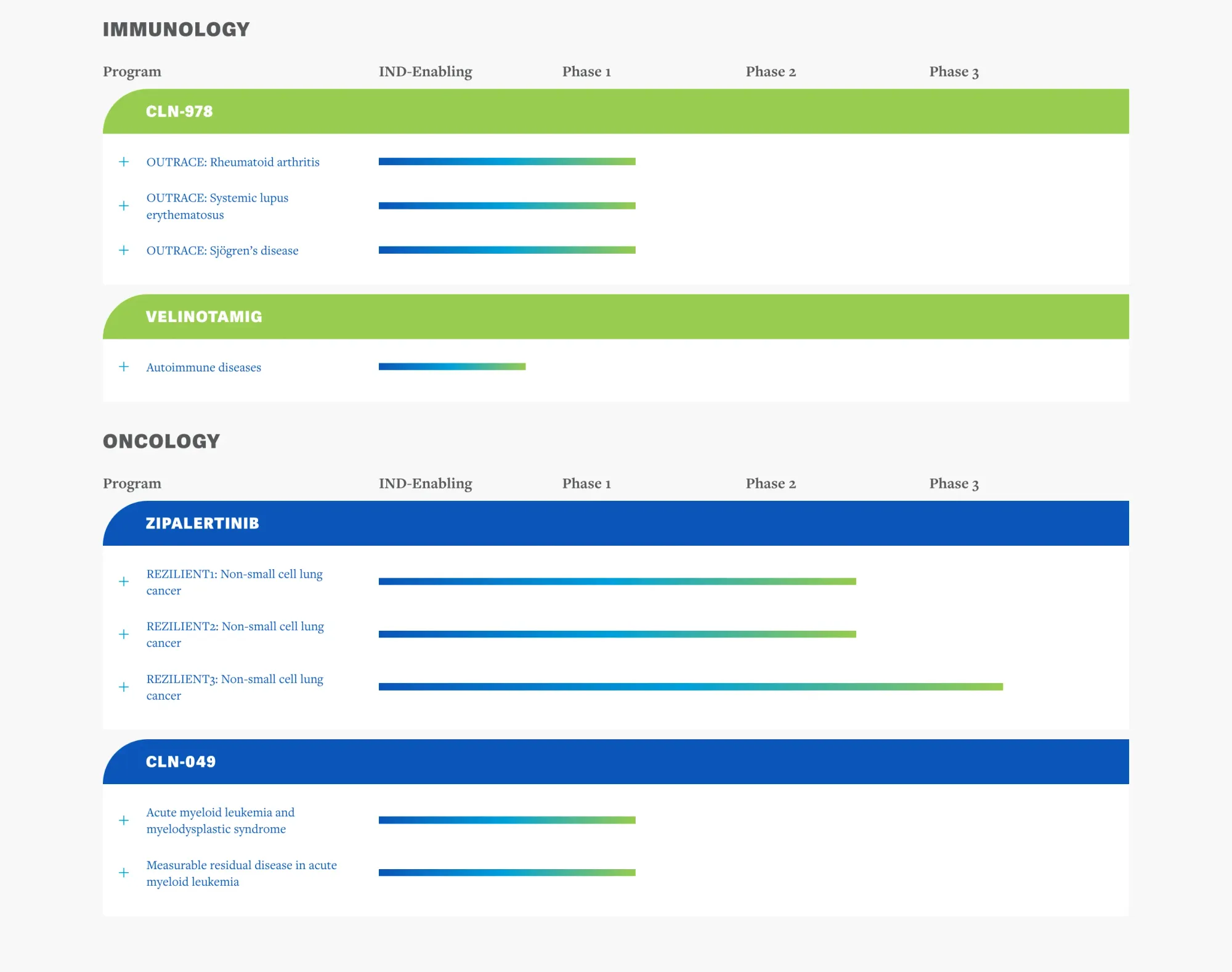
FDA Breakthrough: New Non-Invasive Biomarker to Revolutionize MASH Drug Development
The FDA accepted non-invasive Liver Stiffness Measurement as a surrogate endpoint for MASH clinical trials, replacing biopsies. This key decision accelerates MASH drug development by enabling safer monitoring, speeding recruitment, and predicting patient outcomes.
This post is for paying subscribers only
Already have an account? Sign in.