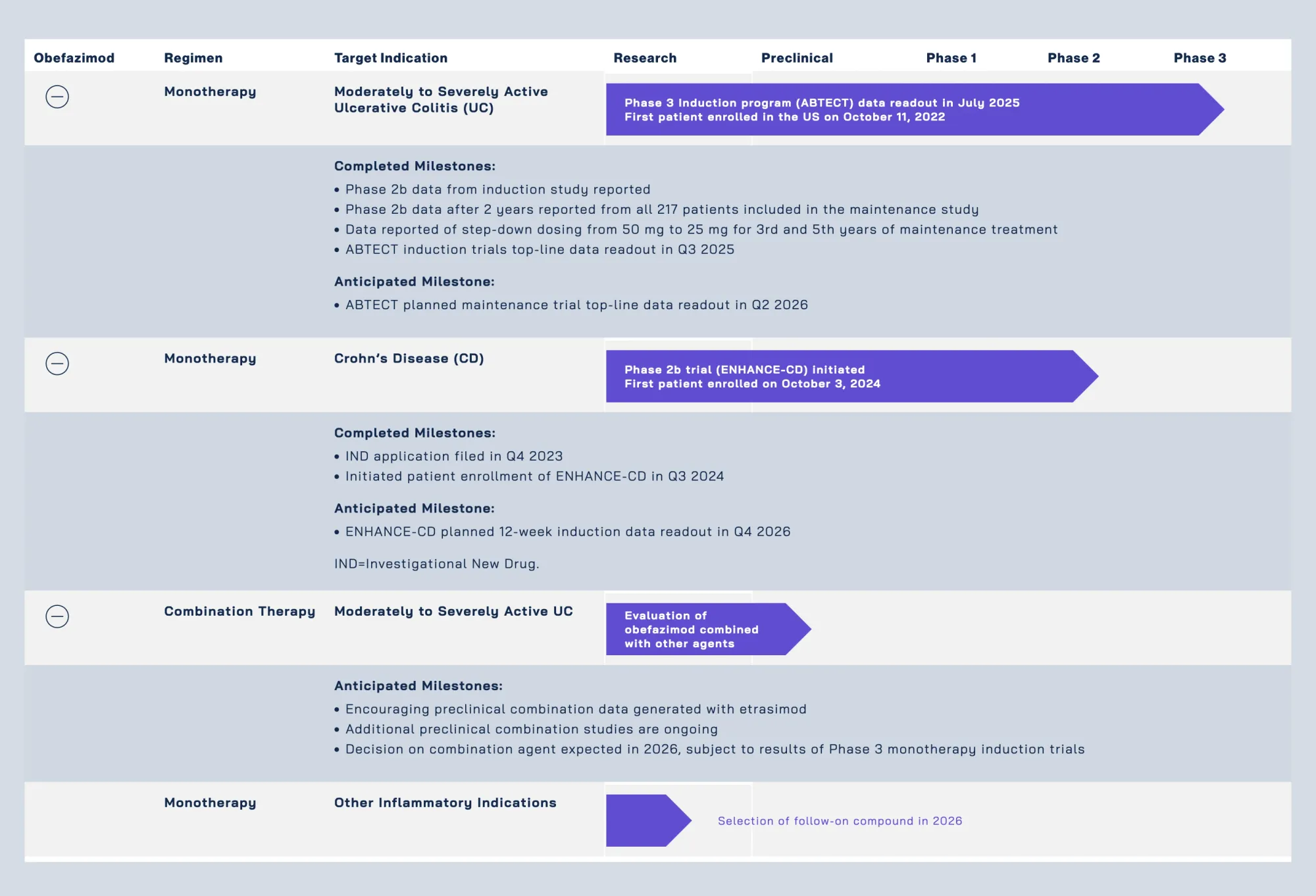
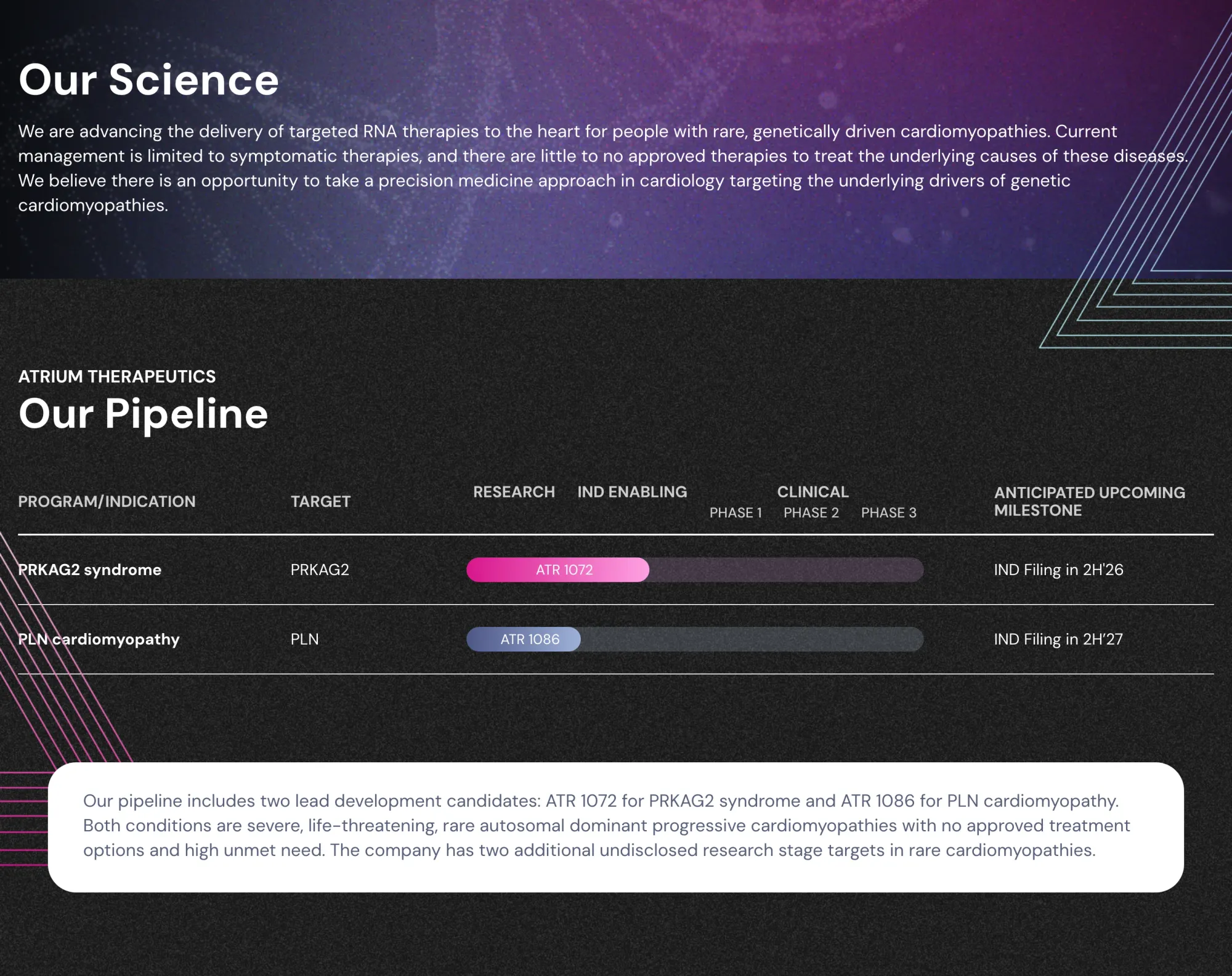
Natera Launches Zenith Genomics as GeneDx Plunges in Rare Disease Testing Showdown
Natera launches Zenith Genomics — its next-generation whole-genome sequencing test for faster rare disease diagnosis. GeneDx shares drop 8% as competition intensifies in the rare-disease market, which carries a $1 trillion economic burden.