Eton Pharmaceuticals Secures Rights to First Generic Treatment for Ultra-Rare Disease
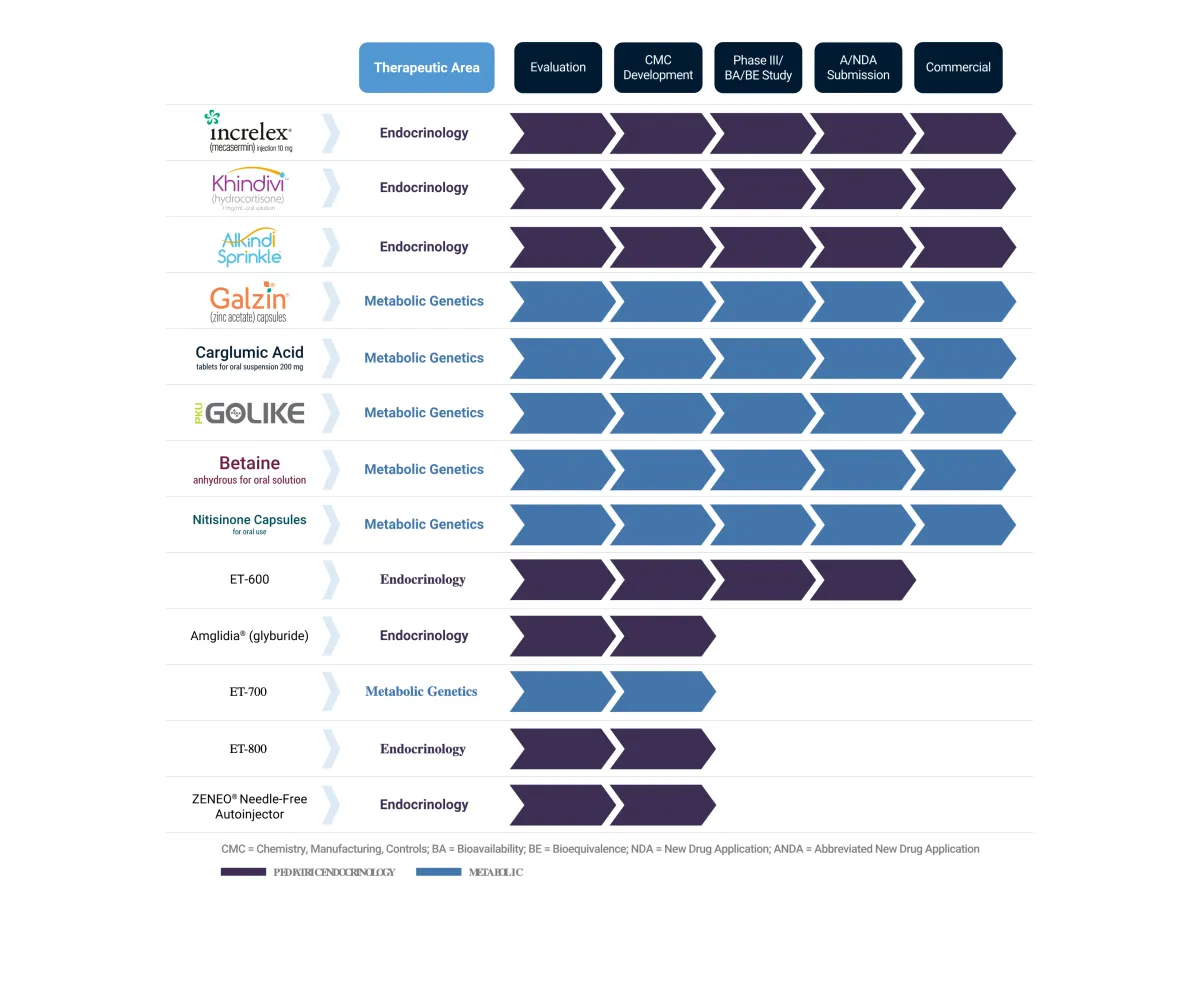
Eton Pharmaceuticals licenses U.S. rights to an ultra-rare disease treatment affecting under 100 patients, expecting FDA approval and mid-2026 launch as first generic alternative.
Already have an account? Sign in.