FDA Grants Breakthrough Status to Alkermes New Narcolepsy Drug
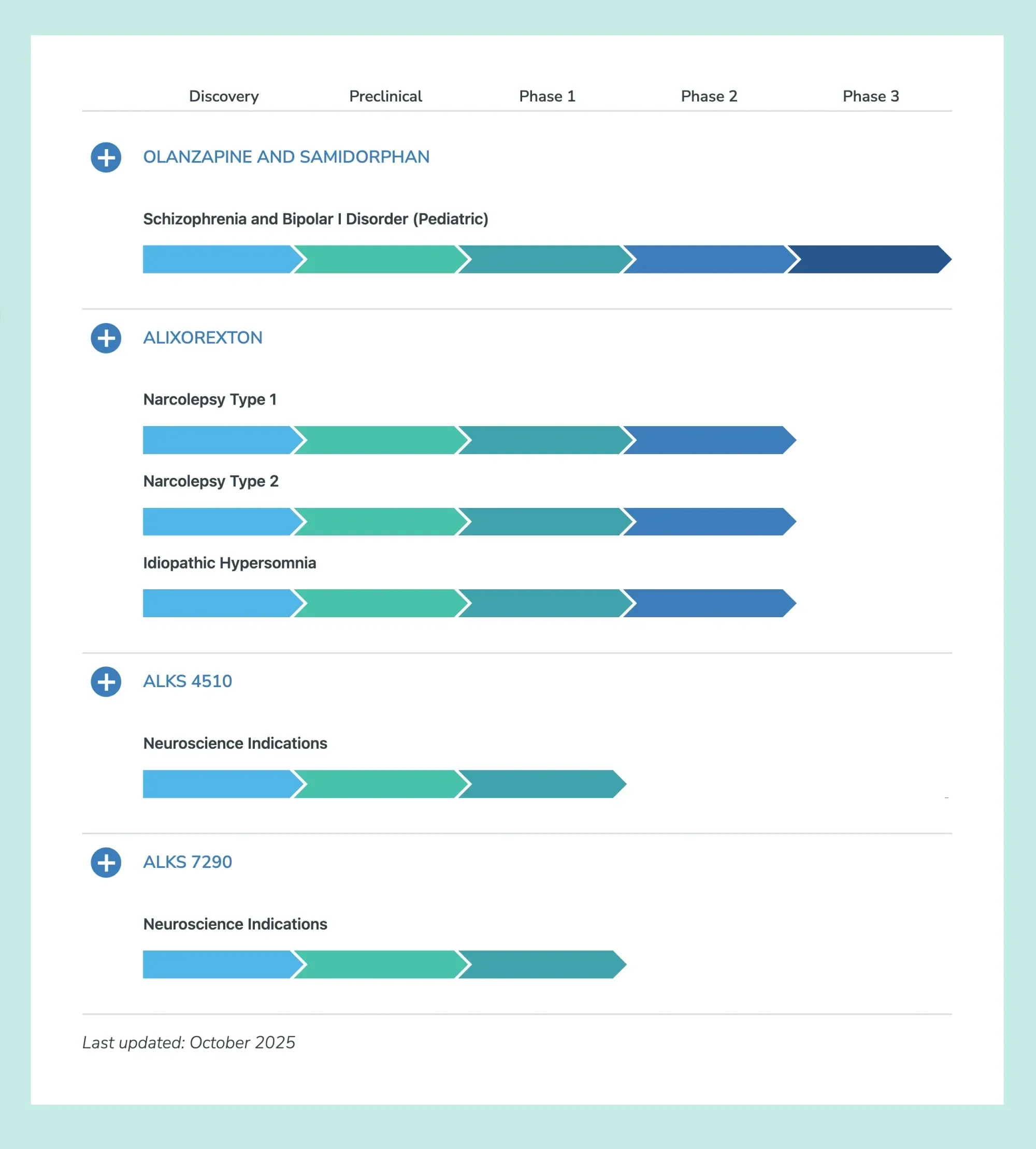
FDA grants Breakthrough Therapy status to alixorexton for narcolepsy type 1. Clinical trials show promising results for daytime wakefulness.

FDA grants Breakthrough Therapy status to alixorexton for narcolepsy type 1. Clinical trials show promising results for daytime wakefulness.

Avadel Pharmaceuticals receives unsolicited $23/share offer from Lundbeck, competing with existing $20/share Alkermes deal. Board reviewing superior proposal.

Truist comments on Alkermes’ Phase 2 Vibrance-2 trial of alixorexton in narcolepsy type 2, noting "limited" details but strong patient engagement and maintaining a Buy rating with a $50 target.

Centessa has great sleep drug data, easing daytime sleepiness. Alkermes stock jumped 7% as its competing drug is still strong.

Alkermes (ALKS) announces $2.1B acquisition of Avadel Pharmaceuticals (AVDL) to gain approved narcolepsy drug Lumryz and enter the growing sleep medicine market. Deal expected Q1 2026.

Alixorexton, a new drug for narcolepsy type 1, shows great promise. It helps patients stay awake longer (28 mins, considered normal), reduces sleepiness, cataplexy, fatigue, and improves focus. It's well-tolerated and moving to Phase 3, signaling a potential new era for treatment.