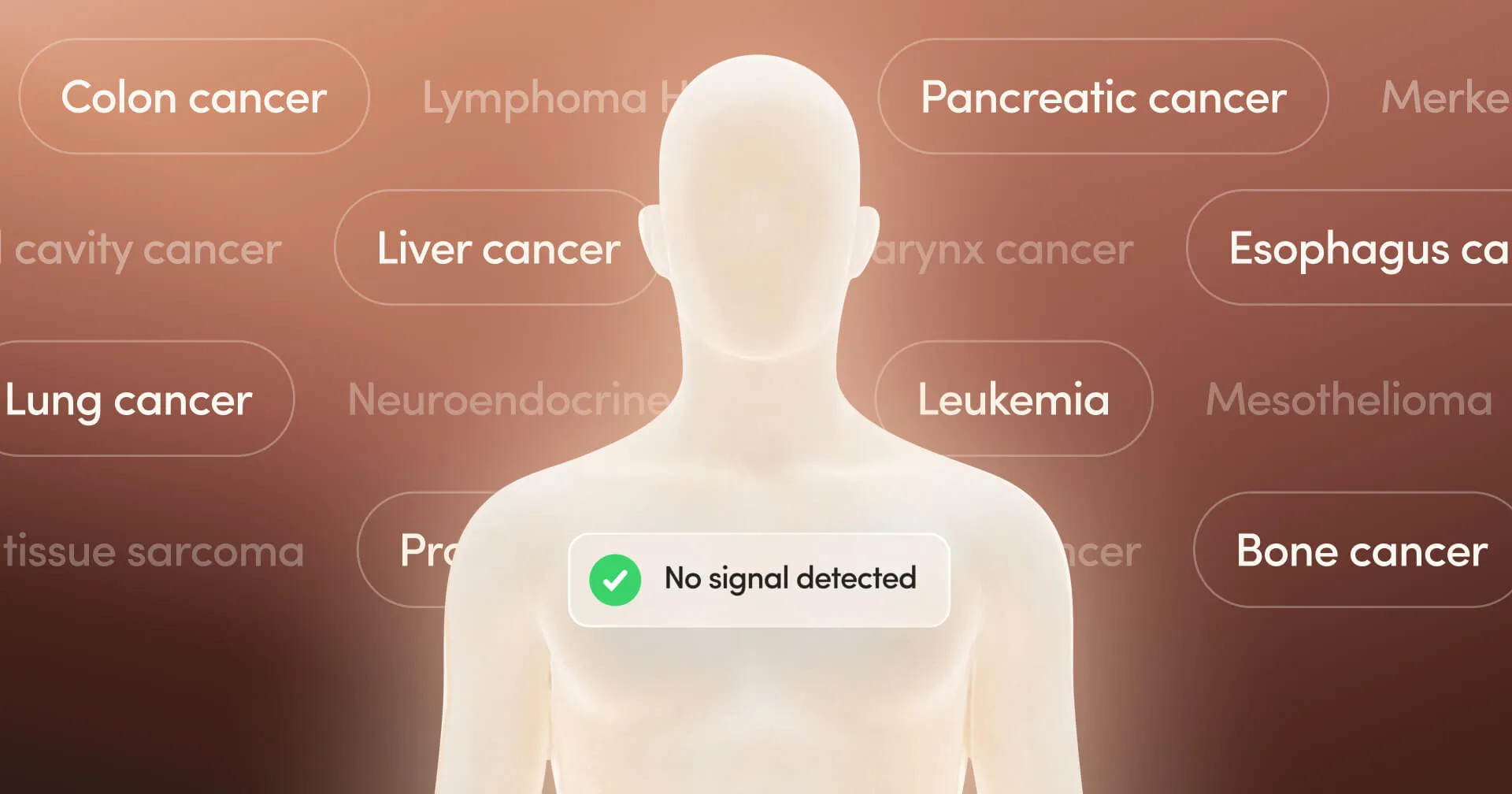
Hims & Hers partners with Grail to offer discounted Galleri cancer detection tests through its Labs subscription service, expanding beyond weight-loss treatments.

Boston Scientific (BSX) reports strong Q4 2025 earnings but shares fall over 10% on modest 2026 guidance. Revenue growth expected to slow to 10-11% from 15.8% in 2026.
Eli Lilly (LLY) beats Q4 expectations and projects strong 2026 growth driven by surging demand for Zepbound and Mounjaro, outpacing rival Novo Nordisk.

Novo Nordisk forecasts 5-13% sales decline for 2026 as Ozempic and Wegovy face intense competition from Eli Lilly and generics, plus US drug pricing pressures.
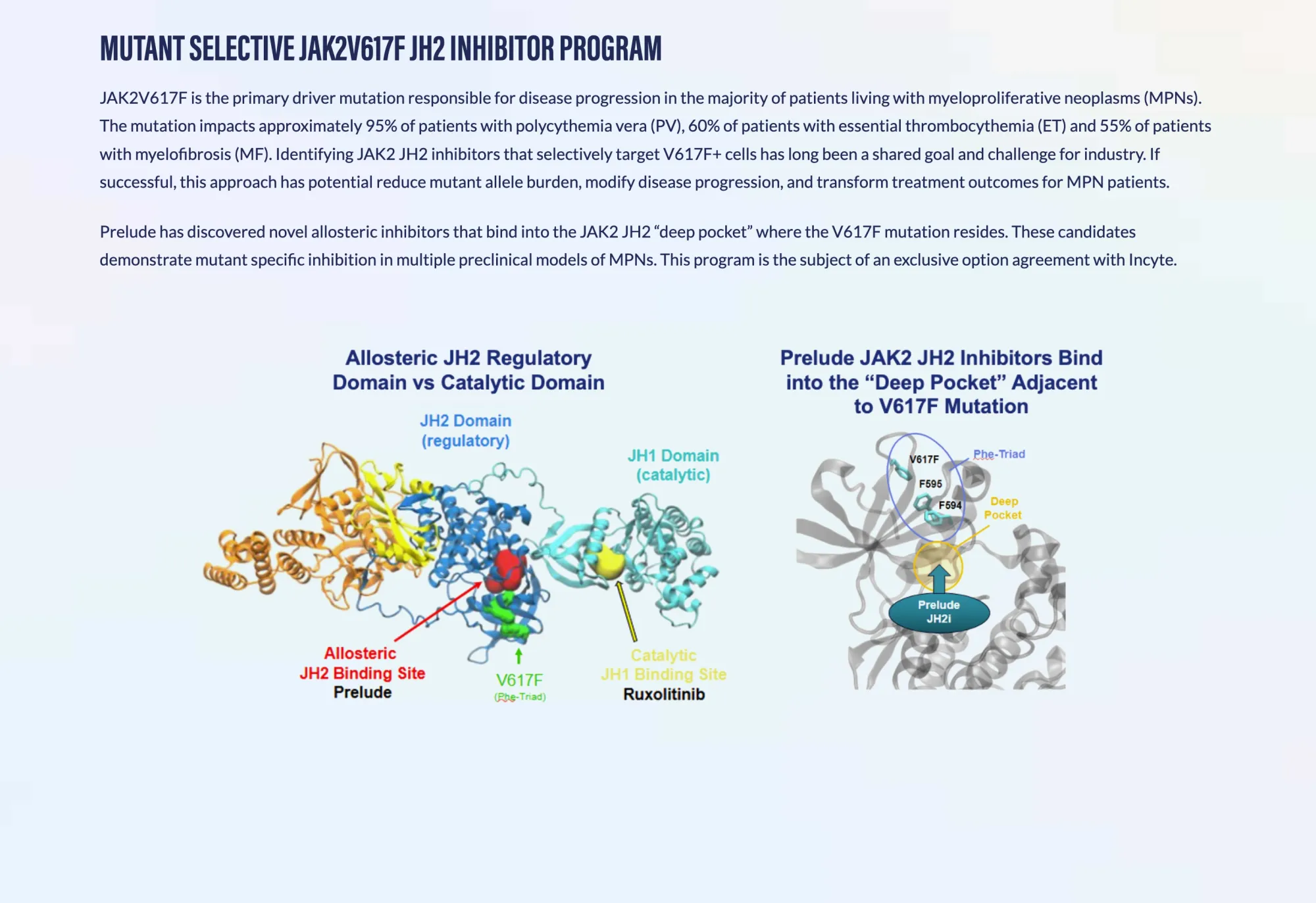
Prelude Therapeutics receives FDA clearance to begin testing PRT12396, a targeted therapy for myeloproliferative neoplasms affecting thousands of patients.