
Morgan Stanley Sees Downside to 2026-27 Consensus for Novo Amid Competition and Patent Risks
Novo Nordisk was downgraded because key drug sales (Ozempic/Wegovy) are slowing. Competition and future patent loss are hurting the stock.

Novo Nordisk was downgraded because key drug sales (Ozempic/Wegovy) are slowing. Competition and future patent loss are hurting the stock.
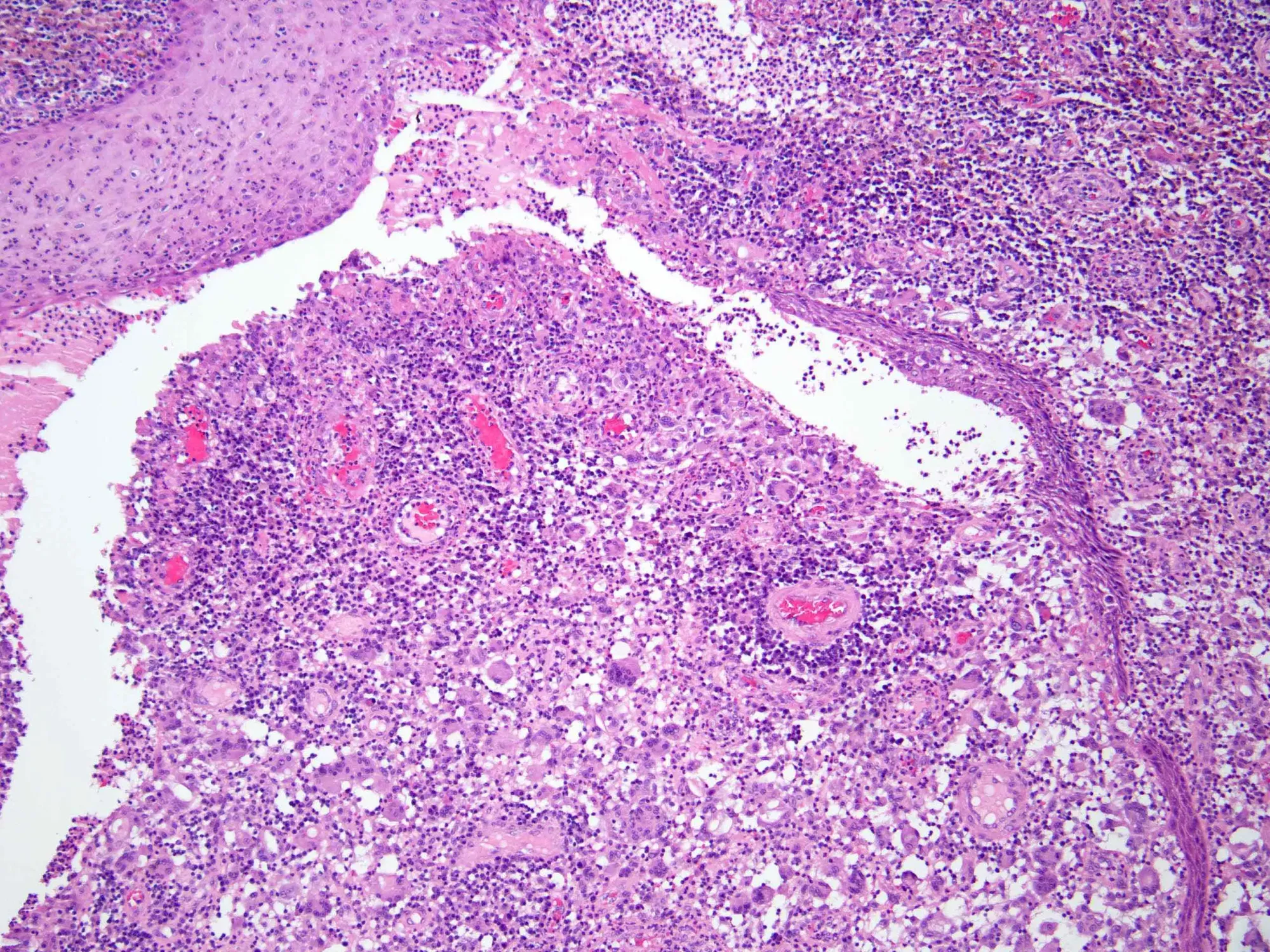
MoonLake announced Phase 3 results for sonelokimab (VELA trials) in HS. While one trial succeeded using the primary analysis, the other faced challenges due to a high placebo response, leading to a stock downgrade.

Crinetics' stock surged after the FDA approved Palsonify, their once-daily oral drug for acromegaly. This rare disorder results in too much growth hormone. Palsonify is the first oral option, offering rapid, consistent control and reduced symptoms. It will be available in early October.

Leerink Partners dramatically upgraded Rapt Therapeutics stock based on the strong outlook for RPT904, a leading drug candidate designed to treat severe allergies, including food allergy, by improving on the current standard treatment, Xolair.
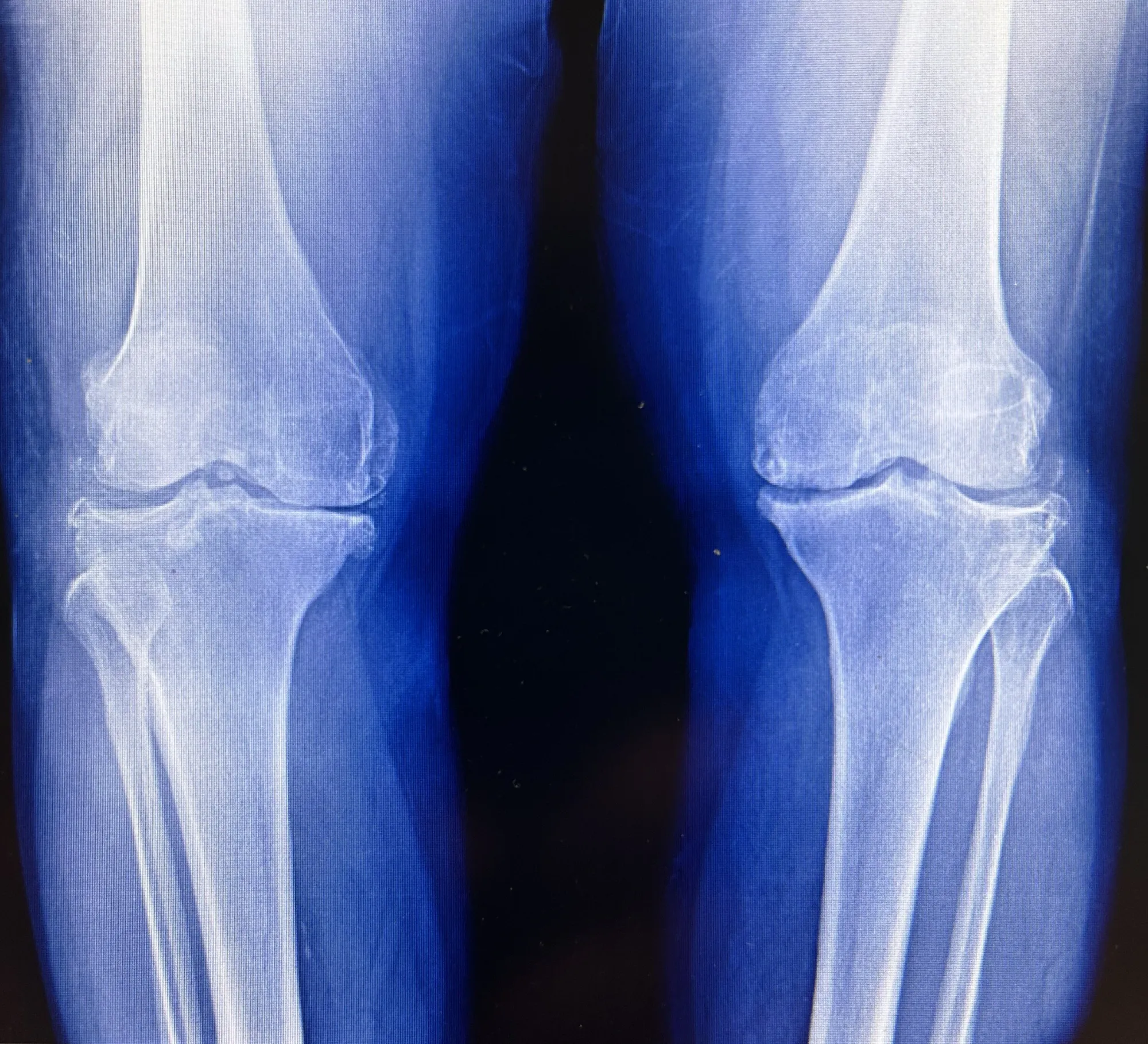
ReNu's second trial for knee pain (OA) missed its main goal, but still showed numerical improvement and function benefits. Organogenesis, confident in the data from all three large studies (1,300+ patients), will ask the FDA to review combined results for approval.

Goldman Sachs cut Apellis stock to "Sell," lowering the price target to $18. Risks include slow near-term growth and lower Syfovre sales.