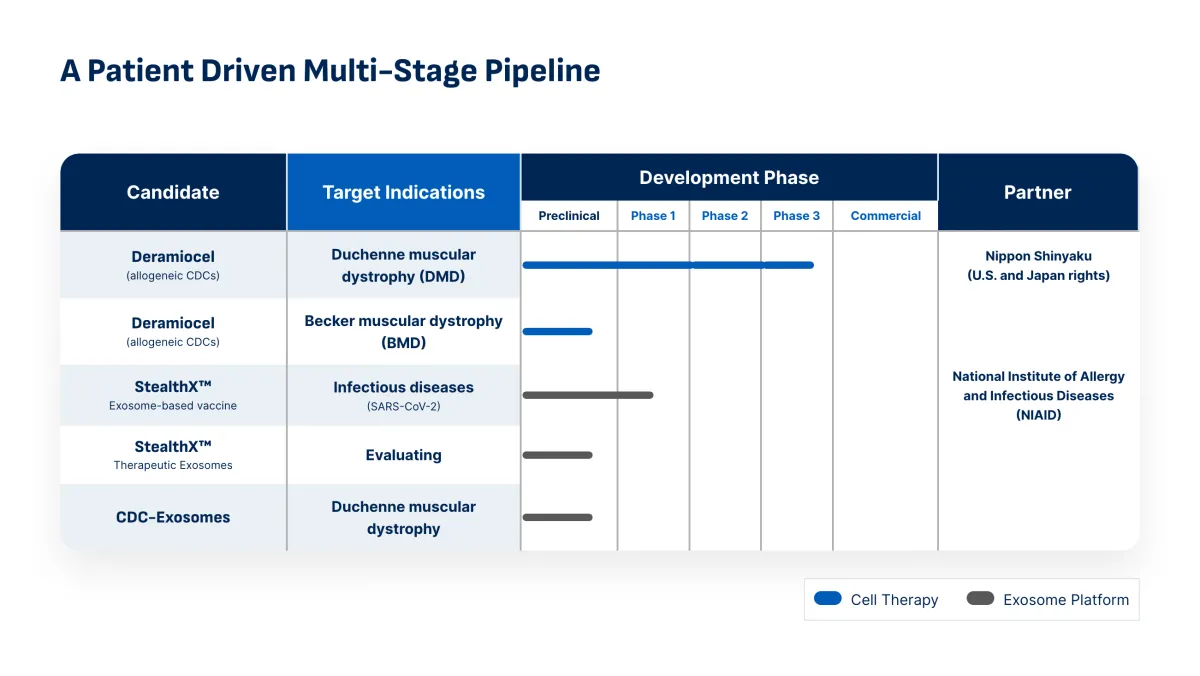
FDA Lifts Rejection and Schedules August 2026 Review for Deramiocel
Capricor Therapeutics receives new PDUFA target date of August 22, 2026 for Deramiocel BLA. Potential first therapy to treat both heart and skeletal muscle damage in Duchenne muscular dystrophy (DMD).